Electronic Structure and the Periodic Table A. Bohr Model of the
... Electronic Structure and the Periodic Table A. Bohr Model of the Atom 1. Solar System Model 2. Created to Fit a “Quantized” Picture of Energy Transfer 3. Basis: Noncontinuous Emission Spectra of the Elements 4. Basic Postulates a. Electrons reside in certain allowed energy states b. Energy absorptio ...
... Electronic Structure and the Periodic Table A. Bohr Model of the Atom 1. Solar System Model 2. Created to Fit a “Quantized” Picture of Energy Transfer 3. Basis: Noncontinuous Emission Spectra of the Elements 4. Basic Postulates a. Electrons reside in certain allowed energy states b. Energy absorptio ...
Electromagnetic Spectrum Wavelength Wavenumber Frequency
... Dative Covalent Bond Coordination number ...
... Dative Covalent Bond Coordination number ...
AP Chemistry
... What is the energy in joules of a mole of photons associated with visible light of wavelength 550 nm? ...
... What is the energy in joules of a mole of photons associated with visible light of wavelength 550 nm? ...
7.4 The Wave Nature of Matter * 7.5 Quantum Mechanics and the Atom
... • Schrödinger's equation can be used derive the energies and orbitals of electrons in atoms Schrödinger equation ...
... • Schrödinger's equation can be used derive the energies and orbitals of electrons in atoms Schrödinger equation ...
1. Millikan did his experiments with the balance of
... forces on a small drop of oil that lies between two electrode plates. By knowing the size of an electric field, the charge on the drops of oil that was dropped (droplets) can be determined. By repeating this experiment several times, he found that the values measured are always multiples of the same ...
... forces on a small drop of oil that lies between two electrode plates. By knowing the size of an electric field, the charge on the drops of oil that was dropped (droplets) can be determined. By repeating this experiment several times, he found that the values measured are always multiples of the same ...
Electrons #1
... In larger atoms, Noble Gasses are used to help abbreviate the notation Ex: We can change P from 1s2 2s2 2p6 3s2 3p3 to [Ne] 3s2 3p3 We can do this because Neon is 1s2 2s2 2p6 Find the closest Noble Gas & build from there ...
... In larger atoms, Noble Gasses are used to help abbreviate the notation Ex: We can change P from 1s2 2s2 2p6 3s2 3p3 to [Ne] 3s2 3p3 We can do this because Neon is 1s2 2s2 2p6 Find the closest Noble Gas & build from there ...
Chapter 13 – Electrons in Atoms
... Electron Arrangement in Atoms Physics and the Quantum Mechanical Model ...
... Electron Arrangement in Atoms Physics and the Quantum Mechanical Model ...
09 Exam 1 Key
... 1. (24)a. Number the following types of light from lowest (#1) to highest (#4) frequency. ...
... 1. (24)a. Number the following types of light from lowest (#1) to highest (#4) frequency. ...
Modern Model of the Atom Student Notes and Assignment
... The ways in which electrons are arranged around the nuclei of atoms are called ELECTRON CONFIGURATIONS. The rules that govern the way the electrons fill the atomic orbitals are: 1. AUFBAU PRINCIPLE - electrons enter orbitals of the lowest energy levels first 2. PAULI EXCLUSION PRINCIPLE - an atomic ...
... The ways in which electrons are arranged around the nuclei of atoms are called ELECTRON CONFIGURATIONS. The rules that govern the way the electrons fill the atomic orbitals are: 1. AUFBAU PRINCIPLE - electrons enter orbitals of the lowest energy levels first 2. PAULI EXCLUSION PRINCIPLE - an atomic ...
Periodic Properties of the Elements
... of the orbital depends only on principle quantum number For atoms with more than 2 electrons, the energy depends on l & n ...
... of the orbital depends only on principle quantum number For atoms with more than 2 electrons, the energy depends on l & n ...
The Address of the Electrons
... order for electrons to share an orbital, they must have different spin ¡ One spin up ¡ One spin down ...
... order for electrons to share an orbital, they must have different spin ¡ One spin up ¡ One spin down ...
(s) If 5.00 moles of zinc is placed into 1.50 L... 34. solution,what is the mass of the hydrogen gas produced?
... Base your answers to questions 34 through 32 on on the following chemical reaction: ...
... Base your answers to questions 34 through 32 on on the following chemical reaction: ...
File - Chemistry 11 Enriched
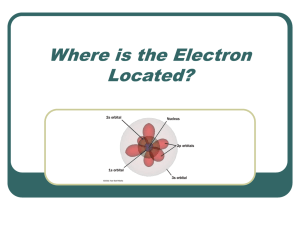
... Each element has a specific electron configuration defining where the electrons are located. In order to understand the location of electrons, we must now look at the atom in three dimensions rather than the planetary early model of the atom. The orbitals are not two dimensional tracks like railroad ...
... Each element has a specific electron configuration defining where the electrons are located. In order to understand the location of electrons, we must now look at the atom in three dimensions rather than the planetary early model of the atom. The orbitals are not two dimensional tracks like railroad ...
Where is the Electron Located?
... Magnetic Quantum Number (m): Indicates the orientation of an orbital around the nucleus. Spin Quantum Number (↓↑): Indicates which way the electron is spinning ...
... Magnetic Quantum Number (m): Indicates the orientation of an orbital around the nucleus. Spin Quantum Number (↓↑): Indicates which way the electron is spinning ...
AP Chemistry Chapter 6 Outline for Concepts to Know 6.1 Wave
... 6.1 Wave Nature of Light Basic anatomy and vocabulary of a wave (wavelength, frequency, amplitude) Relationship between speed, wavelength and frequency c= 3.00x108 m/s order of categories of electromagnetic radiation order and approximate range of visible radiation 4 – 8(x10-7) m 6.2 Quant ...
... 6.1 Wave Nature of Light Basic anatomy and vocabulary of a wave (wavelength, frequency, amplitude) Relationship between speed, wavelength and frequency c= 3.00x108 m/s order of categories of electromagnetic radiation order and approximate range of visible radiation 4 – 8(x10-7) m 6.2 Quant ...
LT1: Electron.NOTES - Simpson County Schools
... What is the quantum mechanical model of the atom and how is it different from the Bohr model? _________________________________________________________________________________________________________ ____________________________________________________________________________________________________ ...
... What is the quantum mechanical model of the atom and how is it different from the Bohr model? _________________________________________________________________________________________________________ ____________________________________________________________________________________________________ ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.