Snc2d Chapter 5 Practice Test
... 6. a) What are valence electrons? B) Why are they important? C) How can you determine the number of valence electrons for an atom from the periodic table (excluding the transition elements) 7. Draw the Lewis dot diagram for S ...
... 6. a) What are valence electrons? B) Why are they important? C) How can you determine the number of valence electrons for an atom from the periodic table (excluding the transition elements) 7. Draw the Lewis dot diagram for S ...
Chemistry - Solutions
... Determine which is more effective, temperature or motion, in increasing the rate of solubility of sugar in water. ...
... Determine which is more effective, temperature or motion, in increasing the rate of solubility of sugar in water. ...
CHEMICAL REACTIONS Chapter 4
... side. Compare those against the number of the atoms of the same element on the right side. 3. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in order to balance the equation. 4. Check your answer to see ...
... side. Compare those against the number of the atoms of the same element on the right side. 3. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in order to balance the equation. 4. Check your answer to see ...
Slide 1
... reaction; it shows the complete formulas of all reactants and products. • However, although this equation shows the reactants and products of the reaction, it does not give a very clear picture of what actually occurs in solution. • The complete ionic equation, better represents the actual forms of ...
... reaction; it shows the complete formulas of all reactants and products. • However, although this equation shows the reactants and products of the reaction, it does not give a very clear picture of what actually occurs in solution. • The complete ionic equation, better represents the actual forms of ...
Chemical Reactions - TSHSChemistry
... 2. Find the number of atoms for each element on the left side. Compare those against the number of the atoms of the same element on the right side. 3. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in o ...
... 2. Find the number of atoms for each element on the left side. Compare those against the number of the atoms of the same element on the right side. 3. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in o ...
All you need to know about Additional Science
... 3.5 Percentage yield Very few chemical reactions have a yield of 100% because: • Reaction is reversible • Some reactants produce unexpected products • Some products are left behind in apparatus • Reactants may not be completely pure • More than one product is produced and it may be difficult to sep ...
... 3.5 Percentage yield Very few chemical reactions have a yield of 100% because: • Reaction is reversible • Some reactants produce unexpected products • Some products are left behind in apparatus • Reactants may not be completely pure • More than one product is produced and it may be difficult to sep ...
Balancing Chemical Equations
... • Describe a chemical reaction by using a word equation and a formula equation. • Relate the conservation of mass to the rearrangement of atoms in a chemical reaction • Write and interpret a balanced chemical equation for a reaction, and relate conservation of mass to the balanced equation ...
... • Describe a chemical reaction by using a word equation and a formula equation. • Relate the conservation of mass to the rearrangement of atoms in a chemical reaction • Write and interpret a balanced chemical equation for a reaction, and relate conservation of mass to the balanced equation ...
MIDTERM REVIEW UNIT 1: Mass/Measurement
... formula if the percent composition is 47.37% carbon, 10.59% hydrogen and 42.04% oxygen and its molar mass is 76.09 g/mol? ...
... formula if the percent composition is 47.37% carbon, 10.59% hydrogen and 42.04% oxygen and its molar mass is 76.09 g/mol? ...
Chemistry I Final Review
... 52. How many grams of potassium bromide should be added to water to prepare 0.50 L of solution with a molarity of 0.125 M? ...
... 52. How many grams of potassium bromide should be added to water to prepare 0.50 L of solution with a molarity of 0.125 M? ...
Types of Chemical Reactions
... of a combination of carbon and hydrogen) to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. This reaction can be expressed as: ...
... of a combination of carbon and hydrogen) to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. This reaction can be expressed as: ...
gr11chemreview
... The following questions highlight the main knowledge and skills from grade 11 chemistry. A good understanding of the concepts covered in grade 11 chemistry is essential for success in grade 12 chemistry and you may need to do some independent review of some material if you do not have a clear unders ...
... The following questions highlight the main knowledge and skills from grade 11 chemistry. A good understanding of the concepts covered in grade 11 chemistry is essential for success in grade 12 chemistry and you may need to do some independent review of some material if you do not have a clear unders ...
+ H 2 SO 4(aq) - Rothschild Science
... element you have NH3 (one nitrogen, three hydrogen)- DON’T mess with these!! Coefficients – small whole number that appears ...
... element you have NH3 (one nitrogen, three hydrogen)- DON’T mess with these!! Coefficients – small whole number that appears ...
CP Chemistry Midterm Study Guide
... 20. What is the solution concentration of 25 g of NaCl mixed in 50 g of H2O? 21. How many grams of MgCl2 are in 500 mL of a 3.2 M solution? 22. How many atoms of sulfur do you have if you have 4 moles? 23. Convert 500 grams of magnesium to moles. 24. If we have 6.02x1023 molecules of oxygen, how man ...
... 20. What is the solution concentration of 25 g of NaCl mixed in 50 g of H2O? 21. How many grams of MgCl2 are in 500 mL of a 3.2 M solution? 22. How many atoms of sulfur do you have if you have 4 moles? 23. Convert 500 grams of magnesium to moles. 24. If we have 6.02x1023 molecules of oxygen, how man ...
Word Equations • a summary
... Use molecular building kits to look at the reaction of: Methane + CH4 ...
... Use molecular building kits to look at the reaction of: Methane + CH4 ...
Conservation of Mass Lab
... In this reaction, the aluminum sulfate looks the most complicated, so start there. Look at what happens with sulfate—since it remains sulfate on the right side of the reaction, treat it as a unit. You have three on the left side and only one on the right side, so place a coefficient of 3 in front of ...
... In this reaction, the aluminum sulfate looks the most complicated, so start there. Look at what happens with sulfate—since it remains sulfate on the right side of the reaction, treat it as a unit. You have three on the left side and only one on the right side, so place a coefficient of 3 in front of ...
Chemical Reactions and Stoichiometry
... b. Law of Conservation of Mass – atoms are neither created nor destroyed, they are just rearranged during a chemical reaction. i. Balanced Chemical Equations - each side of the equation has the same number of atoms (and, therefore, mass) of each element. Example: Burning solid carbon in the presence ...
... b. Law of Conservation of Mass – atoms are neither created nor destroyed, they are just rearranged during a chemical reaction. i. Balanced Chemical Equations - each side of the equation has the same number of atoms (and, therefore, mass) of each element. Example: Burning solid carbon in the presence ...
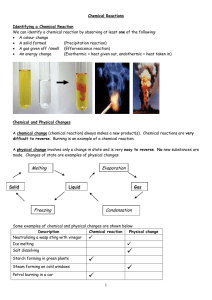
Chemical Reactions
... A word equation is a simple way of showing a chemical reaction. The reactants are shown on the left hand side and the products are shown on the right hand side. The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be a ...
... A word equation is a simple way of showing a chemical reaction. The reactants are shown on the left hand side and the products are shown on the right hand side. The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be a ...
Chemistry Chapter 12
... -with limiting reactants are talking about substances on the left hand side of the reaction equation -for this type of problem, must be given the quantity of two different reactants -the reactant that is completely consumed is called the limiting reactant -this substance is usually the more expensiv ...
... -with limiting reactants are talking about substances on the left hand side of the reaction equation -for this type of problem, must be given the quantity of two different reactants -the reactant that is completely consumed is called the limiting reactant -this substance is usually the more expensiv ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.