Learning Outcomes
... (g) define relative molecular mass, Mr, and calculate relative molecular mass (and relative formula mass) as the sum of relative atomic masses................................................................. 18 (h) calculate the percentage mass of an element in a compound when given appropriate info ...
... (g) define relative molecular mass, Mr, and calculate relative molecular mass (and relative formula mass) as the sum of relative atomic masses................................................................. 18 (h) calculate the percentage mass of an element in a compound when given appropriate info ...
reaction kinetics and mechanistic studies of nitric oxide
... kinetic aspects of this process, including feasibility, stoichiometry, reaction pathways and the effects of various process parameters [4]. This was followed by work on NO absorption by aqueous persulfate (Na2S2O8) activated by temperature. The use of persulfate was suggested to reduce the process c ...
... kinetic aspects of this process, including feasibility, stoichiometry, reaction pathways and the effects of various process parameters [4]. This was followed by work on NO absorption by aqueous persulfate (Na2S2O8) activated by temperature. The use of persulfate was suggested to reduce the process c ...
chemical equilibrium type 1
... As we increase the pressure of a gaseous system at equilibrium, either by decreasing the volume of the system or by adding more of the equilibrium mixture, we introduce a stress by increasing the number of molecules per unit of volume. In accordance with Le Chatelier's principle, a chemical reaction ...
... As we increase the pressure of a gaseous system at equilibrium, either by decreasing the volume of the system or by adding more of the equilibrium mixture, we introduce a stress by increasing the number of molecules per unit of volume. In accordance with Le Chatelier's principle, a chemical reaction ...
Problem 1-2 - IPN-Kiel
... Give four compounds in which oxygen has another oxidation number than –II. Do not name more than one compound for each different oxidation number. ...
... Give four compounds in which oxygen has another oxidation number than –II. Do not name more than one compound for each different oxidation number. ...
PDF of Chapter 6 Foundations of Chemistry
... Because water is a compound, its composition does not vary. Pure water is always made up of the same atoms in the same combinations. Therefore, a chemical formula can be used to describe the atoms that make up water (H2O). Salt water is a homogeneous mixture, or solution. The solute (NaCl) and the s ...
... Because water is a compound, its composition does not vary. Pure water is always made up of the same atoms in the same combinations. Therefore, a chemical formula can be used to describe the atoms that make up water (H2O). Salt water is a homogeneous mixture, or solution. The solute (NaCl) and the s ...
Homework1-4-Answers
... 17. Balance the following chemical equation: (Section: 3.7) C4H10 + O2 CO2 + H2O Ans: 2(C4H10) + 13 O2 8 CO2 + 10 H2O 18. Balance the following chemical equation: (Section: 3.7) C + Fe2O3 Fe + CO Ans: 3C + Fe2O3 2Fe + 3CO 19. Refer to the (unbalanced) equation CS2 + CaO CO2 + CaS. How many ...
... 17. Balance the following chemical equation: (Section: 3.7) C4H10 + O2 CO2 + H2O Ans: 2(C4H10) + 13 O2 8 CO2 + 10 H2O 18. Balance the following chemical equation: (Section: 3.7) C + Fe2O3 Fe + CO Ans: 3C + Fe2O3 2Fe + 3CO 19. Refer to the (unbalanced) equation CS2 + CaO CO2 + CaS. How many ...
Laboratory Practices from Physical Chemistry
... temperature Tk. Only the end of the flask neck (1–2 cm) has to project from the bath. During sample evaporation, all air from the flask escapes and the flask contains just sample vapor. Sample evaporation process can be monitored using a small piece of filter paper set to the flask neck. When vapor ...
... temperature Tk. Only the end of the flask neck (1–2 cm) has to project from the bath. During sample evaporation, all air from the flask escapes and the flask contains just sample vapor. Sample evaporation process can be monitored using a small piece of filter paper set to the flask neck. When vapor ...
Chapter 1: Chemistry: The Study of Change
... percent yield of the reaction if 28.2 g PI3 is obtained from the reaction of 48.0 g of I2 with excess phosphorus? (Section: 3.10) 2P(s) + 3I2(s) 2PI3(s) Ans: 54.3% 23. What is the limiting reagent when 27.0 g of P and 68.0 g of I2 react according to the following chemical equation? (Section: 3.9) ...
... percent yield of the reaction if 28.2 g PI3 is obtained from the reaction of 48.0 g of I2 with excess phosphorus? (Section: 3.10) 2P(s) + 3I2(s) 2PI3(s) Ans: 54.3% 23. What is the limiting reagent when 27.0 g of P and 68.0 g of I2 react according to the following chemical equation? (Section: 3.9) ...
Chapter 15: Chemical Equilibrium
... The equilibrium concentrations of ammonia and hydrogen chloride do not depend on the amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporate ...
... The equilibrium concentrations of ammonia and hydrogen chloride do not depend on the amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporate ...
Chemical Reactions
... 1. Assign a letter for each coefficient in the reaction 2. Determine an algebraic equation to solve for the value of each coefficient ...
... 1. Assign a letter for each coefficient in the reaction 2. Determine an algebraic equation to solve for the value of each coefficient ...
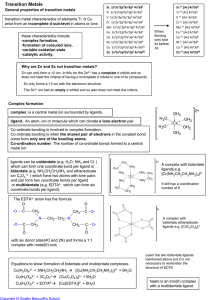
Transition Metals
... It cannot be conc HCl as the Cl- ions would be oxidised to Cl2 by MnO4It cannot be nitric acid as this is an oxidising agent. It oxidises Fe2+ to Fe3+ It cannot be conc H2SO4 as this is an oxidising agent. It cannot be ethanoic acid as this is a weak acid and not supply the large amount of acid need ...
... It cannot be conc HCl as the Cl- ions would be oxidised to Cl2 by MnO4It cannot be nitric acid as this is an oxidising agent. It oxidises Fe2+ to Fe3+ It cannot be conc H2SO4 as this is an oxidising agent. It cannot be ethanoic acid as this is a weak acid and not supply the large amount of acid need ...
g - Highline Community College
... Practice – Predict whether DSsystem is + or − for each of the following • A hot beaker burning your fingers DS is + • Water vapor condensing DS is − • Separation of oil and vinegar salad dressing DS is − DS is + • Dissolving sugar in tea • 2 PbO2(s) 2 PbO(s) + O2(g) DS is + • 2 NH3(g) N2(g) + 3 ...
... Practice – Predict whether DSsystem is + or − for each of the following • A hot beaker burning your fingers DS is + • Water vapor condensing DS is − • Separation of oil and vinegar salad dressing DS is − DS is + • Dissolving sugar in tea • 2 PbO2(s) 2 PbO(s) + O2(g) DS is + • 2 NH3(g) N2(g) + 3 ...
Kinetics Presentation - Chemistrybyscott.org
... text, as well as selected animations and videos. For animations and videos to run properly, we recommend that you run this PowerPoint presentation from the PowerLecture disc inserted in your computer. Also, for the mathematical symbols to display properly, you must install the supplied font called “ ...
... text, as well as selected animations and videos. For animations and videos to run properly, we recommend that you run this PowerPoint presentation from the PowerLecture disc inserted in your computer. Also, for the mathematical symbols to display properly, you must install the supplied font called “ ...
Stoichiometry of Formulas and Equations
... the amounts of substances consumed and produced in a reaction. Suppose you are a polymer chemist preparing a new plastic: how much of this new material will a given polymerization reaction yield? Or suppose you’re a chemical engineer studying rocket engine thrust: what amount of exhaust gases will a ...
... the amounts of substances consumed and produced in a reaction. Suppose you are a polymer chemist preparing a new plastic: how much of this new material will a given polymerization reaction yield? Or suppose you’re a chemical engineer studying rocket engine thrust: what amount of exhaust gases will a ...
2014_S4_CHM_NORMAL (ALL)
... 43. The electronic structure of a compound formed between an element X and chlorine is shown below. (Only electrons in the outermost shells are shown.) What would be the formula of the compound formed between X and magnesium? A. ...
... 43. The electronic structure of a compound formed between an element X and chlorine is shown below. (Only electrons in the outermost shells are shown.) What would be the formula of the compound formed between X and magnesium? A. ...
PDF File
... ~ tenfold faster than that for the substrate with a 2′-fluoro group at U(–1), despite the weaker electron-withdrawing ability of 2′-OH than 2′-F [2]. As a 2′-fluoro group contains lone-pair electrons that can accept hydrogen bonds but cannot donate hydrogen bonds, the higher reactivity of the substr ...
... ~ tenfold faster than that for the substrate with a 2′-fluoro group at U(–1), despite the weaker electron-withdrawing ability of 2′-OH than 2′-F [2]. As a 2′-fluoro group contains lone-pair electrons that can accept hydrogen bonds but cannot donate hydrogen bonds, the higher reactivity of the substr ...
FREE Sample Here - We can offer most test bank and
... 3. The French chemist Antoine Lavoisier found that the weight of objects before burning and the weight of the products after burning were equal. He concluded that the total weight did not change during a process. Which of these best describes Lavoisier's conclusion? a. From observation, Lavoisier cr ...
... 3. The French chemist Antoine Lavoisier found that the weight of objects before burning and the weight of the products after burning were equal. He concluded that the total weight did not change during a process. Which of these best describes Lavoisier's conclusion? a. From observation, Lavoisier cr ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.