Exam #: Printed Name: Signature: PHYSICS DEPARTMENT
... Begin each answer on the same page as the question, but continue on additional blank pages if necessary. Write only on one side of each page. Each page should contain work related to only one problem. When you start a new problem, start a new page. If you need to leave your seat, wait until everyone ...
... Begin each answer on the same page as the question, but continue on additional blank pages if necessary. Write only on one side of each page. Each page should contain work related to only one problem. When you start a new problem, start a new page. If you need to leave your seat, wait until everyone ...
Exam #: Printed Name: Signature: PHYSICS DEPARTMENT
... In the Einstein model for a solid, atoms are treated as one-dimensional quantum mechanical oscillators that can each accept an arbitrary number of energy units above the ground state. Recall that the multiplicity Ω(N, q) = (q + N − 1)!/(q!(N − 1)!) gives the number of states available to a system co ...
... In the Einstein model for a solid, atoms are treated as one-dimensional quantum mechanical oscillators that can each accept an arbitrary number of energy units above the ground state. Recall that the multiplicity Ω(N, q) = (q + N − 1)!/(q!(N − 1)!) gives the number of states available to a system co ...
Electrons in Atoms
... matter can gain or lose energy only in small, specific amounts called quanta Quantum – minimum amount of energy that can be gained or lost by an atom Equantum = hv where “E” is energy, “h” is Planck’s constant, and “v” is velocity Planck’s constant = 6.626x10-34 J·s Planck’s theory: for a give ...
... matter can gain or lose energy only in small, specific amounts called quanta Quantum – minimum amount of energy that can be gained or lost by an atom Equantum = hv where “E” is energy, “h” is Planck’s constant, and “v” is velocity Planck’s constant = 6.626x10-34 J·s Planck’s theory: for a give ...
Solutions
... b) Why did Thomson come up with, or devise, this particular model? {5 pts} c) Describe Ernest Rutherford’s model of the atom. {5 pts} d) Why did Rutherford come up with, or devise, this particular model? {5 pts} (a) Thomson’s model was called the PLUM PUDDING MODEL. It consisted of an amorphous blob ...
... b) Why did Thomson come up with, or devise, this particular model? {5 pts} c) Describe Ernest Rutherford’s model of the atom. {5 pts} d) Why did Rutherford come up with, or devise, this particular model? {5 pts} (a) Thomson’s model was called the PLUM PUDDING MODEL. It consisted of an amorphous blob ...
151b650e7a25cfd
... 4- Region D Vdc is so high that even a minimally-ionizing particle will produce a very large voltage pulse. The initial ionization produced by the radiation triggers a complete gas breakdown as an avalanche of electrons heads towards and spreads along the centre wire. This region is called the Geig ...
... 4- Region D Vdc is so high that even a minimally-ionizing particle will produce a very large voltage pulse. The initial ionization produced by the radiation triggers a complete gas breakdown as an avalanche of electrons heads towards and spreads along the centre wire. This region is called the Geig ...
Problem set 2
... Quantum Mechanics 1, Spring 2011 CMI Problem set 2 Due by the beginning of class on Friday January 21, 2011 g Classical motion for zero angular momentum in a − r potential Let us try to model a hydrogen atom as a simple classical mechanical system. It is assumed to have an infinitely heavy point-lik ...
... Quantum Mechanics 1, Spring 2011 CMI Problem set 2 Due by the beginning of class on Friday January 21, 2011 g Classical motion for zero angular momentum in a − r potential Let us try to model a hydrogen atom as a simple classical mechanical system. It is assumed to have an infinitely heavy point-lik ...
Quantum
... Example 1: What is the energy of a photon of yellow-green light (l = 555 nm)? First we find f from wave equation: c = fl ...
... Example 1: What is the energy of a photon of yellow-green light (l = 555 nm)? First we find f from wave equation: c = fl ...
PHYS-2020: General Physics II Problem Set 3, Spring 2017
... 8. At rest, a car’s horn sounds the note A (440 Hz). The horn is sounded while the car is moving down the street. A bicyclist moving in the same direction with one-third the car’s speed hears a frequency of 415 Hz. (a) Is the cyclist ahead or behind the car? (b) What is the speed of the car? 9. Two ...
... 8. At rest, a car’s horn sounds the note A (440 Hz). The horn is sounded while the car is moving down the street. A bicyclist moving in the same direction with one-third the car’s speed hears a frequency of 415 Hz. (a) Is the cyclist ahead or behind the car? (b) What is the speed of the car? 9. Two ...
chemistry in the 8th grade
... configuration. This would be 2 electrons in the first shell and 8 electrons in any shell after the first one. Atoms can undergo chemical reactions by losing, gaining, or sharing electrons to achieve this stable configuration. If atoms have 3 or fewer outer electrons, they can lose these electrons to ...
... configuration. This would be 2 electrons in the first shell and 8 electrons in any shell after the first one. Atoms can undergo chemical reactions by losing, gaining, or sharing electrons to achieve this stable configuration. If atoms have 3 or fewer outer electrons, they can lose these electrons to ...
Slide 1
... This appears to say that a photon of a certain wavelength has mass! Proved by Arthur Compton in 1922 E-M radiation is both waves & little packets of energy and matter called photons De Broglie 1923: if light has wave-particle duality, then matter, which is particle-like, must also be wavelike under ...
... This appears to say that a photon of a certain wavelength has mass! Proved by Arthur Compton in 1922 E-M radiation is both waves & little packets of energy and matter called photons De Broglie 1923: if light has wave-particle duality, then matter, which is particle-like, must also be wavelike under ...
HonorsChem.final.rev.probs
... 16. For the following reaction: 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) a) Determine the number of grams of KOH that will be produced when 97 g of potassium are used. b) Determine the number of liters of H2 gas that will be produced when 6.5 X 1024 molecules of water are reacted. ...
... 16. For the following reaction: 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) a) Determine the number of grams of KOH that will be produced when 97 g of potassium are used. b) Determine the number of liters of H2 gas that will be produced when 6.5 X 1024 molecules of water are reacted. ...
Two-particle systems
... This state means that if the spin of one particle is up, then the spin of the other particle must be down. Such state can not be separated into the product state as neither particle is in definite state of being spin up or spin down. Equation (1) above assumes that we can tell which particle is part ...
... This state means that if the spin of one particle is up, then the spin of the other particle must be down. Such state can not be separated into the product state as neither particle is in definite state of being spin up or spin down. Equation (1) above assumes that we can tell which particle is part ...
Compound vs Element chart
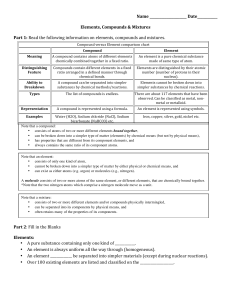
... Note that a compound: • consists of atoms of two or more different elements bound together, • can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), • has properties that are different from its component elements, and • always contains the same rat ...
... Note that a compound: • consists of atoms of two or more different elements bound together, • can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), • has properties that are different from its component elements, and • always contains the same rat ...
Transition metal configurations and limitations of the orbital
... The Hartree-Fock method provides a better orbital approximation since it includes the effect of inter-electronic repulsions, hut only does so in a time-averaged manner. This method fails to account for the instantaneous changes in the configuration of an atom. The energy contribution due to this eff ...
... The Hartree-Fock method provides a better orbital approximation since it includes the effect of inter-electronic repulsions, hut only does so in a time-averaged manner. This method fails to account for the instantaneous changes in the configuration of an atom. The energy contribution due to this eff ...
Document
... • The chemical characteristics of atoms are based upon the number of e- that are in the outer energy levels. • These are called valance electrons. They are only s and p sublevel e-. • For the sulfur atom, 1s2 2s2p6 3s2p4, there are 6 valence e-. ...
... • The chemical characteristics of atoms are based upon the number of e- that are in the outer energy levels. • These are called valance electrons. They are only s and p sublevel e-. • For the sulfur atom, 1s2 2s2p6 3s2p4, there are 6 valence e-. ...
The Wave
... And then there was a problem… However, in the early 20th century, several effects were observed which could not be understood using the wave theory of light. So other experiments were done and found it could behave as both:: 1) The Photo-Electric Effect (particle) 2) The Compton Effect (particle) 3 ...
... And then there was a problem… However, in the early 20th century, several effects were observed which could not be understood using the wave theory of light. So other experiments were done and found it could behave as both:: 1) The Photo-Electric Effect (particle) 2) The Compton Effect (particle) 3 ...
Bose-Einstein Condensation
... • Hot Li7 atoms, emitted from an oven at 800 K, form an atomic beam. • The atomic beam is slowed by an oppositely directed laser beam, and deflected by a second laser beam towards a magnetic and optical trap. • Another laser beam collimates the deflected atomic beam, and optically pumps it, so that ...
... • Hot Li7 atoms, emitted from an oven at 800 K, form an atomic beam. • The atomic beam is slowed by an oppositely directed laser beam, and deflected by a second laser beam towards a magnetic and optical trap. • Another laser beam collimates the deflected atomic beam, and optically pumps it, so that ...
Modern Physics Review
... the emission of photoelectrons. If the intensity of this radiation is increased, what will happen to the number of the emissions of ...
... the emission of photoelectrons. If the intensity of this radiation is increased, what will happen to the number of the emissions of ...
quantum-theory-of-the-atom2
... of both a particle and a wave, we can start to understand the emission spectra of atoms. One in particular, hydrogen (shown below) ...
... of both a particle and a wave, we can start to understand the emission spectra of atoms. One in particular, hydrogen (shown below) ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.