Chemistry 221
... 1. Two compounds have formulas A2B6 and CD3. One of these compounds is an ionic compound and one a molecular one. How can you tell from the formulas which one is ionic? a) For the ionic compound, if the negative ion has a –1 charge, what is the charge of the positive ion? b) Suggest two ions that mi ...
... 1. Two compounds have formulas A2B6 and CD3. One of these compounds is an ionic compound and one a molecular one. How can you tell from the formulas which one is ionic? a) For the ionic compound, if the negative ion has a –1 charge, what is the charge of the positive ion? b) Suggest two ions that mi ...
test one
... 5. Given the equation dy = −y(y − 2)(y − 99), find each equilibrium (constant) solution and dt discuss the stability of each equilibrium solution. continue to page two ...
... 5. Given the equation dy = −y(y − 2)(y − 99), find each equilibrium (constant) solution and dt discuss the stability of each equilibrium solution. continue to page two ...
Intro to Chemical Equations note
... oxygen, or nitrogen are by themselves in an equation, they are shown as DIATOMIC ELEMENTS. H2 O2 N2 F2 Cl2 Br2 I2 ...
... oxygen, or nitrogen are by themselves in an equation, they are shown as DIATOMIC ELEMENTS. H2 O2 N2 F2 Cl2 Br2 I2 ...
Document
... UNIT QUESTION: How do I justify and solve the solution to a system of equations or inequalities? Standard: MCC9-12.A.REI.1, 3, 5, 6, and 12 ...
... UNIT QUESTION: How do I justify and solve the solution to a system of equations or inequalities? Standard: MCC9-12.A.REI.1, 3, 5, 6, and 12 ...
Naming Ionic Compounds
... e.g. calcium chloride…. write down CaCl o figure out the ionic charge of each element from the periodic table – normally metals have a positive charge and non metals have a negative charge e.g. Ca has a +2charge and Cl has a -1charge o add a number to the elements so that the total charge of the com ...
... e.g. calcium chloride…. write down CaCl o figure out the ionic charge of each element from the periodic table – normally metals have a positive charge and non metals have a negative charge e.g. Ca has a +2charge and Cl has a -1charge o add a number to the elements so that the total charge of the com ...
study guide first semester chemistry
... measurements accurate? Precise? Explain. ( You need to know the actual value to know if any measurements are accurate. They are not precise) 4. Identify the number of significant digits in each of the following measurements: a. 520mL (2) b. 0.0102ms (3) 5. Determine the volume of a box that measures ...
... measurements accurate? Precise? Explain. ( You need to know the actual value to know if any measurements are accurate. They are not precise) 4. Identify the number of significant digits in each of the following measurements: a. 520mL (2) b. 0.0102ms (3) 5. Determine the volume of a box that measures ...
Balancing Equations Notes
... 4. It is often easiest to start balancing with an element that appears only once on each side of the arrow. These elements must have the same coefficient. Next balance elements that appear only once on each side but have different numbers of atoms. Finally balance elements that are in two formulas i ...
... 4. It is often easiest to start balancing with an element that appears only once on each side of the arrow. These elements must have the same coefficient. Next balance elements that appear only once on each side but have different numbers of atoms. Finally balance elements that are in two formulas i ...
Math 1320, Section 10 Quiz IV Solutions 20 Points Please answer
... Now, we can simplify what’s under the root sign in both equations, because 9 is a perfect square which divides 27. Then ...
... Now, we can simplify what’s under the root sign in both equations, because 9 is a perfect square which divides 27. Then ...
Balancing Chemical Equations
... o we cannot change the formulas therefore we must change the number of molecules o we do this by adding coefficients in front of the formulas coefficient = number written in front to the chemical symbol or formula balanced chemical equation = when reactants & products contain an equal number of atom ...
... o we cannot change the formulas therefore we must change the number of molecules o we do this by adding coefficients in front of the formulas coefficient = number written in front to the chemical symbol or formula balanced chemical equation = when reactants & products contain an equal number of atom ...
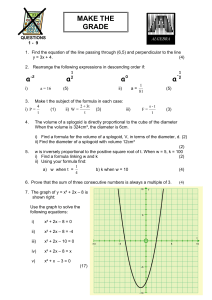
Find the equation of the line passing through (6,5) and
... can factorise quadratics using the difference of two square can factorise harder quadratics (a>1) can simplify and solve simple equations involving algebraic fractions can solve quadratic equations using the quadratic formula can solve simple quadratic equations using completing the square can sketc ...
... can factorise quadratics using the difference of two square can factorise harder quadratics (a>1) can simplify and solve simple equations involving algebraic fractions can solve quadratic equations using the quadratic formula can solve simple quadratic equations using completing the square can sketc ...
Chapter 7 - Midway ISD
... less oxygen gets –ite ending If more than 2 oxyanions, hypo- is added for less then the –ite and per- is added for more than –ate ...
... less oxygen gets –ite ending If more than 2 oxyanions, hypo- is added for less then the –ite and per- is added for more than –ate ...