* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Milk is a suspension of lipids and proteins. At... soluble because they have either a net positive or net...
SNARE (protein) wikipedia , lookup
Theories of general anaesthetic action wikipedia , lookup
Magnesium transporter wikipedia , lookup
Protein phosphorylation wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Circular dichroism wikipedia , lookup
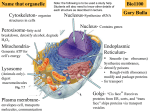
Endomembrane system wikipedia , lookup
Implicit solvation wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Signal transduction wikipedia , lookup
Type three secretion system wikipedia , lookup
List of types of proteins wikipedia , lookup
Protein moonlighting wikipedia , lookup
Bacterial microcompartment wikipedia , lookup
Western blot wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Milk is a suspension of lipids and proteins. At the pH of milk (about 6.4) these proteins are soluble because they have either a net positive or net negative charge. The charge keeps the proteins from interacting with each other by repelling each other yet and allows for sufficient solvation. The proteins do not get close enough for short range interactions like van der Waals forces to take effect. Bacteria produce acidic byproducts that decrease the pH as does the addition of vinegar. Several of the major proteins in milk must have isoelectric points in the acidic range. At their isoelectric points the proteins have no net charge. While the proteins have no net charge, their surfaces still carry multiple positive and negative regions. The proteins begin to interact through this network of positive and negative charges. Other interactions, such as van der Waals forces and the hydrophobic effect increase the total attractive force. The proteins thus begin to aggregate and eventually the particles grow large enough that they precipitate leading to the appearance of coagulation.