Primary electrons make random elastic and inelastic collision either
... Bremsstrahlung (continuum) X-rays, deceleration of primary electrons (with primary energy E0) in the Columbic field of the nucleus, a type of ‘braking’ effect…. i.e. as to pass though the stronger electric filed, close to nuclei, it will suffer a “quantum jump” to a low energy state, which will make ...
... Bremsstrahlung (continuum) X-rays, deceleration of primary electrons (with primary energy E0) in the Columbic field of the nucleus, a type of ‘braking’ effect…. i.e. as to pass though the stronger electric filed, close to nuclei, it will suffer a “quantum jump” to a low energy state, which will make ...
King Abdulaziz University, Department of Physics, Jeddah
... Electrons are treated as an ideal neutral gas, and their total energy depends on the temperature and applied field. ...
... Electrons are treated as an ideal neutral gas, and their total energy depends on the temperature and applied field. ...
Electron Configuration - Warren County Public Schools
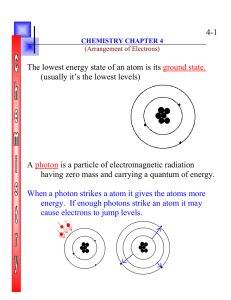
... • Light has wave-like properties but can also be thought of as a stream of particles. • Each particle of light carries a quantum of energy. • He called these particles photons. • A photon is a particle of electromagnetic radiation having zero mass and carrying a quantum of energy. ...
... • Light has wave-like properties but can also be thought of as a stream of particles. • Each particle of light carries a quantum of energy. • He called these particles photons. • A photon is a particle of electromagnetic radiation having zero mass and carrying a quantum of energy. ...
Review Exam #1 - Seattle Central College
... Characteristics (Electrons have Mass and Momentum) Electromagnetic Radiation (Light) has Both Wave Characteristics (Waves can be Diffracted by Crystals) and Particle Characteristics (Photons have Momentum, but No Mass) Emission spectra of elements demonstrate that the energy of electrons is quantize ...
... Characteristics (Electrons have Mass and Momentum) Electromagnetic Radiation (Light) has Both Wave Characteristics (Waves can be Diffracted by Crystals) and Particle Characteristics (Photons have Momentum, but No Mass) Emission spectra of elements demonstrate that the energy of electrons is quantize ...
The Nature of the Atom The Nature of the Atom
... The Nature of the Atom • Why do we need standing matter waves? The answer to this question naturally comes when the Schrödinger equation is solved for the hydrogen atom. In 1926, Schrödinger proposed a wave equation that described how matter waves change in space and time. The Schrödinger equati ...
... The Nature of the Atom • Why do we need standing matter waves? The answer to this question naturally comes when the Schrödinger equation is solved for the hydrogen atom. In 1926, Schrödinger proposed a wave equation that described how matter waves change in space and time. The Schrödinger equati ...
Chemistry I Honors – Semester Exam Review – Fall 2000
... Hydrogen atoms have specific energy levels. Therefore, the atoms can only gain or lose certain amounts of energy. When atoms lose energy, they emit photons which correspond to the lines in the emission spectrum. The more energy lost, the more energy the photon has. Bohr’s model stated that electrons ...
... Hydrogen atoms have specific energy levels. Therefore, the atoms can only gain or lose certain amounts of energy. When atoms lose energy, they emit photons which correspond to the lines in the emission spectrum. The more energy lost, the more energy the photon has. Bohr’s model stated that electrons ...
x 100 QUANTUM NUMBERS AND SYMBOLS
... number is 3. What are the possible value sets (l, ml, ms) for the other three quantum numbers? 2. Is it possible for an electron in an atom to have these quantum numbers: n=2, l =1, ml =3, ms =1/2? Why or why not? ...
... number is 3. What are the possible value sets (l, ml, ms) for the other three quantum numbers? 2. Is it possible for an electron in an atom to have these quantum numbers: n=2, l =1, ml =3, ms =1/2? Why or why not? ...
4-1 The lowest energy state of an atom is its ground state. (usually
... When a photon strikes a atom it gives the atoms more energy. If enough photons strike an atom it may cause electrons to jump levels. ...
... When a photon strikes a atom it gives the atoms more energy. If enough photons strike an atom it may cause electrons to jump levels. ...
1st Term Review
... 14. Based on the gold foil experiment, what did Rutherford conclude about the atom? 15. An atom of chromium-60 contains how many protons, neutron and electrons? 16. What is the difference between a compound and an element? 17. What is the electron configuration of a neutral calcium atom? 18. Atomic ...
... 14. Based on the gold foil experiment, what did Rutherford conclude about the atom? 15. An atom of chromium-60 contains how many protons, neutron and electrons? 16. What is the difference between a compound and an element? 17. What is the electron configuration of a neutral calcium atom? 18. Atomic ...
Atomic Structure Practice Answers
... 5. This is a possible configuration for a transition metal atom. B 6. This electron configuration is not possible. A 7. This is a possible configuration for a transition metal ion. C 8-11 refer to the following: A. B. C. D. E. ...
... 5. This is a possible configuration for a transition metal atom. B 6. This electron configuration is not possible. A 7. This is a possible configuration for a transition metal ion. C 8-11 refer to the following: A. B. C. D. E. ...
Lecture28
... • When light is incident on a system of atoms, there is usually a net absorption. This is because when the system is in thermal equilibrium there are many more atoms in the ground state than in excited states. • If there are more atoms in excited states than in the ground state (population inversion ...
... • When light is incident on a system of atoms, there is usually a net absorption. This is because when the system is in thermal equilibrium there are many more atoms in the ground state than in excited states. • If there are more atoms in excited states than in the ground state (population inversion ...
Chapter 7 NotesAA
... Electrons coming from excited to ground state which gives off a photon of light (h) ...
... Electrons coming from excited to ground state which gives off a photon of light (h) ...
Atomic Theory Study Guide - Reading Community Schools
... 2. Name orbitals given its quantum numbers, or identify quantum numbers for given orbital. 3. Sketch the relative shapes, sizes, and spatial orientations of s, p, and d orbitals of the hydrogen atom. 4. Apply the concept of effective nuclear charge, Zeff, to describe the difference in orbital energi ...
... 2. Name orbitals given its quantum numbers, or identify quantum numbers for given orbital. 3. Sketch the relative shapes, sizes, and spatial orientations of s, p, and d orbitals of the hydrogen atom. 4. Apply the concept of effective nuclear charge, Zeff, to describe the difference in orbital energi ...
HW 12 - stKFUPM
... A Nobel Laureate (Chemistry & Peace) who used the shell model greatly in his teaching and research is ...
... A Nobel Laureate (Chemistry & Peace) who used the shell model greatly in his teaching and research is ...
Chapter 4
... Particle Nature of Light Bohr Model of the H atom 1913 – Danish physicist – Niels Bohr Single e- circled around nucleus in allowed paths or orbits e- has fixed E when in this orbit (lowest E closest to nucleus) Lot of empty space between nucleus and e- in which e- cannot be in E increas ...
... Particle Nature of Light Bohr Model of the H atom 1913 – Danish physicist – Niels Bohr Single e- circled around nucleus in allowed paths or orbits e- has fixed E when in this orbit (lowest E closest to nucleus) Lot of empty space between nucleus and e- in which e- cannot be in E increas ...
Glossary Chapter 4
... simultaneously both the position and velocity of an electron or any other particle (99) highest occupied energy level the electron-containing main energy level with the highest principal quantum number (110) ...
... simultaneously both the position and velocity of an electron or any other particle (99) highest occupied energy level the electron-containing main energy level with the highest principal quantum number (110) ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.