Cleaning Up With Atom Economy
... students. Be sure the students understand how to calculate molecular weights and use stoichiometric relationships. The calculations are a little tricky since there are three moles of sodium hydroxide and soap per one mole of triglyceride. You may wish to minimize the time involved by providing the m ...
... students. Be sure the students understand how to calculate molecular weights and use stoichiometric relationships. The calculations are a little tricky since there are three moles of sodium hydroxide and soap per one mole of triglyceride. You may wish to minimize the time involved by providing the m ...
NAME…………… - Kcse Online
... Delivery tube is removed from the water before heating is stopped at the end of the experiment. (1mk) ______________________________________________________________________ ______________________________________________________________________ ________________________________________________________ ...
... Delivery tube is removed from the water before heating is stopped at the end of the experiment. (1mk) ______________________________________________________________________ ______________________________________________________________________ ________________________________________________________ ...
SrF 2(s)
... to go from name to formula: first part is the same as before…look up the symbol for each ion then balance the charges using subscripts, then for the hydrate part…add “ x H2O” where x is the number given in the prefix Examples: iron (III) nitrate nonahydrate = sodium chlorate tetrahydrate = nickel ( ...
... to go from name to formula: first part is the same as before…look up the symbol for each ion then balance the charges using subscripts, then for the hydrate part…add “ x H2O” where x is the number given in the prefix Examples: iron (III) nitrate nonahydrate = sodium chlorate tetrahydrate = nickel ( ...
IPC – First Semester Exam Review Be able to classify an example
... o Valence electrons determine an element’s reactivity Elements in the same group have similar reactivity since they have the same number of valence electrons. Reactivity is how elements and compounds react to other substances. o Noble gases are nonreactive (inert) because their valence energy leve ...
... o Valence electrons determine an element’s reactivity Elements in the same group have similar reactivity since they have the same number of valence electrons. Reactivity is how elements and compounds react to other substances. o Noble gases are nonreactive (inert) because their valence energy leve ...
Unit 1 Review, pages 138–145
... (d) The chemical formula for phosphorous trichloride is PCl3. (e) The chemical formula for dinitrogen tetraoxide is N2O4. 43. The two sources of most molecular compounds are living things (renewable resources) and fossil fuels (non-renewable resources). 44. Reusing an object means repurposing the ob ...
... (d) The chemical formula for phosphorous trichloride is PCl3. (e) The chemical formula for dinitrogen tetraoxide is N2O4. 43. The two sources of most molecular compounds are living things (renewable resources) and fossil fuels (non-renewable resources). 44. Reusing an object means repurposing the ob ...
2 - TEST BANK 360
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
File - Meissnerscience.com
... Hydrated Ionic Compounds A ______ is an _______ compound that has ___________ from a water solution and has _____ molecules incorporated into their crystal structure. Hydrates have a ________________ of water molecules ___________ bonded to each _________________. Compounds that have no water molec ...
... Hydrated Ionic Compounds A ______ is an _______ compound that has ___________ from a water solution and has _____ molecules incorporated into their crystal structure. Hydrates have a ________________ of water molecules ___________ bonded to each _________________. Compounds that have no water molec ...
Unit A Remediation Review
... a) Sulfuric acid is neutralized by sodium hydroxide to produce sodium sulfate and water. b) When propane (C3H10) is burned, carbon dioxide and water are formed. c) Zinc metal reacts with hydrochloric acid to produce hydrogen gas and zinc oxide. d) Sodium metal will react with oxygen to form sodium o ...
... a) Sulfuric acid is neutralized by sodium hydroxide to produce sodium sulfate and water. b) When propane (C3H10) is burned, carbon dioxide and water are formed. c) Zinc metal reacts with hydrochloric acid to produce hydrogen gas and zinc oxide. d) Sodium metal will react with oxygen to form sodium o ...
physical setting chemistry
... (1) They have identical molecular and identical properties. (2) They have identical molecular and different properties. (3) They have different molecular and identical properties. (4) They have different molecular and different properties. ...
... (1) They have identical molecular and identical properties. (2) They have identical molecular and different properties. (3) They have different molecular and identical properties. (4) They have different molecular and different properties. ...
classification of chemical reactions
... Example: rusting of iron burning of wood Physical change a change that does not produce a new substance a change in appearance or state Example: chopping wood melting ice Evidence of chemical reactions ...
... Example: rusting of iron burning of wood Physical change a change that does not produce a new substance a change in appearance or state Example: chopping wood melting ice Evidence of chemical reactions ...
Document
... Pure substances whose molecules are composed of atoms of different elements in fixed ratios Are represented by formulas Molecular formula-symbols and subscripts are used to represent elements and their ratios in each molecule of a compound Structural formula-shows how the atoms are connected in a ...
... Pure substances whose molecules are composed of atoms of different elements in fixed ratios Are represented by formulas Molecular formula-symbols and subscripts are used to represent elements and their ratios in each molecule of a compound Structural formula-shows how the atoms are connected in a ...
File
... • To balance an equation means to change the numbers of each molecule involved, so that the same number of atoms of each element appear on the reactants side and on the products side. • Chemical equations balance on an atomic level, not molecular. • You cannot change the formula of a substance, i.e. ...
... • To balance an equation means to change the numbers of each molecule involved, so that the same number of atoms of each element appear on the reactants side and on the products side. • Chemical equations balance on an atomic level, not molecular. • You cannot change the formula of a substance, i.e. ...
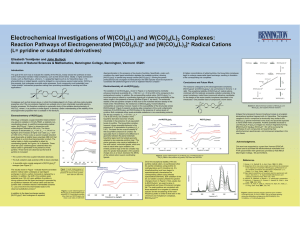
Electrochemical Investigations of W(CO) (L) and W(CO) (L) Complexes:
... active multinuclear transition metal complexes, such as that shown below. Ideally, a highly luminescent moeity, such as Re(CO)3(LL), where LL = a polypyridyl ligand such as 2,2’-bipyridine (bpy), 1,10phenanthroline or related ligands, would be bridged to a non-emissive second metal center, W(CO)5 in ...
... active multinuclear transition metal complexes, such as that shown below. Ideally, a highly luminescent moeity, such as Re(CO)3(LL), where LL = a polypyridyl ligand such as 2,2’-bipyridine (bpy), 1,10phenanthroline or related ligands, would be bridged to a non-emissive second metal center, W(CO)5 in ...
Chapter 2 Matter and Components F11 110
... 3. All atoms of an element have the same number of protons and electrons, which determines the chemical behavior of the element. Isotopes of an element differ in the number of neutrons, and thus in mass number. A sample of the element is treated as though its atoms have an average mass. ...
... 3. All atoms of an element have the same number of protons and electrons, which determines the chemical behavior of the element. Isotopes of an element differ in the number of neutrons, and thus in mass number. A sample of the element is treated as though its atoms have an average mass. ...
Chapter 2 Matter and Components F11 110pt
... The Modern Reassessment of the Atomic Theory 1. All matter is composed of atoms. The atom is the smallest body that retains the unique identity of the element. 2. Atoms of one element cannot be converted into atoms of another element in a chemical reaction. Elements can only be converted into other ...
... The Modern Reassessment of the Atomic Theory 1. All matter is composed of atoms. The atom is the smallest body that retains the unique identity of the element. 2. Atoms of one element cannot be converted into atoms of another element in a chemical reaction. Elements can only be converted into other ...
A2 Chemistry key word list
... The enthalpy change that takes place when one mole of gaseous atoms forms from the element in its standard state. ...
... The enthalpy change that takes place when one mole of gaseous atoms forms from the element in its standard state. ...
AP Chemistry Summer Assignment Summer 2015 Ms. Osquist
... (a) Al(s) + Cl2(g) AlCl3(s) (b) C2H4(g) + O2(g) CO2(g) + H2O(g) (c) Li(s) + N2(g) Li3N(s) (d) PbCO3(s) PbO(s) + CO2(g) (e) C7H8O2(l) + O2(g) CO2(g) + H2O(g) 4.4 Write a balanced chemical equation and net-ionic equation for the precipitation reaction that occurs when the following aqueous s ...
... (a) Al(s) + Cl2(g) AlCl3(s) (b) C2H4(g) + O2(g) CO2(g) + H2O(g) (c) Li(s) + N2(g) Li3N(s) (d) PbCO3(s) PbO(s) + CO2(g) (e) C7H8O2(l) + O2(g) CO2(g) + H2O(g) 4.4 Write a balanced chemical equation and net-ionic equation for the precipitation reaction that occurs when the following aqueous s ...
Help us improve Wikipedia by supporting it financially
... [edit] Nomenclature The naming of elements precedes the atomic theory of matter, although at the time it was not known which chemicals were elements and which compounds. When it was learned, existing names (e.g., gold, mercury, iron) were kept in most countries, and national differences emerged over ...
... [edit] Nomenclature The naming of elements precedes the atomic theory of matter, although at the time it was not known which chemicals were elements and which compounds. When it was learned, existing names (e.g., gold, mercury, iron) were kept in most countries, and national differences emerged over ...
A Review of High School Chemistry
... Here are some ideas of DALTON’S ATOMIC THEORY: 1. An element is composed of small indivisible particles called ATOMS. 2. All atoms of a given element have identical properties which are unique from other elements. 3. Atoms cannot be created, destroyed or converted into other elements. 4. MOLECULES a ...
... Here are some ideas of DALTON’S ATOMIC THEORY: 1. An element is composed of small indivisible particles called ATOMS. 2. All atoms of a given element have identical properties which are unique from other elements. 3. Atoms cannot be created, destroyed or converted into other elements. 4. MOLECULES a ...
Unit 2.2 Test Review Key
... 8.5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances 8.5F recognize whether a chemical equation containing coefficients is balanced or not and how that relates to the law of conservation of ...
... 8.5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances 8.5F recognize whether a chemical equation containing coefficients is balanced or not and how that relates to the law of conservation of ...
Fall Final Review Honors
... STRATEGY: Start by reading through your notes to refresh your memory on these topics. Then, use this review sheet as a starting point to identify the areas on which you need to spend more study time. For those areas, go back to homework assignments, quizzes, and reviews to practice more problems. I ...
... STRATEGY: Start by reading through your notes to refresh your memory on these topics. Then, use this review sheet as a starting point to identify the areas on which you need to spend more study time. For those areas, go back to homework assignments, quizzes, and reviews to practice more problems. I ...
1st semester exam review
... • Unknown element X has two isotopes. 6X has a 7.59 % abundance. 7X has a 92.41% abundance. What is the atomic mass of the unknown element? ...
... • Unknown element X has two isotopes. 6X has a 7.59 % abundance. 7X has a 92.41% abundance. What is the atomic mass of the unknown element? ...