Chemical Reactions
... • Because carbon has 4 valence electrons, each atom can form 4 bonds. • Carbon can combine in many ways with itself and other elements to form all living things. ...
... • Because carbon has 4 valence electrons, each atom can form 4 bonds. • Carbon can combine in many ways with itself and other elements to form all living things. ...
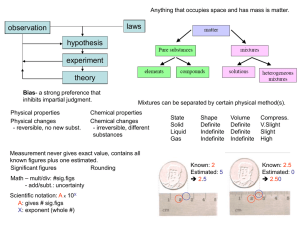
Chapter 2: Matter
... The raw materials that make up sand are transformed into something new when heated at very high temperatures. *Key Concept* ...
... The raw materials that make up sand are transformed into something new when heated at very high temperatures. *Key Concept* ...
File - Mr. Gittermann
... • Electrons: Subatomic particle with a negative charge found in a certain region of space around the nucleus called the electron cloud; kept close to the atom due to the attraction between the opposite charges of the electron and proton ...
... • Electrons: Subatomic particle with a negative charge found in a certain region of space around the nucleus called the electron cloud; kept close to the atom due to the attraction between the opposite charges of the electron and proton ...
Chapter 3 Discovering the atom and subatomic particles (History of
... 3.4 The electron was the first subatomic particle discovered Electron was found when Joseph John Thomson and Robert Millikan studied the cathode ray tube. They found that the cathode ray particles were much lighter than hydrogen atoms, indicating that the atom was not the smallest particle of matte ...
... 3.4 The electron was the first subatomic particle discovered Electron was found when Joseph John Thomson and Robert Millikan studied the cathode ray tube. They found that the cathode ray particles were much lighter than hydrogen atoms, indicating that the atom was not the smallest particle of matte ...
Chapter 3 Discovering the atom and subatomic particles (History of
... 3.4 The electron was the first subatomic particle discovered Electron was found when Joseph John Thomson and Robert Millikan studied the cathode ray tube. They found that the cathode ray particles were much lighter than hydrogen atoms, indicating that the atom was not the smallest particle of matte ...
... 3.4 The electron was the first subatomic particle discovered Electron was found when Joseph John Thomson and Robert Millikan studied the cathode ray tube. They found that the cathode ray particles were much lighter than hydrogen atoms, indicating that the atom was not the smallest particle of matte ...
Chemistry 102B What`s in an atom? Before “Chemistry” Other Early
... • John Dalton (early 1800s) – noted that elements that combined to form more than one kind of compound, did so in proportions of integers by mass. “Law of Multiple Proportions” ...
... • John Dalton (early 1800s) – noted that elements that combined to form more than one kind of compound, did so in proportions of integers by mass. “Law of Multiple Proportions” ...
chapters 1-4
... A compound is a distinct substance that contains two or more elements combined in a definite proportion by weight. Compounds can be decomposed chemically into simpler substances – that is, into simpler compounds or elements. ...
... A compound is a distinct substance that contains two or more elements combined in a definite proportion by weight. Compounds can be decomposed chemically into simpler substances – that is, into simpler compounds or elements. ...
- Lexington JHS
... Elements-found on the periodic table An element is a pure substance that cannot be broken down into other substances. (made of only one type of atom) Ex. C, H, O, S, or Fe ...
... Elements-found on the periodic table An element is a pure substance that cannot be broken down into other substances. (made of only one type of atom) Ex. C, H, O, S, or Fe ...
The Two-Body Problem
... • For a two particle system, it is a point always on the line joining the two particles. • The barycenter is closer to the heavier of the two masses. ...
... • For a two particle system, it is a point always on the line joining the two particles. • The barycenter is closer to the heavier of the two masses. ...
Chemical Reaction Basics
... Advanced Chemistry – Chapter 8 A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
... Advanced Chemistry – Chapter 8 A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
File - Mr. Sault`s Classroom
... atom we have. Each molecule has 1 Al and 3 Cl We have to multiply that by the coefficient to get the total # of atoms. 1x3 = 3 Al 3x3 = 9 Cl ...
... atom we have. Each molecule has 1 Al and 3 Cl We have to multiply that by the coefficient to get the total # of atoms. 1x3 = 3 Al 3x3 = 9 Cl ...
Atoms, compounds and elements - Mrs. Tes de Luna`s Science Class
... ◦ The first part of his theory states that all matter is made of atoms, which are indivisible. ◦ The second part of the theory says all atoms of a given element are identical in mass and properties. ◦ The third part says compounds are combinations of two or more different types of atoms. ◦ The fourt ...
... ◦ The first part of his theory states that all matter is made of atoms, which are indivisible. ◦ The second part of the theory says all atoms of a given element are identical in mass and properties. ◦ The third part says compounds are combinations of two or more different types of atoms. ◦ The fourt ...
Atoms - Red Hook Central Schools
... 400 b.c. Greeks • Greeks philosophers ponder the nature of matter: what is it made of? • Democritus: basic particle of matter = “atom” which means “indivisble”. Envisions these to be “hard spheres” • Aristotle: does not believe in atoms ...
... 400 b.c. Greeks • Greeks philosophers ponder the nature of matter: what is it made of? • Democritus: basic particle of matter = “atom” which means “indivisble”. Envisions these to be “hard spheres” • Aristotle: does not believe in atoms ...
ATOMS
... • Most matter is in the form of COMPOUNDS or mixtures of compounds. For example: salt (NaCl), water (H20), carbon dioxide (CO2) • Compounds have properties UNLIKE those of their elements. For example: Salt—Sodium (Na) is a shiny, soft, gray, explosive metal with water & Chlorine (Cl) is a yellowish- ...
... • Most matter is in the form of COMPOUNDS or mixtures of compounds. For example: salt (NaCl), water (H20), carbon dioxide (CO2) • Compounds have properties UNLIKE those of their elements. For example: Salt—Sodium (Na) is a shiny, soft, gray, explosive metal with water & Chlorine (Cl) is a yellowish- ...
Elementary my dear Watson review
... they form molecules. A molecule could be made up of atoms of the same kind. For example: O2 (oxygen) and 03 (ozone). A molecule could also be made up of different atoms. We often call these compounds. Here are some examples: CO2, H20, C6H1206 ...
... they form molecules. A molecule could be made up of atoms of the same kind. For example: O2 (oxygen) and 03 (ozone). A molecule could also be made up of different atoms. We often call these compounds. Here are some examples: CO2, H20, C6H1206 ...
Analysis of most common difficulties on Exam 1 last
... stronger the IM forces, the ___ the boiling point, or in which gas are forces strongest) Calculations involving the gas laws Density of gases Reading phase diagrams and understanding meaning of vapor pressure ...
... stronger the IM forces, the ___ the boiling point, or in which gas are forces strongest) Calculations involving the gas laws Density of gases Reading phase diagrams and understanding meaning of vapor pressure ...
Chemistry Final Study Guide
... 43. The three major categories of elements on the periodic table are the __________, __________, and __________. 44. The first group on the periodic table is called the __________ __________, and they are very reactive due to the fact that they tend to lose one __________. 45. Electrons in the outer ...
... 43. The three major categories of elements on the periodic table are the __________, __________, and __________. 44. The first group on the periodic table is called the __________ __________, and they are very reactive due to the fact that they tend to lose one __________. 45. Electrons in the outer ...
ChemicalBondingPowerpoint
... framework of organic compounds, and functional groups containing H, N, or O atoms bonded to C determine their behavior (Table 2.1). ...
... framework of organic compounds, and functional groups containing H, N, or O atoms bonded to C determine their behavior (Table 2.1). ...
Chapter 2 - Chemical Context of Life
... Hydrogen bonds occur when H is covalently bonded to an electronegative atom and attracted to another electronegative atom. Water ...
... Hydrogen bonds occur when H is covalently bonded to an electronegative atom and attracted to another electronegative atom. Water ...
EX3504-03
... Problem Six (15 marks) a. Explain the reason why Hartree-Fock fails for H2 when the internuclear distance is large. b. List three commonly used quantum chemistry methods that take into account of the electron correlation. c. If we want to calculate the ground state energy of a small molecule accurat ...
... Problem Six (15 marks) a. Explain the reason why Hartree-Fock fails for H2 when the internuclear distance is large. b. List three commonly used quantum chemistry methods that take into account of the electron correlation. c. If we want to calculate the ground state energy of a small molecule accurat ...
CHEMISTRY
... become IONS- they have a charge. Ex. a Hydrogen atom that loses it’s electron is called a hydrogen ion, H+, or a proton. ...
... become IONS- they have a charge. Ex. a Hydrogen atom that loses it’s electron is called a hydrogen ion, H+, or a proton. ...
Activity 2 - The Bohr Atom
... Experiments by Geiger and Marsden, the ‘Gold-foil’ or ‘Rutherford’ experiment showed that almost all the mass and positive charge of the nucleus was concentrated into a tiny sphere at the centre of the atom, which prompted Rutherford’s planetary model, of electrons orbiting the nucleus, like planets ...
... Experiments by Geiger and Marsden, the ‘Gold-foil’ or ‘Rutherford’ experiment showed that almost all the mass and positive charge of the nucleus was concentrated into a tiny sphere at the centre of the atom, which prompted Rutherford’s planetary model, of electrons orbiting the nucleus, like planets ...