Notes-15 - KSU Physics
... The example in He shows that we can think that for each atom, there are electron orbitals, designated by n . Depending on the number of electrons available, one can put each electron in one of the orbitals. Each one of these orbitals for a fixed n is called a subshell, and each fixed n is called ...
... The example in He shows that we can think that for each atom, there are electron orbitals, designated by n . Depending on the number of electrons available, one can put each electron in one of the orbitals. Each one of these orbitals for a fixed n is called a subshell, and each fixed n is called ...
The Modern Atomic Model
... Bohr Model of the Atom (review) •Energy levels contain electrons. •Electrons travel around the nucleus. •Different orbitals varied by different quantum (energy). •Gaps between energy levels were not equal. ...
... Bohr Model of the Atom (review) •Energy levels contain electrons. •Electrons travel around the nucleus. •Different orbitals varied by different quantum (energy). •Gaps between energy levels were not equal. ...
8.5C Vocabulary
... Electron Shell Outermost Electron Shell Valence Electrons Metals Non-Metals Metalloids Noble Gas Reactivity ...
... Electron Shell Outermost Electron Shell Valence Electrons Metals Non-Metals Metalloids Noble Gas Reactivity ...
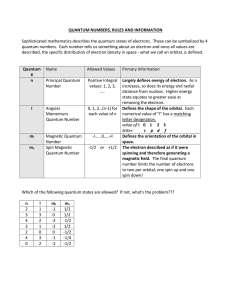
Chapter 7, Quantum Nos.
... For the H atom the orbital energy depends only on n, so all orbitals with the same value of n have the same energy. This is not true, however, for any other atom! The H atom orbitals may be used to approximate the orbitals for multi-electron atoms. But since these atoms have more than one electron, ...
... For the H atom the orbital energy depends only on n, so all orbitals with the same value of n have the same energy. This is not true, however, for any other atom! The H atom orbitals may be used to approximate the orbitals for multi-electron atoms. But since these atoms have more than one electron, ...
Study Guide: Chapter 4 - the Arrangement of Electrons in Atoms
... 2. Understand the relationship between energy of light and its frequency; Know how to calculate the energy of light given its frequency, and the frequency given its energy (MEMORIZE FORMULA; Planck’s constant will be given on the test) – work a few practice problems 3. Know what a line emission spec ...
... 2. Understand the relationship between energy of light and its frequency; Know how to calculate the energy of light given its frequency, and the frequency given its energy (MEMORIZE FORMULA; Planck’s constant will be given on the test) – work a few practice problems 3. Know what a line emission spec ...
Chp7,Quantum_Num
... For the H atom the orbital energy depends only on n, so all orbitals with the same value of n have the same energy. This is not true, however, for any other atom! The H atom orbitals may be used to approximate the orbitals for multi-electron atoms. But since these atoms have more than one electron, ...
... For the H atom the orbital energy depends only on n, so all orbitals with the same value of n have the same energy. This is not true, however, for any other atom! The H atom orbitals may be used to approximate the orbitals for multi-electron atoms. But since these atoms have more than one electron, ...
Electron Configuration Class Notes
... Energy moves in waves, but it can act as particles (photons). Louie de Broglie – “matter waves” Postulated that since light shows a “dual nature” – has wave properties as well as particulate properties, then matter should also be able to move - not only as particles - but also as waves! - this prope ...
... Energy moves in waves, but it can act as particles (photons). Louie de Broglie – “matter waves” Postulated that since light shows a “dual nature” – has wave properties as well as particulate properties, then matter should also be able to move - not only as particles - but also as waves! - this prope ...
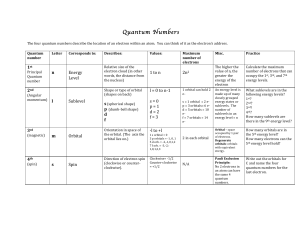
Quantum Number Table
... Sophisticated mathematics describes the quantum states of electrons. These can be symbolized by 4 quantum numbers. Each number tells us something about an electron and once all values are described, the specific distribution of electron density in space - what we call an orbital, is defined. ...
... Sophisticated mathematics describes the quantum states of electrons. These can be symbolized by 4 quantum numbers. Each number tells us something about an electron and once all values are described, the specific distribution of electron density in space - what we call an orbital, is defined. ...
Periodic Trends
... share electrons in order to acquire a full set of eight valence electrons. The octet rule is useful for predicting what types of ions an element is likely to form. ...
... share electrons in order to acquire a full set of eight valence electrons. The octet rule is useful for predicting what types of ions an element is likely to form. ...
Advanced Simulation Activity - Westgate Mennonite Collegiate
... associated with pure elements as you have seen. What models of the atom did it replace? Why? For this, take a look at the models of the atom that have come before: 1. Switch from Experiment to Prediction in the upper left hand corner of the simulation. Highlight the Billiard Ball model (Dalton’s mod ...
... associated with pure elements as you have seen. What models of the atom did it replace? Why? For this, take a look at the models of the atom that have come before: 1. Switch from Experiment to Prediction in the upper left hand corner of the simulation. Highlight the Billiard Ball model (Dalton’s mod ...
Ch. 4: Electron Configuration
... • Ground state: An atom’s lowest energy state • Excited state: Higher potential energy than ground state. • Photon: A particle of electromagnetic radiation having zero mass and carrying a quantum of energy (i.e., packet of light) • Only certain wavelengths of light are emitted by hydrogen atoms when ...
... • Ground state: An atom’s lowest energy state • Excited state: Higher potential energy than ground state. • Photon: A particle of electromagnetic radiation having zero mass and carrying a quantum of energy (i.e., packet of light) • Only certain wavelengths of light are emitted by hydrogen atoms when ...
the bohr-sommerfeld model of the atom
... atom as consisting of a massive, compact and positively charged nucleus and a negatively charged, point-like electron of mass m moving with constant speed v in a circular orbit around the nucleus (see Fig. 1). The centripetal force F~ required to keep the electron in a circular orbit is the electric ...
... atom as consisting of a massive, compact and positively charged nucleus and a negatively charged, point-like electron of mass m moving with constant speed v in a circular orbit around the nucleus (see Fig. 1). The centripetal force F~ required to keep the electron in a circular orbit is the electric ...
Bohr model
In atomic physics, the Rutherford–Bohr model or Bohr model, introduced by Niels Bohr in 1913, depicts the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity. After the cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911) came the Rutherford–Bohr model or just Bohr model for short (1913). The improvement to the Rutherford model is mostly a quantum physical interpretation of it. The Bohr model has been superseded, but the quantum theory remains sound.The model's key success lay in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. While the Rydberg formula had been known experimentally, it did not gain a theoretical underpinning until the Bohr model was introduced. Not only did the Bohr model explain the reason for the structure of the Rydberg formula, it also provided a justification for its empirical results in terms of fundamental physical constants.The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell atom. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. However, because of its simplicity, and its correct results for selected systems (see below for application), the Bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. A related model was originally proposed by Arthur Erich Haas in 1910, but was rejected. The quantum theory of the period between Planck's discovery of the quantum (1900) and the advent of a full-blown quantum mechanics (1925) is often referred to as the old quantum theory.