solliqsol - chemmybear.com
... (b) A solution is prepared by dissolving 2.53 grams of p-dichlorobenzene (molecular weight 147.0) in 25.86 grams of naphthalene (molecular weight 128.2). Calculate the molality of the p-dichlorobenzene solution. (c) The freezing point of pure naphthalene is determined to be 80.2C. The solution prep ...
... (b) A solution is prepared by dissolving 2.53 grams of p-dichlorobenzene (molecular weight 147.0) in 25.86 grams of naphthalene (molecular weight 128.2). Calculate the molality of the p-dichlorobenzene solution. (c) The freezing point of pure naphthalene is determined to be 80.2C. The solution prep ...
Chemical Reactivity as Described by Quantum Chemical Methods
... pioneering work by Heitler and London [2] on the hydrogen molecule in 1928 providing insight into, to quote Pauling, the Nature of the Chemical Bond [3]. However Quantum Chemistry is, at least in our opinion, more than the mere application of quantum mechanical principles to molecules and their inte ...
... pioneering work by Heitler and London [2] on the hydrogen molecule in 1928 providing insight into, to quote Pauling, the Nature of the Chemical Bond [3]. However Quantum Chemistry is, at least in our opinion, more than the mere application of quantum mechanical principles to molecules and their inte ...
kcse chemistry questions
... Describe how the following reagents can be used to prepare lead sulphate solid potassium sulphate, solid lead carbonate, dilute nitric acid and distilled water. Explain why the enthalpy of neutralization of ethanoic acid with sodium hydroxide is different from that of hydrochloric acid with sodium h ...
... Describe how the following reagents can be used to prepare lead sulphate solid potassium sulphate, solid lead carbonate, dilute nitric acid and distilled water. Explain why the enthalpy of neutralization of ethanoic acid with sodium hydroxide is different from that of hydrochloric acid with sodium h ...
Higher Chemistry Resources Guide - Glow Blogs
... A potential energy diagram can be used to show the energy pathway for a reaction. The enthalpy change is the energy difference between products and reactants. It can be calculated from a potential energy diagram. The enthalpy change has a negative value for exothermic reactions and a positive value ...
... A potential energy diagram can be used to show the energy pathway for a reaction. The enthalpy change is the energy difference between products and reactants. It can be calculated from a potential energy diagram. The enthalpy change has a negative value for exothermic reactions and a positive value ...
A Low-Fluorine Solution with the F/Ba Mole Ratio of 2 for the
... fluorine content in this solution is only about 10.3% of that used in the conventional TFA-MOD solution, which is much lower than that of any reported low-fluorine studies. YBCO thin films with favorable superconducting performance could be obtained repeatedly using this solution. The behaviour of t ...
... fluorine content in this solution is only about 10.3% of that used in the conventional TFA-MOD solution, which is much lower than that of any reported low-fluorine studies. YBCO thin films with favorable superconducting performance could be obtained repeatedly using this solution. The behaviour of t ...
Thermochemistry - Pearson Canada
... atural gas consists mostly of methane, CH 4. As we learned in Chapter 4, the complete combustion of a hydrocarbon, such as methane, yields carbon dioxide and water as products. More important, however, is another “product” of this reaction, which we have not previously mentioned: heat. This heat can ...
... atural gas consists mostly of methane, CH 4. As we learned in Chapter 4, the complete combustion of a hydrocarbon, such as methane, yields carbon dioxide and water as products. More important, however, is another “product” of this reaction, which we have not previously mentioned: heat. This heat can ...
Higher Chemistry Resources Guide - Glow Blogs
... additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are only included as helpful suggestions. Practitioners should also refer to the SQA website for the most up to date Course and Uni ...
... additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are only included as helpful suggestions. Practitioners should also refer to the SQA website for the most up to date Course and Uni ...
PPT - Gmu - George Mason University
... The Chemistry 211/212 General Chemistry courses taught at George Mason are intended for those students enrolled in a science /engineering oriented curricula, with particular emphasis on chemistry, biochemistry, and biology The material on these slides is taken primarily from the course text but the ...
... The Chemistry 211/212 General Chemistry courses taught at George Mason are intended for those students enrolled in a science /engineering oriented curricula, with particular emphasis on chemistry, biochemistry, and biology The material on these slides is taken primarily from the course text but the ...
Document
... reaction is started with [H2 ]0 = 0.76 M, [N2]0 = 0.60 M and [NH3]0= 0.48 M. Which of the following is correct as the reaction comes to equilibrium? A) The concentration of N2will increase B) The concentration of H2will decrease C) The concentration of NH3will decrease D) The concentration of both N ...
... reaction is started with [H2 ]0 = 0.76 M, [N2]0 = 0.60 M and [NH3]0= 0.48 M. Which of the following is correct as the reaction comes to equilibrium? A) The concentration of N2will increase B) The concentration of H2will decrease C) The concentration of NH3will decrease D) The concentration of both N ...
Benzylamine reacts with nitrous acid to form unstable
... Q2. Arrange the following (i) In decreasing order of basic strength in gas phase: C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3 (ii) In increasing order of boiling point: C2H5OH, (CH3)2NH, C2H5NH2 (iii) In increasing order of solubility in water: C6H5NH2, (C2H5)2NH, C2H5NH2. Ans. (i) The given compounds can ...
... Q2. Arrange the following (i) In decreasing order of basic strength in gas phase: C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3 (ii) In increasing order of boiling point: C2H5OH, (CH3)2NH, C2H5NH2 (iii) In increasing order of solubility in water: C6H5NH2, (C2H5)2NH, C2H5NH2. Ans. (i) The given compounds can ...
Contents - MCAT Prep Course
... To find the ∆Hf° for As2, use the following equation. Remember that the ∆Hf° = 0 for pure elements in their natural state. ∆H° = Σ∆Hf°products - ∆Hf°reactants ∆Hf°reactants = ∆Hf°products - ∆H° 2(-786kJ) – (-1794) = +222kJ Since there is only one mole of As2, ∆Hf° for As2 = +222kJ/mol ...
... To find the ∆Hf° for As2, use the following equation. Remember that the ∆Hf° = 0 for pure elements in their natural state. ∆H° = Σ∆Hf°products - ∆Hf°reactants ∆Hf°reactants = ∆Hf°products - ∆H° 2(-786kJ) – (-1794) = +222kJ Since there is only one mole of As2, ∆Hf° for As2 = +222kJ/mol ...
Oxygen Carriers Materials for Chemical
... number was seen for any of these oxygen carriers. Reduction and oxidation kinetics of oxygen carriers of NiO/MgAl2O4 and Mn3O4/Mg-ZrO2 for CLC were investigated using methane and air in a TGA. At high temperature both oxygen carriers reacted rapidly under both reducing and oxidizing conditions and t ...
... number was seen for any of these oxygen carriers. Reduction and oxidation kinetics of oxygen carriers of NiO/MgAl2O4 and Mn3O4/Mg-ZrO2 for CLC were investigated using methane and air in a TGA. At high temperature both oxygen carriers reacted rapidly under both reducing and oxidizing conditions and t ...
REVIEW and answers
... Potential energy of reactants: 100 kJ Potential energy of products: 200 kJ Potential energy of activated complex: 450 kJ Activation energy: 350 kJ H: + 100 kJ Exo- or endo- thermic? endothermic ...
... Potential energy of reactants: 100 kJ Potential energy of products: 200 kJ Potential energy of activated complex: 450 kJ Activation energy: 350 kJ H: + 100 kJ Exo- or endo- thermic? endothermic ...
AP Chemistry Unit 7- Homework Problems Equilibrium and Ksp
... 1. Will a ppt of CaCO3 (Ksp= 3.4 x10-9) form if [Ca+2] = 4 x10-6 M and [CO3-2] = 4 x10-3? Q = [4 x10-6][4x10-3] = 1.6x10-8 >> 3.4x10-9 so yes, ppt 2. Will a ppt of Ag2CrO4 (Ksp = 1.1 x10-12 ) form if [Ag+] = 3x10-4 and [CrO4-2] = 2x10-4? Q = [3 x10-4]2 [2x10-4] = 1.8x10-11 >> 1.1x10-12 so yes, ppt 3 ...
... 1. Will a ppt of CaCO3 (Ksp= 3.4 x10-9) form if [Ca+2] = 4 x10-6 M and [CO3-2] = 4 x10-3? Q = [4 x10-6][4x10-3] = 1.6x10-8 >> 3.4x10-9 so yes, ppt 2. Will a ppt of Ag2CrO4 (Ksp = 1.1 x10-12 ) form if [Ag+] = 3x10-4 and [CrO4-2] = 2x10-4? Q = [3 x10-4]2 [2x10-4] = 1.8x10-11 >> 1.1x10-12 so yes, ppt 3 ...
File - Roden`s AP Chemistry
... (a) A solution containing 3.23 grams of an unknown compound dissolved in 100.0 grams of water freezes at -0.97C. The solution does not conduct electricity. Calculate the molecular weight of the compound. (The molal freezing point depression constant for water is 1.86C kg mole-1) (b) Elemental anal ...
... (a) A solution containing 3.23 grams of an unknown compound dissolved in 100.0 grams of water freezes at -0.97C. The solution does not conduct electricity. Calculate the molecular weight of the compound. (The molal freezing point depression constant for water is 1.86C kg mole-1) (b) Elemental anal ...
theodore l. brown h. eugene lemay, jr. bruce e. bursten catherine j
... system, or transmission in any form or by any means, electronic, mechanical, photocopying, recording, or likewise. To obtain permission(s) to use material from this work, please submit a written request to Pearson Education, Inc., Permissions Department, 1900 E. Lake Ave., Glenview, IL 60025. Many o ...
... system, or transmission in any form or by any means, electronic, mechanical, photocopying, recording, or likewise. To obtain permission(s) to use material from this work, please submit a written request to Pearson Education, Inc., Permissions Department, 1900 E. Lake Ave., Glenview, IL 60025. Many o ...
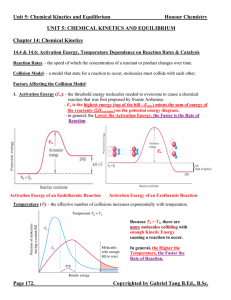
Unit 5: Chemical Kinetics and Equilibrium
... reactant and product concentrations (Equilibrium Expression) has a constant value, K – equilibrium constant. Equilibrium Expression: - an expression relating the concentrations or pressures of the reactants and products when they are at the state of equilibrium. - it takes the form of the individual ...
... reactant and product concentrations (Equilibrium Expression) has a constant value, K – equilibrium constant. Equilibrium Expression: - an expression relating the concentrations or pressures of the reactants and products when they are at the state of equilibrium. - it takes the form of the individual ...
Name:
... According to these results, what would be the initial rate (in mol/(L·s)) if all three concentrations are: [BrO3-]=[Br-]=[H+]=0.20 mol/L? 2. Use the following diagram to answer the questions below. a) Is the reaction exothermic or endothermic? Explain. b) What letter represents the activation energy ...
... According to these results, what would be the initial rate (in mol/(L·s)) if all three concentrations are: [BrO3-]=[Br-]=[H+]=0.20 mol/L? 2. Use the following diagram to answer the questions below. a) Is the reaction exothermic or endothermic? Explain. b) What letter represents the activation energy ...
Precision, accuracy and significant figures
... ammonia cleaning solution is needed to make 250 mL of a 1.50% m/v solution? b What mass of ammonia is present in 150 mL of the 1.50% solution? ...
... ammonia cleaning solution is needed to make 250 mL of a 1.50% m/v solution? b What mass of ammonia is present in 150 mL of the 1.50% solution? ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.