Nuclear Chemistry - Ector County ISD.
... change (since there is no change in the total number of nuclear particles), however the atomic number will increase by one (because the neutron transmutates into an additional proton). An example of this is the decay of the isotope of carbon named carbon-14 into the element nitrogen: ...
... change (since there is no change in the total number of nuclear particles), however the atomic number will increase by one (because the neutron transmutates into an additional proton). An example of this is the decay of the isotope of carbon named carbon-14 into the element nitrogen: ...
radioactivity-ppt
... Alpha particles may be completely stopped by a sheet of paper, beta particles by aluminum shielding. Gamma rays, however, can only be reduced by much more ...
... Alpha particles may be completely stopped by a sheet of paper, beta particles by aluminum shielding. Gamma rays, however, can only be reduced by much more ...
The discovery of the natural radioactive decay of uranium in 1896 by
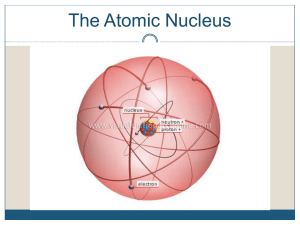
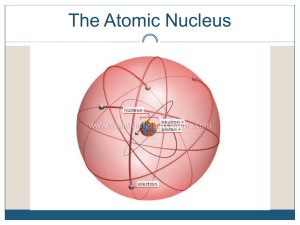
... different atomic weights owing to variations in the number of neutrons. Atoms of the same element with differing atomic weights are called isotopes. Radioactive decay is a spontaneous process in which an isotope (the parent) loses particles from its nucleus to form an isotope of a new element (the d ...
... different atomic weights owing to variations in the number of neutrons. Atoms of the same element with differing atomic weights are called isotopes. Radioactive decay is a spontaneous process in which an isotope (the parent) loses particles from its nucleus to form an isotope of a new element (the d ...
Ch. 21.1 Nuclear Radiation
... particles from certain elements. (like uranium) – also known as radioactive decay ...
... particles from certain elements. (like uranium) – also known as radioactive decay ...
Grade 10S Physics T3W5 material
... in a leaf is magnesium used? If lead accumulates in the body until it kills, which part of the body does it destroy, and where is it stored? Are atoms of iodine able to leave solid iodine into a saturated solution of iodine? 5. When rocks form, a certain amount of radioactive material is locked up i ...
... in a leaf is magnesium used? If lead accumulates in the body until it kills, which part of the body does it destroy, and where is it stored? Are atoms of iodine able to leave solid iodine into a saturated solution of iodine? 5. When rocks form, a certain amount of radioactive material is locked up i ...
Chapter 19 Radioactive Material An Isotope is an element with a
... uranium atoms split. The hot water turns into steam which then spins a turbine that is hooked u to a generator. To prevent the uranium from creating to much heat that might melt the walls ...
... uranium atoms split. The hot water turns into steam which then spins a turbine that is hooked u to a generator. To prevent the uranium from creating to much heat that might melt the walls ...
Chapter 16 – Nuclear Energy
... • 50% of people exposed to 450 rems die. • 800 or more rems always fatal (no effective treatment). • In time, survivors can develop cancer. • Ex: X-ray = 0.1 to 1 rem ...
... • 50% of people exposed to 450 rems die. • 800 or more rems always fatal (no effective treatment). • In time, survivors can develop cancer. • Ex: X-ray = 0.1 to 1 rem ...
Nuclear Fission
... Radioactivity • Radioactive atoms: unstable atoms that decay and emit particles and energy from their nuclei – Not all elements are radioactive • Most cases it is only certain isotopes that are radioactive – Example: »H – 1 = »H – 2 = »H – 3 = ...
... Radioactivity • Radioactive atoms: unstable atoms that decay and emit particles and energy from their nuclei – Not all elements are radioactive • Most cases it is only certain isotopes that are radioactive – Example: »H – 1 = »H – 2 = »H – 3 = ...