Chapter 3
... In Chapter 2, we saw the importance of relative numbers of atoms in the formation of compounds. We also learned how relative masses of atoms can be based on the arbitrary choice of the carbon-12 atom as a standard (Section 2.4). Now, we introduce a concept that enables us to deal with actual rather ...
... In Chapter 2, we saw the importance of relative numbers of atoms in the formation of compounds. We also learned how relative masses of atoms can be based on the arbitrary choice of the carbon-12 atom as a standard (Section 2.4). Now, we introduce a concept that enables us to deal with actual rather ...
Chemistry Review Module Chapter 1
... bit, for example, red iron oxide (rust) has Fe3+ ions, black iron oxide (wustite) contains Fe2+ ions. Note also, that most negative ions have the name ending changed to –ide. ...
... bit, for example, red iron oxide (rust) has Fe3+ ions, black iron oxide (wustite) contains Fe2+ ions. Note also, that most negative ions have the name ending changed to –ide. ...
Characterization of Multi-constituent Substances for REACH
... In addition to the actual analytical testing, it is important to make use of all the available information that might help to predict or define the theoretical composition of the substance. This should include details of the chemical process(es) involved in manufacture, the starting materials used, ...
... In addition to the actual analytical testing, it is important to make use of all the available information that might help to predict or define the theoretical composition of the substance. This should include details of the chemical process(es) involved in manufacture, the starting materials used, ...
1 - Grygla School
... Evidence that a chemical change may be happening generally falls into one of the categories described below and shown in Figure 5. The more of these signs you observe, the more likely a chemical change is taking place. But be careful! Some physical changes also have one or more of these signs. a. Th ...
... Evidence that a chemical change may be happening generally falls into one of the categories described below and shown in Figure 5. The more of these signs you observe, the more likely a chemical change is taking place. But be careful! Some physical changes also have one or more of these signs. a. Th ...
Core_Class_Science_Chemistry_for_the_web 838.3 KB
... Qualitative data is obtained through observations that describe color, smell, shape or other physical characteristics that is related to the 5 senses A hypothesis is a tentative explanation for what has been observed. Quantitative data is obtained from numerical observations that describe how much, ...
... Qualitative data is obtained through observations that describe color, smell, shape or other physical characteristics that is related to the 5 senses A hypothesis is a tentative explanation for what has been observed. Quantitative data is obtained from numerical observations that describe how much, ...
Chemistry booklet
... The mole concept gives us a means of managing fixed numbers of atoms / ions / molecules in ...
... The mole concept gives us a means of managing fixed numbers of atoms / ions / molecules in ...
National 5 - Deans Community High School
... Copy the graph showing reaction A and add the corresponding curves which could have been obtained for experiments B, C and D. (Label each curve clearly.) 18. The collision theory states that for two molecules to react, they must first collide with one another. Use the collision theory to explain the ...
... Copy the graph showing reaction A and add the corresponding curves which could have been obtained for experiments B, C and D. (Label each curve clearly.) 18. The collision theory states that for two molecules to react, they must first collide with one another. Use the collision theory to explain the ...
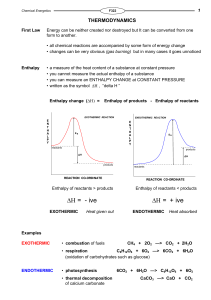
Enthalpy change
... for diatomic gases, the bond enthalpy is twice the enthalpy of atomisation the smaller the bond enthalpy, the weaker the bond and the easier it is to break Some mean bond enthalpies (in kJ mol-1) (values may differ slightly in other texts) H-H C-C C=C C≡C C-O C=O C-H C-N C-F C-Cl C-Br ...
... for diatomic gases, the bond enthalpy is twice the enthalpy of atomisation the smaller the bond enthalpy, the weaker the bond and the easier it is to break Some mean bond enthalpies (in kJ mol-1) (values may differ slightly in other texts) H-H C-C C=C C≡C C-O C=O C-H C-N C-F C-Cl C-Br ...
Enzymes: “Helper” Protein molecules
... Each enzyme is the specific helper to a specific reaction each enzyme needs to be the right shape for the job enzymes are named for the reaction they help ...
... Each enzyme is the specific helper to a specific reaction each enzyme needs to be the right shape for the job enzymes are named for the reaction they help ...
Stoichiometric Calculations
... Stoichiometry Calculations with Volumes At a given temperature and pressure, the space a sample of a gas takes up (it's volume) is proportional to the number of moles of gas molecules ...
... Stoichiometry Calculations with Volumes At a given temperature and pressure, the space a sample of a gas takes up (it's volume) is proportional to the number of moles of gas molecules ...
File
... Every chemical reaction obeys the Law of Conservation of Mass (aka the Law of Conservation of Matter). This states that matter is neither created nor destroyed. What this means to us is that whatever mass you start out with is the same amount of mass you end up with. For example, in the above equat ...
... Every chemical reaction obeys the Law of Conservation of Mass (aka the Law of Conservation of Matter). This states that matter is neither created nor destroyed. What this means to us is that whatever mass you start out with is the same amount of mass you end up with. For example, in the above equat ...
The Major Classes of Chemical Reactions
... • mole-mass-number conversions (Section 3.1) • molarity and mole-volume conversions (Section 3.5) • balancing chemical equations (Section 3.3) • calculating amounts of reactants and products (Section 3.4) ...
... • mole-mass-number conversions (Section 3.1) • molarity and mole-volume conversions (Section 3.5) • balancing chemical equations (Section 3.3) • calculating amounts of reactants and products (Section 3.4) ...
Experiment 15: Reduction and Oxidation of Organic Compounds
... Once you have determined that the reaction has gone to completion, add 30 mL of water to the flask, swirl and transfer the contents to a separatory funnel. Add 30 mL of diethyl ether to the sep funnel (use a little to rinse out the residue from the Erlenmeyer flask). Next, add 2 mL of saturated sodi ...
... Once you have determined that the reaction has gone to completion, add 30 mL of water to the flask, swirl and transfer the contents to a separatory funnel. Add 30 mL of diethyl ether to the sep funnel (use a little to rinse out the residue from the Erlenmeyer flask). Next, add 2 mL of saturated sodi ...
File - chemistryattweed
... 2. Calcium carbonate decomposes to calcium oxide and carbon dioxide. What mass of calcium carbonate is decomposed if 2.28L of carbon dioxide gas is collected at 25°C and 101 kPa. Identify natural and industrial sources of sulfur dioxide and oxides of nitrogen. ...
... 2. Calcium carbonate decomposes to calcium oxide and carbon dioxide. What mass of calcium carbonate is decomposed if 2.28L of carbon dioxide gas is collected at 25°C and 101 kPa. Identify natural and industrial sources of sulfur dioxide and oxides of nitrogen. ...
9/10/10 1 Chemistry 121: Atomic and Molecular Chemistry
... • ie., analyze samples of carbon dioxide gas different sources, find the same ratio by mass of carbon to oxygen. The law of multiple proportions: Two (or more) elements can combine to form more than one compound. Different compounds made up of the same elements differ in the number of atoms of each ...
... • ie., analyze samples of carbon dioxide gas different sources, find the same ratio by mass of carbon to oxygen. The law of multiple proportions: Two (or more) elements can combine to form more than one compound. Different compounds made up of the same elements differ in the number of atoms of each ...
chap-4-atomic-weights
... Lussac) by Amedeo Avogadro: At any given temperature and pressure, equal volumes of gases contain equal #s of particles. It’s worth trying to comprehend what a crazy notion this was: it seemed to mean that all gas particles (H2, O2, CH4, C2H6, C2H2, C3H8, etc) were the same size. But this sounds, ah ...
... Lussac) by Amedeo Avogadro: At any given temperature and pressure, equal volumes of gases contain equal #s of particles. It’s worth trying to comprehend what a crazy notion this was: it seemed to mean that all gas particles (H2, O2, CH4, C2H6, C2H2, C3H8, etc) were the same size. But this sounds, ah ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.