Science 10 - SharpSchool
... give the atom name for the first element (with the prefix if there is more than one) then give the name for the second element with “ide” ending and include the prefix Note: if the first element is hydrogen, do not put a prefix (these are acids!) Prefixes 1 = mono 2 = di 3 = tri Try These 1. CO( ...
... give the atom name for the first element (with the prefix if there is more than one) then give the name for the second element with “ide” ending and include the prefix Note: if the first element is hydrogen, do not put a prefix (these are acids!) Prefixes 1 = mono 2 = di 3 = tri Try These 1. CO( ...
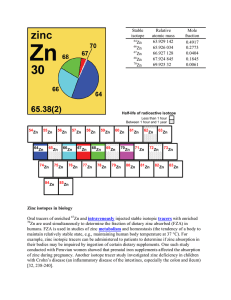
Zinc isotopes in biology Oral tracers of enriched Zn and
... slightly more than that of the proton. All atoms contain neutrons in their nucleus except for protium (1H). [return] positron – the antimatter counterpart of the electron, with a mass identical to that of the electron and an equal but opposite (positive) charge. positron emission tomography (PET) sc ...
... slightly more than that of the proton. All atoms contain neutrons in their nucleus except for protium (1H). [return] positron – the antimatter counterpart of the electron, with a mass identical to that of the electron and an equal but opposite (positive) charge. positron emission tomography (PET) sc ...
Chemical Reactions Chemical Arithmetic
... form an ionic compound (a salt) and water • Oxidation-Reduction reactions in which one or more electrons are transferred between reactants ...
... form an ionic compound (a salt) and water • Oxidation-Reduction reactions in which one or more electrons are transferred between reactants ...
Haley CHM2045 Final Review
... 2. A 1.0 L mixture of He, Ar, and Ne has a total pressure of 654 mmHg at 298 K. If the partial pressure of He is 378 mmHg and the partial pressure of Ne is 112 mmHg, what is the partial pressure of Ar? 3. Lithium reacts with nitrogen gas in the following reaction, 6Li + N2 —> 2Li3N What mass of lith ...
... 2. A 1.0 L mixture of He, Ar, and Ne has a total pressure of 654 mmHg at 298 K. If the partial pressure of He is 378 mmHg and the partial pressure of Ne is 112 mmHg, what is the partial pressure of Ar? 3. Lithium reacts with nitrogen gas in the following reaction, 6Li + N2 —> 2Li3N What mass of lith ...
Chemistry I Exam
... (2) LiOH + Na → NaOH + Li (3) NaCl + Li → LiCl + Na (4) NaNO3 + K → KNO3 + Na (5) 2 Al + 3 ZnCl2 → 2 AlCl3 + 3 Zn (6) 2 Ag + Pb(NO3)2 → 2 AgNO3 + Pb A. 1 and 6 ...
... (2) LiOH + Na → NaOH + Li (3) NaCl + Li → LiCl + Na (4) NaNO3 + K → KNO3 + Na (5) 2 Al + 3 ZnCl2 → 2 AlCl3 + 3 Zn (6) 2 Ag + Pb(NO3)2 → 2 AgNO3 + Pb A. 1 and 6 ...
File - Science with Mr. Louie
... In chemistry, we often use numbers that are either very large (1 mole = 602 200 000 000 000 000 000 000 atoms) or very small (the mass of an electron = 0.000 000 000 000 000 000 000 000 000 000 910 939 kg). Writing numbers with so many digits would be tedious and difficult. To make writing very larg ...
... In chemistry, we often use numbers that are either very large (1 mole = 602 200 000 000 000 000 000 000 atoms) or very small (the mass of an electron = 0.000 000 000 000 000 000 000 000 000 000 910 939 kg). Writing numbers with so many digits would be tedious and difficult. To make writing very larg ...
Chemical Equations Balancing Chemical Equations Try One…
... This is called the skeleton equation. Try to read it like a sentence- Gaseous ammonia is added dd d to oxygen gas to produce d nitrogen i monoxide, id which is given off as a gas, and water -NOW BALANCE IT -by adding coefficients-DO NOT CHANGE SUBSCRIPTS 1. start at the formula with the highest subs ...
... This is called the skeleton equation. Try to read it like a sentence- Gaseous ammonia is added dd d to oxygen gas to produce d nitrogen i monoxide, id which is given off as a gas, and water -NOW BALANCE IT -by adding coefficients-DO NOT CHANGE SUBSCRIPTS 1. start at the formula with the highest subs ...
Scandium and Yttrium - Mercyhurst University
... coordination. Two compounds were particularly interesting, [Sc(CH2Si(CH3)3)3(12-crown-4)] (4) and Y(CH2Si(CH3)3)3(12-crown-4)] (5) (shown on the next page), because of their difference in coordination. ...
... coordination. Two compounds were particularly interesting, [Sc(CH2Si(CH3)3)3(12-crown-4)] (4) and Y(CH2Si(CH3)3)3(12-crown-4)] (5) (shown on the next page), because of their difference in coordination. ...
atom - Zanichelli online per la scuola
... Dalton’s law of multiple proportions The law of multiple proportions states that when two elements combine to give more than one compound, the masses of one element that combine with the fixed mass of the other are in a ratio of small whole numbers. ...
... Dalton’s law of multiple proportions The law of multiple proportions states that when two elements combine to give more than one compound, the masses of one element that combine with the fixed mass of the other are in a ratio of small whole numbers. ...
Dear Students, Welcome to AP Chemistry, a little early. We will have
... 4. Memorize the chemical symbols for elements on the periodic table (PT). For example, Mg represents Magnesium. You can use a PT on every assessment, but the name of the element is NOT listed. The periodic table that you will use all year and on the AP Chemistry Exam is provided in this packet. Also ...
... 4. Memorize the chemical symbols for elements on the periodic table (PT). For example, Mg represents Magnesium. You can use a PT on every assessment, but the name of the element is NOT listed. The periodic table that you will use all year and on the AP Chemistry Exam is provided in this packet. Also ...
E:\My Documents\sch3u\SCH3Ureview.wpd
... 6) If you had 6.5 g of liquid Nitrogen and allowed it to fill a balloon as Nitrogen gas: a) How many Nitrogen molecules would you have? b) How many Nitrogen atoms would you have? c) Why are these two numbers not the same? 7) What is the percent composition by mass of the compounds: a) H2O b) CO2 c) ...
... 6) If you had 6.5 g of liquid Nitrogen and allowed it to fill a balloon as Nitrogen gas: a) How many Nitrogen molecules would you have? b) How many Nitrogen atoms would you have? c) Why are these two numbers not the same? 7) What is the percent composition by mass of the compounds: a) H2O b) CO2 c) ...
RXN-4-STUDENTS - Rothschild Science
... element you have NH3 (one nitrogen, three hydrogen)- DON’T mess with these!! Coefficients – small whole number that appears ...
... element you have NH3 (one nitrogen, three hydrogen)- DON’T mess with these!! Coefficients – small whole number that appears ...
in-class assignment - hrsbstaff.ednet.ns.ca
... see and hear it as titanium dioxide. So far the ions I've shown were of single atoms, for example, Na+, K+, Ti4+, Cl-, and O2-. However, a large number of compounds have ions that are made from two or more elements. They call them polyatomic ions. "Poly" meaning "many" and "atomic" referring to atom ...
... see and hear it as titanium dioxide. So far the ions I've shown were of single atoms, for example, Na+, K+, Ti4+, Cl-, and O2-. However, a large number of compounds have ions that are made from two or more elements. They call them polyatomic ions. "Poly" meaning "many" and "atomic" referring to atom ...
Writing And Balancing Equations
... On the left, Zn and HCl are reactants On the right, ZnCl2 and H2 are products The arrow () means “yields” The up arrow () indicated a gas product The 2 in front of HCl is a coefficient The small 2 to the right of H is a subscript ...
... On the left, Zn and HCl are reactants On the right, ZnCl2 and H2 are products The arrow () means “yields” The up arrow () indicated a gas product The 2 in front of HCl is a coefficient The small 2 to the right of H is a subscript ...
Electrochemistry
... Rules for Assigning Oxidation Numbers (In order of priority): 1. The oxidation number of any pure element is _________. 2. The oxidation number of a monatomic ion is __________ to its charge. 3. The ______ of the oxidation numbers in a compound is zero if ____________, or equal to the ___________ if ...
... Rules for Assigning Oxidation Numbers (In order of priority): 1. The oxidation number of any pure element is _________. 2. The oxidation number of a monatomic ion is __________ to its charge. 3. The ______ of the oxidation numbers in a compound is zero if ____________, or equal to the ___________ if ...
Midterm Review Answers
... sodium fluoride, and sodium hydroxide you need to separate the barium, mercury(II), and magnesium ions. How would you go about separating these ions? Discuss your experimental procedure and defend your answer. Using the solubility rules, chloride compounds are generally soluble, but mercury is an ex ...
... sodium fluoride, and sodium hydroxide you need to separate the barium, mercury(II), and magnesium ions. How would you go about separating these ions? Discuss your experimental procedure and defend your answer. Using the solubility rules, chloride compounds are generally soluble, but mercury is an ex ...
2016-2017 Summer Assignment AP Chem 2017 Summer
... d. Nitrogen e. Helium f. Krypton g. Fluorine ...
... d. Nitrogen e. Helium f. Krypton g. Fluorine ...
2014MSC(ORGANIC(CHEMISTRY!
... ! Polar!covalent!bonds!are!formed!through!the!sharing!of!electrons!between!neutral!atoms!–! it!is!polar!where!the!electrons!are!attracted!stronger!to!one!atom!over!the!other.!! ! Therefore,!the!electron!distribution!between!the!atoms!is!not!symmetrical,!and!atoms!have! a!partial!negative!or!positive ...
... ! Polar!covalent!bonds!are!formed!through!the!sharing!of!electrons!between!neutral!atoms!–! it!is!polar!where!the!electrons!are!attracted!stronger!to!one!atom!over!the!other.!! ! Therefore,!the!electron!distribution!between!the!atoms!is!not!symmetrical,!and!atoms!have! a!partial!negative!or!positive ...
Chemistry Final Exam Review 2013
... 57. Which equation is correctly balanced? a. Cu + H2SO4 CuSO4 + H2O + SO2 b. 2Na + 2H2O 2NaOH+ H2 c. 2Fe + 3O2 Fe2O3 d. 4Cu + S8 8Cu2S 58. What coefficients are required to balance this equation? __Fe2O3 + __CO __Fe + __CO2 a. 2, 6, 3,6 b. 1, 3, 2, 3 c. 1, 1, 2, 2 d. 1, 1, 2, 1 59. Which ex ...
... 57. Which equation is correctly balanced? a. Cu + H2SO4 CuSO4 + H2O + SO2 b. 2Na + 2H2O 2NaOH+ H2 c. 2Fe + 3O2 Fe2O3 d. 4Cu + S8 8Cu2S 58. What coefficients are required to balance this equation? __Fe2O3 + __CO __Fe + __CO2 a. 2, 6, 3,6 b. 1, 3, 2, 3 c. 1, 1, 2, 2 d. 1, 1, 2, 1 59. Which ex ...
The Chemical Context of Life PPT
... Electron Distribution and Chemical Properties • The chemical behavior of an atom is determined by the distribution of electrons in electron energy levels and sublevels • The periodic table of the elements shows the electron distribution for each element—think of it as a giant BINGO card ...
... Electron Distribution and Chemical Properties • The chemical behavior of an atom is determined by the distribution of electrons in electron energy levels and sublevels • The periodic table of the elements shows the electron distribution for each element—think of it as a giant BINGO card ...