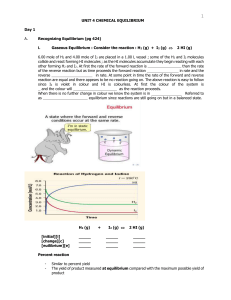
Unit 3: 1 Equilibrium and the Constant, K
... thermodynamics (a later unit). However, we can extract a piece of that unit for our current work, because we will study an experimentally measurable quantity called the equilibrium constant. In short, most reactions occur so as to produce products. A reaction with a large equilibrium constant procee ...
... thermodynamics (a later unit). However, we can extract a piece of that unit for our current work, because we will study an experimentally measurable quantity called the equilibrium constant. In short, most reactions occur so as to produce products. A reaction with a large equilibrium constant procee ...
Synthesis of Imidazolium Room-Temperature Ionic
... pure by 1H NMR. Several purification protocols can be utilized to remove colored impurities either at the stage of the bromide or later (10), if spectroscopic grade ionic liquids are desired. Carrying out the reaction in a solvent ensures that the resulting ionic liquid does not develop a brown colo ...
... pure by 1H NMR. Several purification protocols can be utilized to remove colored impurities either at the stage of the bromide or later (10), if spectroscopic grade ionic liquids are desired. Carrying out the reaction in a solvent ensures that the resulting ionic liquid does not develop a brown colo ...
Document
... magnitude of ΔH remains the same, but its sign changes. 2. When the balanced equation for a reaction is multiplied by an integer, the value of ΔH for that reaction must be multiplied by the same integer. ...
... magnitude of ΔH remains the same, but its sign changes. 2. When the balanced equation for a reaction is multiplied by an integer, the value of ΔH for that reaction must be multiplied by the same integer. ...
A Few Things You Might Want To Know
... Mixtures can be heterogeneous or homogeneous (= solutions). They consist of substances that can be separated by physical changes (distillation, crystallization, chromatography). Substances can be either elements or compounds. Compounds can be separated into elements by chemical changes (redox reacti ...
... Mixtures can be heterogeneous or homogeneous (= solutions). They consist of substances that can be separated by physical changes (distillation, crystallization, chromatography). Substances can be either elements or compounds. Compounds can be separated into elements by chemical changes (redox reacti ...
Document
... Check It Out! Example 2 Write the ratio 36 inches to 4 feet in simplest form. First convert feet to inches. 4 feet = 4 ● 12 inches = 48 inches ...
... Check It Out! Example 2 Write the ratio 36 inches to 4 feet in simplest form. First convert feet to inches. 4 feet = 4 ● 12 inches = 48 inches ...
WORD - SSS Chemistry
... Write a balanced chemical equation for each of the following, and classify each as synthesis, decomposition, single replacement, double replacement, neutralization or combustion. a) ...
... Write a balanced chemical equation for each of the following, and classify each as synthesis, decomposition, single replacement, double replacement, neutralization or combustion. a) ...
Unit 3: Thermochemistry
... the energy released when bonds form. ie. energy is absorbed exothermic reaction - the energy required to break bonds is less than the energy released when bonds form. ie. energy is produced ...
... the energy released when bonds form. ie. energy is absorbed exothermic reaction - the energy required to break bonds is less than the energy released when bonds form. ie. energy is produced ...
2 - TEST BANK 360
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
... Millikan performed a series of experiments in which he obtained the charge on the electron by observing how a charged drop of oil falls in the presence and in the absence of an electric field. An atomizer introduces a fine mist of oil drops into the top chamber (Figure 2.6). Several drops happen to ...
Reaction Kinetics - National Open University of Nigeria
... CHM 407: Reaction Kinetics concerns with the speed or rates of chemical reactions. The study of reaction rates allows for the prediction of how fast it will take a reaction mixture to reach equilibrium. It also account for how the reaction rate would be optimised by controlling certain factors such ...
... CHM 407: Reaction Kinetics concerns with the speed or rates of chemical reactions. The study of reaction rates allows for the prediction of how fast it will take a reaction mixture to reach equilibrium. It also account for how the reaction rate would be optimised by controlling certain factors such ...
Stoichiometric Calculations
... sides have the same number of each kind of atom. As a result, the Law of Conservation of Mass is obeyed, since no atoms can be created nor destroyed. Therefore, the quantities that are conserved in a balanced equation are mass and atoms. Conserved means that the amount that appears before the arrow ...
... sides have the same number of each kind of atom. As a result, the Law of Conservation of Mass is obeyed, since no atoms can be created nor destroyed. Therefore, the quantities that are conserved in a balanced equation are mass and atoms. Conserved means that the amount that appears before the arrow ...
Chemistry Challenge Problems
... was made by the German chemist Johann Wolfgang Döbereiner (1780–1849). In 1816, Döbereiner noticed that the then accepted atomic mass of strontium (50) was midway between the atomic masses of calcium (27.5) and barium (72.5). Note that the accepted atomic masses for these elements today are very dif ...
... was made by the German chemist Johann Wolfgang Döbereiner (1780–1849). In 1816, Döbereiner noticed that the then accepted atomic mass of strontium (50) was midway between the atomic masses of calcium (27.5) and barium (72.5). Note that the accepted atomic masses for these elements today are very dif ...
Exemplar Paper
... Ca(OCl)2(s) + 2HCl(aq) → Ca(OH)2(aq) + 2Cl2(g) alculate the mass of calcium hypochlorite that would be needed to C produce 0·096 litres of chlorine gas. (Take the molar volume of chlorine gas to be 24 litres mol−1.) ...
... Ca(OCl)2(s) + 2HCl(aq) → Ca(OH)2(aq) + 2Cl2(g) alculate the mass of calcium hypochlorite that would be needed to C produce 0·096 litres of chlorine gas. (Take the molar volume of chlorine gas to be 24 litres mol−1.) ...
chemistry-c7-what-you-should
... I in the context of the Haber process: a. I understand that the reaction between hydrogen and nitrogen to form ammonia is a reversible reaction b. I understand how the yield of ammonia is increased by recycling unreacted hydrogen and nitrogen c. I can explain the effect of changing temperature and p ...
... I in the context of the Haber process: a. I understand that the reaction between hydrogen and nitrogen to form ammonia is a reversible reaction b. I understand how the yield of ammonia is increased by recycling unreacted hydrogen and nitrogen c. I can explain the effect of changing temperature and p ...
Chemistry
... particle of matter. It translates to mean something that is indivisible. In the eighteenth century, chemist, John Dalton, revived the term when he suggested that each element was made up of unique atoms and the atoms of an element are all the same. At that time, there were about 35 known elements. T ...
... particle of matter. It translates to mean something that is indivisible. In the eighteenth century, chemist, John Dalton, revived the term when he suggested that each element was made up of unique atoms and the atoms of an element are all the same. At that time, there were about 35 known elements. T ...
Organic Chemistry II
... Organic synthesis is very important in that it allows the experimenter to make new compounds from compounds that might be more readily available. Sometimes a synthesis reaction is easy to do and other times great effort and care must be given. This experiment will require good technique as the cyclo ...
... Organic synthesis is very important in that it allows the experimenter to make new compounds from compounds that might be more readily available. Sometimes a synthesis reaction is easy to do and other times great effort and care must be given. This experiment will require good technique as the cyclo ...
1b-Redox FIB notes and practice
... This compound of sodium chloride consists of Na1+ ions and Cl1- ions. Thus, the oxidation number of sodium is +1 and of chlorine is -1. Notice that when writing oxidation numbers, the sign is put before the number. Sodium has an ionic charge of 1+ and an oxidation number of +1. In calcium fluoride, ...
... This compound of sodium chloride consists of Na1+ ions and Cl1- ions. Thus, the oxidation number of sodium is +1 and of chlorine is -1. Notice that when writing oxidation numbers, the sign is put before the number. Sodium has an ionic charge of 1+ and an oxidation number of +1. In calcium fluoride, ...
Chemistry Curriculum Map - Belle Vernon Area School District
... the mass number of an isotope and the average atomic mass of an element. Anchor: CHEM.A.2.3 – Explain how periodic trends in the properties of atoms allow for the prediction of physical and chemical properties. Eligible Content: CHEM.A.2.3.2 – Explain how the periodicity of chemical properties led t ...
... the mass number of an isotope and the average atomic mass of an element. Anchor: CHEM.A.2.3 – Explain how periodic trends in the properties of atoms allow for the prediction of physical and chemical properties. Eligible Content: CHEM.A.2.3.2 – Explain how the periodicity of chemical properties led t ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.