unit-4-notes-1_enthalpy-and-entropy
... the left (starting with reactants) or from the right (starting with products) Just a little term before we summarize: The word macroscopic means large scale or visible or observable. (The opposite is microscopic, which means too small to see eg. molecular level). Some macroscopic properties are tota ...
... the left (starting with reactants) or from the right (starting with products) Just a little term before we summarize: The word macroscopic means large scale or visible or observable. (The opposite is microscopic, which means too small to see eg. molecular level). Some macroscopic properties are tota ...
ap chemistry 2005/2006
... identification as physical or chemical change. Lab: Determining the Specific Heat of an Unknown Metal – the specific heat capacity of a nail will be experimentally determined by measuring the temperature change of water and of the nail after it has been heated to approximately 850 degrees Celsius in ...
... identification as physical or chemical change. Lab: Determining the Specific Heat of an Unknown Metal – the specific heat capacity of a nail will be experimentally determined by measuring the temperature change of water and of the nail after it has been heated to approximately 850 degrees Celsius in ...
AP Chemistry
... of solution spilled from the polystyrene cup before all of the solution than they are in CaCl2(s) solute dissolved. How does this affect the calculated value for the heat of solution of the ionic compound? (A) The calculated value is too large because less water was cooled as the remaining solute di ...
... of solution spilled from the polystyrene cup before all of the solution than they are in CaCl2(s) solute dissolved. How does this affect the calculated value for the heat of solution of the ionic compound? (A) The calculated value is too large because less water was cooled as the remaining solute di ...
- Academy Test Bank
... OBJ: Perform calculations using the mole. TOP: stoichiometry | mass and moles of substance 24. A sample of gallium(III) sulfite, Ga2(SO3)3, contains 1.95 mol of sulfite ions. The number of moles of gallium(III) ions in the sample is A) 1.30 mol. B) 2.92 mol. C) 1.95 mol. D) 5.84 mol. E) 3.90 mol. A ...
... OBJ: Perform calculations using the mole. TOP: stoichiometry | mass and moles of substance 24. A sample of gallium(III) sulfite, Ga2(SO3)3, contains 1.95 mol of sulfite ions. The number of moles of gallium(III) ions in the sample is A) 1.30 mol. B) 2.92 mol. C) 1.95 mol. D) 5.84 mol. E) 3.90 mol. A ...
Chemistry - Sanskriti School
... a) Availability of carbon dioxide b) Availability of sea forest c) Diffusion of gases into water d) Due to dissolved oxygen in water ...
... a) Availability of carbon dioxide b) Availability of sea forest c) Diffusion of gases into water d) Due to dissolved oxygen in water ...
ChemConnections
... same types of atoms in the same physical state, the more atoms per molecule, the more types of motion available to it and, thus, the higher its entropy. (d) 4 mol S2. The two samples contain the same number of sulfur atoms, but different numbers of molecules. Despite the greater complexity of S8 , t ...
... same types of atoms in the same physical state, the more atoms per molecule, the more types of motion available to it and, thus, the higher its entropy. (d) 4 mol S2. The two samples contain the same number of sulfur atoms, but different numbers of molecules. Despite the greater complexity of S8 , t ...
Bifunctional Asymmetric Catalysis: Cooperative Lewis Acid/Base
... to be the best cocatalyst across the board; ironically, indiumbased Lewis acids have infrequently been used in organic synthesis, and their coordination chemistry is not well-explored.17 One reason may be that InIII binds to many ligands reversibly and with comparatively low affinity.18 The most str ...
... to be the best cocatalyst across the board; ironically, indiumbased Lewis acids have infrequently been used in organic synthesis, and their coordination chemistry is not well-explored.17 One reason may be that InIII binds to many ligands reversibly and with comparatively low affinity.18 The most str ...
CHE 110 Dr. Nicholas Bizier Office DS 337b email
... Using a conversion factor-fraction whose numerator and denominator express the same quantity given in different units. ...
... Using a conversion factor-fraction whose numerator and denominator express the same quantity given in different units. ...
The Oxidation States of Tin
... Introduction: This lab sought to prepare the compounds tin(IV) iodide and tin(II) iodide. This experiment was performed so that an unusual characteristic of certain metals could be demonstrated. Some metals have the ability to maintain two or more oxidation states even though they are not a part of ...
... Introduction: This lab sought to prepare the compounds tin(IV) iodide and tin(II) iodide. This experiment was performed so that an unusual characteristic of certain metals could be demonstrated. Some metals have the ability to maintain two or more oxidation states even though they are not a part of ...
Chapter 8 - Cengage Learning
... Would you say ammonia (NH3) is mostly nitrogen or mostly hydrogen? Your answer depends on if you are looking at the number of atoms or the mass of the atoms. In terms of numbers of atoms, ammonia is ¾ hydrogen (there are four atoms making up an ammonia molecule, and three of them are hydrogen). But ...
... Would you say ammonia (NH3) is mostly nitrogen or mostly hydrogen? Your answer depends on if you are looking at the number of atoms or the mass of the atoms. In terms of numbers of atoms, ammonia is ¾ hydrogen (there are four atoms making up an ammonia molecule, and three of them are hydrogen). But ...
Series 9
... The objective of the present section is to illustrate the application of the conservation of energy principle to reacting systems. The forms of the conservation of energy principle introduced previously remain valid whether or not a chemical reaction occurs within the system. However, the methods us ...
... The objective of the present section is to illustrate the application of the conservation of energy principle to reacting systems. The forms of the conservation of energy principle introduced previously remain valid whether or not a chemical reaction occurs within the system. However, the methods us ...
Problem 1: A brief history of life in the universe
... of the planet’s atmosphere (which in turn depends on the distance from the sun, internal temperature, etc.), tectonic activity, and the existence of life. As the sun generated heat, light, and solar wind through nuclear fusion of hydrogen to helium, the primitive inner planets (Mercury, Venus, Earth ...
... of the planet’s atmosphere (which in turn depends on the distance from the sun, internal temperature, etc.), tectonic activity, and the existence of life. As the sun generated heat, light, and solar wind through nuclear fusion of hydrogen to helium, the primitive inner planets (Mercury, Venus, Earth ...
Chemical Technology - Engineers Institute of India
... 1. Chemical and allied Industry have first rank among all manufacturing industry both in capital assets and importance to the country economy. 2. Chemical Industry plays important role in every part of life. For example foods, drugs, petroleum, and fertilizer industry 3. Chemical Industry is differe ...
... 1. Chemical and allied Industry have first rank among all manufacturing industry both in capital assets and importance to the country economy. 2. Chemical Industry plays important role in every part of life. For example foods, drugs, petroleum, and fertilizer industry 3. Chemical Industry is differe ...
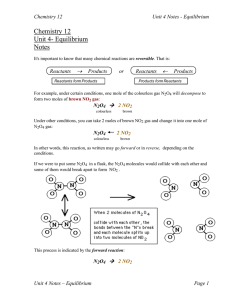
CHEMICAL EQUILIBRIUM (Part II)II)
... Constants Knowing the equilibrium constant (Kc or ) and/or the initial concentrations of KP) and/or the initial concentrations of reactants and products for a given reaction allows you to predict several features of that reaction, such as: o whether the reaction tends to occur or not. or not ...
... Constants Knowing the equilibrium constant (Kc or ) and/or the initial concentrations of KP) and/or the initial concentrations of reactants and products for a given reaction allows you to predict several features of that reaction, such as: o whether the reaction tends to occur or not. or not ...
3 - LPS
... Unit One Experiment – 2: Mass and Change ............................................ 49 Unit One Experiment – 3: Density and Relationships ............................... 53 Unit One Experiment – 4: Measurement and Uncertainty ......................... 58 ...
... Unit One Experiment – 2: Mass and Change ............................................ 49 Unit One Experiment – 3: Density and Relationships ............................... 53 Unit One Experiment – 4: Measurement and Uncertainty ......................... 58 ...
Chapter 1: conversion problems, metric system, significant figures
... liquid at 27 C. The heat for this process will involve a few steps – You will have to calculate the heat change from cooling the steam from 400.K to 373K (the heat capacity of steam is 2.0J/(K*g), then the heat of condensation using the latent heat of condensation of water which is 2.26kJ/g (the lat ...
... liquid at 27 C. The heat for this process will involve a few steps – You will have to calculate the heat change from cooling the steam from 400.K to 373K (the heat capacity of steam is 2.0J/(K*g), then the heat of condensation using the latent heat of condensation of water which is 2.26kJ/g (the lat ...
honors chemistry harvard-westlake second semester final exam
... a. How many moles of gas have been collected? b. Write the chemical reaction, including the correct numerical energy term on the appropriate side c. Assuming that the zinc has been completely consumed, how many grams of zinc were used in this reaction? d. If the resulting solution was evaporated, wh ...
... a. How many moles of gas have been collected? b. Write the chemical reaction, including the correct numerical energy term on the appropriate side c. Assuming that the zinc has been completely consumed, how many grams of zinc were used in this reaction? d. If the resulting solution was evaporated, wh ...
Gases - chemmybear.com
... would reduce the number (or frequency) of collisions of gas molecules with the surface of the balloon; [OR decrease the momentum change that occurs when the gas molecules strike the balloon surface] . In order to maintain a constant pressure vs the external pressure, the volume must decrease. (b) Th ...
... would reduce the number (or frequency) of collisions of gas molecules with the surface of the balloon; [OR decrease the momentum change that occurs when the gas molecules strike the balloon surface] . In order to maintain a constant pressure vs the external pressure, the volume must decrease. (b) Th ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.























