ELECTRONS IN ATOMS
... Most of an atom’s mass is concentrated in the small, positively charged nucleus.The electrons surround the nucleus and the rest of the atom is empty space. Electrons are arranged in concentric circular paths around the nucleus. ...
... Most of an atom’s mass is concentrated in the small, positively charged nucleus.The electrons surround the nucleus and the rest of the atom is empty space. Electrons are arranged in concentric circular paths around the nucleus. ...
Chapter 5 PPT/Notes B
... • The Aufbau principle says electrons fill the lowest energy levels first. • Hund’s rule says to place one electron in each orbital before placing the 2nd one of opposite ...
... • The Aufbau principle says electrons fill the lowest energy levels first. • Hund’s rule says to place one electron in each orbital before placing the 2nd one of opposite ...
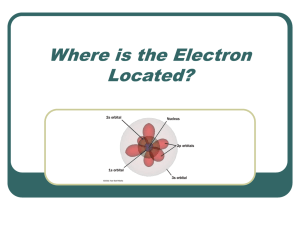
Where is the Electron Located?
... Magnetic Quantum Number (m): Indicates the orientation of an orbital around the nucleus. Spin Quantum Number (↓↑): Indicates which way the electron is spinning ...
... Magnetic Quantum Number (m): Indicates the orientation of an orbital around the nucleus. Spin Quantum Number (↓↑): Indicates which way the electron is spinning ...
CHEMISTRY 1A
... c. The maximum number of orbitals that may be associated with the following set of quantum numbers n = 5 and l = 3 is _________. d. The maximum number of electrons that may be associated with the quantum number set n = 4, l = 1, and ml = 1 is _________. ...
... c. The maximum number of orbitals that may be associated with the following set of quantum numbers n = 5 and l = 3 is _________. d. The maximum number of electrons that may be associated with the quantum number set n = 4, l = 1, and ml = 1 is _________. ...
Pauli Exclusion Principle Quiz
... Pauli Exclusion Principle Quiz 1. The location of any electron in an atom can be described by ____ unique quantum numbers. ...
... Pauli Exclusion Principle Quiz 1. The location of any electron in an atom can be described by ____ unique quantum numbers. ...
Quantum Theory of the Atom
... D. Electrons do not move around the nucleus in circular orbits E. Laid ground for later atomic ...
... D. Electrons do not move around the nucleus in circular orbits E. Laid ground for later atomic ...
Chapter 3 Study Guide
... i. Tells you the order in which electrons fill up an atom. b. Rules to obey when determining electron configurations: i. Pauli Exclusion Principle: no two electrons in the same orbital can have the same 4 quantum numbers. ii. aufbau principle: electrons fill up the lowest available energy levels fir ...
... i. Tells you the order in which electrons fill up an atom. b. Rules to obey when determining electron configurations: i. Pauli Exclusion Principle: no two electrons in the same orbital can have the same 4 quantum numbers. ii. aufbau principle: electrons fill up the lowest available energy levels fir ...
Chemistry Notes
... A. All matter in the universe is composed of atoms. B. Parts of an atom 1. Electron Shell – The outer part of an atom. This is where the electrons are found. a) Electrons – Negatively charged particles found in the electron shell. Electrons orbit the nucleus. 2. Nucleus – The center of the atom. Com ...
... A. All matter in the universe is composed of atoms. B. Parts of an atom 1. Electron Shell – The outer part of an atom. This is where the electrons are found. a) Electrons – Negatively charged particles found in the electron shell. Electrons orbit the nucleus. 2. Nucleus – The center of the atom. Com ...
Aufbau Diagram Directions
... Pauli Exclusion: an atomic orbital may describe at most 2 electrons (each electron will have a different spin) Hund’s Rule: When electrons occupy orbitals of equal energy, one electron enters each orbital until all the orbitas contain one electron, then a second electron is added to each orbital. Ho ...
... Pauli Exclusion: an atomic orbital may describe at most 2 electrons (each electron will have a different spin) Hund’s Rule: When electrons occupy orbitals of equal energy, one electron enters each orbital until all the orbitas contain one electron, then a second electron is added to each orbital. Ho ...
Mr. Knittel`s Final Review Sheet I Answers
... Rutherford placed a small, dense positive charge in the center of the atomic model surrounded by a blob of electrons. Although Rutherford’s model is often called the planetary model, be careful not to mix it up with Bohr – Rutherford’s model did NOT have fixed orbits for the electrons. Realizing tha ...
... Rutherford placed a small, dense positive charge in the center of the atomic model surrounded by a blob of electrons. Although Rutherford’s model is often called the planetary model, be careful not to mix it up with Bohr – Rutherford’s model did NOT have fixed orbits for the electrons. Realizing tha ...
09 Exam 1 Key
... 1. (24)a. Number the following types of light from lowest (#1) to highest (#4) frequency. ...
... 1. (24)a. Number the following types of light from lowest (#1) to highest (#4) frequency. ...
Chemistry Science Notebook
... List the three reasons scientists found Rutherford’s nuclear atomic model to be fundamentally incomplete. ...
... List the three reasons scientists found Rutherford’s nuclear atomic model to be fundamentally incomplete. ...
ATOMIC STRUCTURE
... En = (-RH)(1/n2) n = 1,2,3,4…. RH = Rydberg constant (2.18 x 10-18 J) n = principle quantun number ...
... En = (-RH)(1/n2) n = 1,2,3,4…. RH = Rydberg constant (2.18 x 10-18 J) n = principle quantun number ...
Modern Atomic Theory
... Light has the characteristics of both a wave and a particle. Light is made up of particles called “photons” …following Einstein's introduction of photons in light waves, one knew that light contains particles which are concentrations of energy incorporated into the wave, suggests that all particles, ...
... Light has the characteristics of both a wave and a particle. Light is made up of particles called “photons” …following Einstein's introduction of photons in light waves, one knew that light contains particles which are concentrations of energy incorporated into the wave, suggests that all particles, ...
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
... anode is reached only by those electrons that have enough kinetic energy Ek to overcome the work eUanode: Ek ≥ eUanode electron collide with many Mercury atoms, if Ugrid < U*, these collisions will always be elastic: no energy loss and anode current (I) will increase; if Ugrid = U*, collisions might ...
... anode is reached only by those electrons that have enough kinetic energy Ek to overcome the work eUanode: Ek ≥ eUanode electron collide with many Mercury atoms, if Ugrid < U*, these collisions will always be elastic: no energy loss and anode current (I) will increase; if Ugrid = U*, collisions might ...
Review Notes - Biochemistry
... 5. Chemical Formula: Where each _ELEMENT_ is represented by its chemical _SYMBOL_ and the _NUMBER__ of atoms is shown in __SUBSCRIPTS__. ...
... 5. Chemical Formula: Where each _ELEMENT_ is represented by its chemical _SYMBOL_ and the _NUMBER__ of atoms is shown in __SUBSCRIPTS__. ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.