Lecture_19-Energy Levels in the Bohr model of the atom
... model in which the single hydrogen electron can only be in certain definite orbits. • In the nth allowed orbit, the electron has orbital angular momentum nh/2π (see Figure on the right). • Bohr proposed that angular momentum is quantized (this will turn out to be correct in general in quantum mechan ...
... model in which the single hydrogen electron can only be in certain definite orbits. • In the nth allowed orbit, the electron has orbital angular momentum nh/2π (see Figure on the right). • Bohr proposed that angular momentum is quantized (this will turn out to be correct in general in quantum mechan ...
Introduction to Quantum Mechanics Homework #3 (Due on April 28
... on the wavefunction given in the textbook. Also calculate the numerical value of E0 in eV unit. 4. 1) What is the virial theorem? 2)Calculate the average potential energy of H1s electron in eV unit using the wavefunction given in the textbook. What is the average kinetic energy of the H1s electron? ...
... on the wavefunction given in the textbook. Also calculate the numerical value of E0 in eV unit. 4. 1) What is the virial theorem? 2)Calculate the average potential energy of H1s electron in eV unit using the wavefunction given in the textbook. What is the average kinetic energy of the H1s electron? ...
power point notes
... Rutherford proposed that the atom consists of a tiny positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus contains almost all of the mass of the atom and consists of protons and neutrons. The number of electrons surrounding the nucleus, equals the number of p ...
... Rutherford proposed that the atom consists of a tiny positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus contains almost all of the mass of the atom and consists of protons and neutrons. The number of electrons surrounding the nucleus, equals the number of p ...
Energy, Heat, and Work* Oh My*
... The Quantum Mechanical Model of the Atom The matter-wave of the electron occupies the space near the nucleus and is continuously influenced by it. The Schrödinger wave equation allows us to solve for the energy states associated with a particular atomic orbital. ...
... The Quantum Mechanical Model of the Atom The matter-wave of the electron occupies the space near the nucleus and is continuously influenced by it. The Schrödinger wave equation allows us to solve for the energy states associated with a particular atomic orbital. ...
Quantum Mechanical Model
... A person can’t stand between rungs/neither can an electron. To move from level to level, an electron must gain or release just the right amount of energy. ...
... A person can’t stand between rungs/neither can an electron. To move from level to level, an electron must gain or release just the right amount of energy. ...
Chapter 4
... c = λν where: c is the speed of light 3.0 x 108 m/s λ is the wavelength ν is the frequency ...
... c = λν where: c is the speed of light 3.0 x 108 m/s λ is the wavelength ν is the frequency ...
Chapter 37 Early Quantum Theory and Models of the Atom
... wave would be a circular standing wave will occur. This yields the same relation that Bohr had proposed. In addition, it makes more reasonable the fact that the electrons do not radiate, as one would otherwise expect from an accelerating charge. ...
... wave would be a circular standing wave will occur. This yields the same relation that Bohr had proposed. In addition, it makes more reasonable the fact that the electrons do not radiate, as one would otherwise expect from an accelerating charge. ...
Chapter 5 : Electrons in Atoms
... Bohr’s model explained the hydrogen’s spectral lines, but failed to explain any other element’s lines. The behavior of electrons is still not fully understood, but it is known they do not move around the nucleus in circular orbits. ...
... Bohr’s model explained the hydrogen’s spectral lines, but failed to explain any other element’s lines. The behavior of electrons is still not fully understood, but it is known they do not move around the nucleus in circular orbits. ...
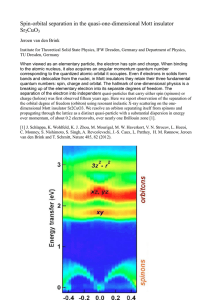
Spin-orbital separation in the quasi-one
... Institute for Theoretical Solid State Physics, IFW Dresden, Germany and Department of Physics, TU Dresden, Germany When viewed as an elementary particle, the electron has spin and charge. When binding to the atomic nucleus, it also acquires an angular momentum quantum number corresponding to the qua ...
... Institute for Theoretical Solid State Physics, IFW Dresden, Germany and Department of Physics, TU Dresden, Germany When viewed as an elementary particle, the electron has spin and charge. When binding to the atomic nucleus, it also acquires an angular momentum quantum number corresponding to the qua ...
Electron discovered 1897, Thomson Atom model 1913, Bohr
... Quantum Mechanics 1925, Heisenberg, Schrödinger ...
... Quantum Mechanics 1925, Heisenberg, Schrödinger ...
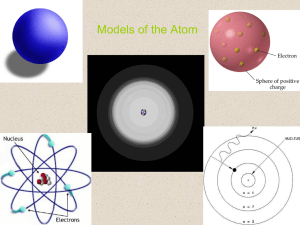
Models of the Atom
... – The quantum mechanical model (electron cloud model): • The modern description of electrons in an atom • Determines the allowed energies an electron can have and how likely it is to find the electron in various locations around the nucleus. ...
... – The quantum mechanical model (electron cloud model): • The modern description of electrons in an atom • Determines the allowed energies an electron can have and how likely it is to find the electron in various locations around the nucleus. ...
DPPs 1 - Career Point
... A neutron of kinetic energy 65 eV collides inelastically with a singly ionized helium atom at rest. It is scattered at an angle of 90º with respect of its original direction. (i) Find the allowed values of the energy of the neutron & that of the atom after collision. (ii) If the atom gets de-excited ...
... A neutron of kinetic energy 65 eV collides inelastically with a singly ionized helium atom at rest. It is scattered at an angle of 90º with respect of its original direction. (i) Find the allowed values of the energy of the neutron & that of the atom after collision. (ii) If the atom gets de-excited ...
Adobe Acrobat file () - Wayne State University Physics and
... electrons, when an electron moves from the n = 1 level to the n = 3 level, the circumference of its orbit becomes 9 times greater. This occurs because (a) there are 3 times as many wavelengths in the new orbit, (b) there are 3 times as many wavelengths and each wavelength is 3 times as long, (c) the ...
... electrons, when an electron moves from the n = 1 level to the n = 3 level, the circumference of its orbit becomes 9 times greater. This occurs because (a) there are 3 times as many wavelengths in the new orbit, (b) there are 3 times as many wavelengths and each wavelength is 3 times as long, (c) the ...
Chapter 11 Review Worksheet
... 16. Define the term ionization energy? Ionization energy = the amount of energy it takes to remove an electron from an element 17. Define the term and atomic radius? Atomic radius = the size of the atom, usually measured from the nucleus to the outer edge 18. How do the ionization energies and atomi ...
... 16. Define the term ionization energy? Ionization energy = the amount of energy it takes to remove an electron from an element 17. Define the term and atomic radius? Atomic radius = the size of the atom, usually measured from the nucleus to the outer edge 18. How do the ionization energies and atomi ...
1. Define the vocabulary on page 88. Section 1
... 3. All forms of electromagnetic radiation move at a constant speed of _____________ through a vacuum. 4. _________ is the distance between corresponding points on adjacent waves. 5. What is the symbol for wavelength? 6. Frequency is defined as _______________________________________. 7. What is the ...
... 3. All forms of electromagnetic radiation move at a constant speed of _____________ through a vacuum. 4. _________ is the distance between corresponding points on adjacent waves. 5. What is the symbol for wavelength? 6. Frequency is defined as _______________________________________. 7. What is the ...
Problem Set 1
... ~ J~ and Jz where J~ is the total angular momentum operator must be eigenfunctions of J. and Jz is its z-component. ( You have a total of six wave functions) 5. Assuming the spin-orbit interaction hamiltonian is given by Hsl = ...
... ~ J~ and Jz where J~ is the total angular momentum operator must be eigenfunctions of J. and Jz is its z-component. ( You have a total of six wave functions) 5. Assuming the spin-orbit interaction hamiltonian is given by Hsl = ...
NAME PERIOD ______ DATE Chapter 5 Sec. 2
... 10. How many orientations are possible for the orbitals related to each of the following sublevels? a. s b. p c. d d. f ...
... 10. How many orientations are possible for the orbitals related to each of the following sublevels? a. s b. p c. d d. f ...
Review 1st Qtr KEY
... c. radiation must be emitted. d. the electron must make a transition from a higher to a lower energy level. ...
... c. radiation must be emitted. d. the electron must make a transition from a higher to a lower energy level. ...
The de Broglie-Bohr Model for the Hydrogen Atom
... properties is subject to constructive and destructive interference. As will be shown this leads naturally to quantization of electron momentum and kinetic energy, and consequently a manifold of allowed energy states for the electron relative to the nucleus. The de Broglie-Bohr model of the hydrogen ...
... properties is subject to constructive and destructive interference. As will be shown this leads naturally to quantization of electron momentum and kinetic energy, and consequently a manifold of allowed energy states for the electron relative to the nucleus. The de Broglie-Bohr model of the hydrogen ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.