Slide 1
... The sum of reactions in the Calvin cycle is the following: 6 CO2 + 12 NADPH + 12 H+ + 18 ATP → C6H12O6 + 6 H2O + 12 NADP+ + 18 ADP + 18 Pi ...
... The sum of reactions in the Calvin cycle is the following: 6 CO2 + 12 NADPH + 12 H+ + 18 ATP → C6H12O6 + 6 H2O + 12 NADP+ + 18 ADP + 18 Pi ...
Chapter 2 Matter
... Matter can be a gas, a liquid, or a solid. Gases have no fixed shape or volume. Gases can be compressed to form liquids. Liquids have no shape, but they do have a volume. Solids are rigid and have a definite shape and volume. ...
... Matter can be a gas, a liquid, or a solid. Gases have no fixed shape or volume. Gases can be compressed to form liquids. Liquids have no shape, but they do have a volume. Solids are rigid and have a definite shape and volume. ...
Section 4.8: Acid-Base Reactions
... reagent (the titrant), that is required to react completely with an exact amount of another reagent, is measured precisely. The method presumes that the titration reaction goes to completion. In most titrations the concentration of titrant is known, in which case the exact amount of analyte, the sub ...
... reagent (the titrant), that is required to react completely with an exact amount of another reagent, is measured precisely. The method presumes that the titration reaction goes to completion. In most titrations the concentration of titrant is known, in which case the exact amount of analyte, the sub ...
Saturday Study Session 1 1st Class Reactions
... • SOLUTION – if it says a solution, then it CAN be broken into ions if it is soluble in water. • Only ionic compounds can become separate ions in a solution. • The 6 strong acids (HCl, HBr, HI, HNO3, HClO4, H2SO4) and the strong bases (group 1 + OH-and Ba, Sr, Ca + OH-) are always written as separat ...
... • SOLUTION – if it says a solution, then it CAN be broken into ions if it is soluble in water. • Only ionic compounds can become separate ions in a solution. • The 6 strong acids (HCl, HBr, HI, HNO3, HClO4, H2SO4) and the strong bases (group 1 + OH-and Ba, Sr, Ca + OH-) are always written as separat ...
ppt Sc10 Review Notes
... 2 KI(aq) + Pb(NO3)2(aq) 2 KNO3(aq) + PbI2(s) NOTE: -SR and DR reactions always happen in solutions so for ionic compounds check solubility table ...
... 2 KI(aq) + Pb(NO3)2(aq) 2 KNO3(aq) + PbI2(s) NOTE: -SR and DR reactions always happen in solutions so for ionic compounds check solubility table ...
Lecture 6
... “The state of greatest order [lowest S] is at the lowest temperature. With increasing temperature, disorder becomes more prevalent.” ...
... “The state of greatest order [lowest S] is at the lowest temperature. With increasing temperature, disorder becomes more prevalent.” ...
2.0 Chem 20 Final Review
... pair of electrons in an adjacent molecule. ▫ Hydrogen nucleus (proton) is simultaneously attracted to two pairs of electrons; one closer (in the same molecule) and one further away (a lone pair on the next molecule) Why do you need a strongly ...
... pair of electrons in an adjacent molecule. ▫ Hydrogen nucleus (proton) is simultaneously attracted to two pairs of electrons; one closer (in the same molecule) and one further away (a lone pair on the next molecule) Why do you need a strongly ...
Thermodynamics Practice Problems Presentation
... Calculate the enthalpy change for the reaction in which hydrogen gas, H2 (g), is combined with fluorine gas, F2(g), to produce 2 moles of hydrogen fluoride gas, HF(g). This reaction is represented by the ...
... Calculate the enthalpy change for the reaction in which hydrogen gas, H2 (g), is combined with fluorine gas, F2(g), to produce 2 moles of hydrogen fluoride gas, HF(g). This reaction is represented by the ...
Review Study Guide for the Final
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
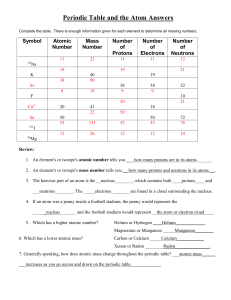
Periodic Table and the Atom Answers
... stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such as “if you have an excess of (compound X)”. This statement shouldn’t worry you… what it really mean ...
... stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such as “if you have an excess of (compound X)”. This statement shouldn’t worry you… what it really mean ...
Thermochemistry - thelapierres.com
... 2SO2(g) + O2(g) → 2SO3(g) ΔH = -198.2 kJ calculate the heat evolved when 87.9 g of SO2 (molar mass = 64.07 g/mol) is converted to SO3. Strategy The thermochemical equation shows that for every 2 moles of SO2 reacted, 198.2 kJ of heat are given off (note the negative sign). Therefore, the conversion ...
... 2SO2(g) + O2(g) → 2SO3(g) ΔH = -198.2 kJ calculate the heat evolved when 87.9 g of SO2 (molar mass = 64.07 g/mol) is converted to SO3. Strategy The thermochemical equation shows that for every 2 moles of SO2 reacted, 198.2 kJ of heat are given off (note the negative sign). Therefore, the conversion ...
XIX. Chemistry, High School
... A. The mass of a mole of CO is exactly half that of a mole of CO2. ...
... A. The mass of a mole of CO is exactly half that of a mole of CO2. ...
File
... cylcoalkene? Briefly explain your choice(s), using either general formulas or units of unsaturation in your answer. (2%) ...
... cylcoalkene? Briefly explain your choice(s), using either general formulas or units of unsaturation in your answer. (2%) ...
Homework Booklet Unit 1 Feb14
... 6. If a copper and sulphur mixture is heated, a red glow is seen to spread through the mixture and this continues even when the heating has stopped. A black solid remains at the end. (a)What is the difference between a mixture and a compound? (b)Give 2 pieces of evidence that suggest that a chemical ...
... 6. If a copper and sulphur mixture is heated, a red glow is seen to spread through the mixture and this continues even when the heating has stopped. A black solid remains at the end. (a)What is the difference between a mixture and a compound? (b)Give 2 pieces of evidence that suggest that a chemical ...
syllabus details - hrsbstaff.ednet.ns.ca
... spacing should be described. A molecular level description of what happens when evaporation, boiling, condensing, melting and freezing occur should be given. Students should understand what is meant by the term diffusion. ...
... spacing should be described. A molecular level description of what happens when evaporation, boiling, condensing, melting and freezing occur should be given. Students should understand what is meant by the term diffusion. ...
02-Atoms-Molecules
... Isotopes are atoms with the same number of protons but different numbers of neutrons ...
... Isotopes are atoms with the same number of protons but different numbers of neutrons ...
Document
... each case, one mole of a particular reactant or product is specified. For example, the enthalpy change involved in the dissolving of one mole of solute is called the molar enthalpy of solution and can be symbolized by ∆Hsol. In Table 1, the substance under consideration in each reaction is highlight ...
... each case, one mole of a particular reactant or product is specified. For example, the enthalpy change involved in the dissolving of one mole of solute is called the molar enthalpy of solution and can be symbolized by ∆Hsol. In Table 1, the substance under consideration in each reaction is highlight ...
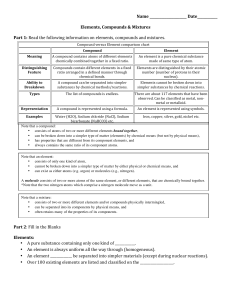
Compound vs Element chart
... • consists of atoms of two or more different elements bound together, • can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), • has properties that are different from its component elements, and • always contains the same ratio of its component at ...
... • consists of atoms of two or more different elements bound together, • can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), • has properties that are different from its component elements, and • always contains the same ratio of its component at ...
Contents and Concepts
... c. Calculate ∆Go from ∆Ho and ∆So. d. Define the standard free energy of formation, ∆Go. e. Calculate ∆Go from standard free energies of formation. f. State the rules for using ∆Go as a criterion for spontaneity g. Interpret the sign of ∆Go. ...
... c. Calculate ∆Go from ∆Ho and ∆So. d. Define the standard free energy of formation, ∆Go. e. Calculate ∆Go from standard free energies of formation. f. State the rules for using ∆Go as a criterion for spontaneity g. Interpret the sign of ∆Go. ...
first test
... 16. Ammonia reacts with diatomic oxygen to form nitric oxide and water vapor: 4NH3 + 5O2 4NO + 6H2O When 40.0 g NH3 and 50.0 g O2 are allowed to react, which is the limiting reagent? A. NH3 B. O2 C. Neither reagent is limiting. ...
... 16. Ammonia reacts with diatomic oxygen to form nitric oxide and water vapor: 4NH3 + 5O2 4NO + 6H2O When 40.0 g NH3 and 50.0 g O2 are allowed to react, which is the limiting reagent? A. NH3 B. O2 C. Neither reagent is limiting. ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.