Chemical Reactions
... Pick up sock and board. Complete the Do Now via QR code or link I will be about 15-30 min late. This should be completed by the time I arrive. http://bit.ly/1LvB4ak ...
... Pick up sock and board. Complete the Do Now via QR code or link I will be about 15-30 min late. This should be completed by the time I arrive. http://bit.ly/1LvB4ak ...
Matter_and_Change2
... Matter with a uniform and definite composition (also called a pure substance). All samples of a substance have identical physical properties. ...
... Matter with a uniform and definite composition (also called a pure substance). All samples of a substance have identical physical properties. ...
Matter_and_Change
... Physical Property- quality or condition that can be observed or measured without changing the substance’s ...
... Physical Property- quality or condition that can be observed or measured without changing the substance’s ...
Chemical Equations
... Chemical formula A representation of a substance in which the elements are represented by their symbols and subscripts represent the number of atoms of each element ...
... Chemical formula A representation of a substance in which the elements are represented by their symbols and subscripts represent the number of atoms of each element ...
Elements, Compounds and Chemical Reactions
... then silicon, and our bodies are oxygen and then carbon. ...
... then silicon, and our bodies are oxygen and then carbon. ...
chemical reactions
... The starting compound is ammonium dichromate. When heated, it begins to decompose into nitrogen gas, water vapor and powdered chromium (III) oxide. It looks like a volcano with ash being spread all over the place. ...
... The starting compound is ammonium dichromate. When heated, it begins to decompose into nitrogen gas, water vapor and powdered chromium (III) oxide. It looks like a volcano with ash being spread all over the place. ...
Chapter 14 – Chemical Reactions
... Gas Formation – _______________________________________ Solid Formation – _______________________________________ Color Change – _______________________________________ Energy Change – _______________________________________ Chemical Formula – shorthand _____________ for a _____________ or a diatomi ...
... Gas Formation – _______________________________________ Solid Formation – _______________________________________ Color Change – _______________________________________ Energy Change – _______________________________________ Chemical Formula – shorthand _____________ for a _____________ or a diatomi ...
2016-02 Chemical Depedency Rate Reform Project
... Chemical dependency rates reform project The 2009 Minnesota legislature directed the Alcohol and Drug Abuse Division (ADAD) to prepare for the 2011 legislature a statewide rate methodology for the Consolidated Chemical Treatment Fund (CCDTF). The methodology will replace county-negotiated rates with ...
... Chemical dependency rates reform project The 2009 Minnesota legislature directed the Alcohol and Drug Abuse Division (ADAD) to prepare for the 2011 legislature a statewide rate methodology for the Consolidated Chemical Treatment Fund (CCDTF). The methodology will replace county-negotiated rates with ...
Introduction to Chemistry
... Ionic- Two elements bond by transferring electrons to create ions that attract together (+ is attracted to - after an electron is transferred) ...
... Ionic- Two elements bond by transferring electrons to create ions that attract together (+ is attracted to - after an electron is transferred) ...
Study Guide – Unit Test (9-27-13)
... There will be other examples included on the test. (Look at notes/old quizzes and worksheets) ...
... There will be other examples included on the test. (Look at notes/old quizzes and worksheets) ...
Chemistry Content Standards
... a. Compare and contrast atomic/molecular motion in solids, liquids, gases, and plasmas. b. Collect data and calculate the amount of heat given off or taken in by chemical or physical processes. c. Analyzing (both conceptually and quantitatively) flow of energy during change of state (phase). Teacher ...
... a. Compare and contrast atomic/molecular motion in solids, liquids, gases, and plasmas. b. Collect data and calculate the amount of heat given off or taken in by chemical or physical processes. c. Analyzing (both conceptually and quantitatively) flow of energy during change of state (phase). Teacher ...
Honors Chemistry Review Packet KEY
... 6. Liquids and gases both have an indefinite shape; while the shape of a solid is definite, the shape of a liquid is indefinite. 7. It is reversible because solid mercury can be melted back into a liquid again. 8. Platinum and copper can have the same mass and volume (extensive properties). They can ...
... 6. Liquids and gases both have an indefinite shape; while the shape of a solid is definite, the shape of a liquid is indefinite. 7. It is reversible because solid mercury can be melted back into a liquid again. 8. Platinum and copper can have the same mass and volume (extensive properties). They can ...
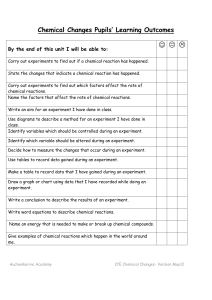
Chemical Reactions Unit Pupils` Learning Outcomes
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
Unit 2: Chemical Reactions
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
Element - the simplest form of matter that can exist under normal
... Element - the simplest form of matter that can exist under normal laboratory conditions Elements are pure substances. Elements cannot be separated into simpler substances by chemical means. Elements are the building blocks for all other substances There are now 117 known elements (as of 2006). All e ...
... Element - the simplest form of matter that can exist under normal laboratory conditions Elements are pure substances. Elements cannot be separated into simpler substances by chemical means. Elements are the building blocks for all other substances There are now 117 known elements (as of 2006). All e ...
Chemical Reaction Basics
... Advanced Chemistry – Chapter 8 A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
... Advanced Chemistry – Chapter 8 A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
chemical warfare world war i
... used tear gas grenades against the German army, to little effect. ...
... used tear gas grenades against the German army, to little effect. ...
Unit 5 Chemical Properties and Changes Video Notes A ______ is a
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
Deconstructed HS-PS1-2
... trends in the periodic table, and knowledge of the patterns of chemical properties.[Clarification Statement: Examples of chemical reactions could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical rea ...
... trends in the periodic table, and knowledge of the patterns of chemical properties.[Clarification Statement: Examples of chemical reactions could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical rea ...