* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download energy
Survey
Document related concepts
Citric acid cycle wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Biosequestration wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Proteolysis wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Enzyme inhibitor wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Drug discovery wikipedia , lookup
Microbial metabolism wikipedia , lookup
Isotopic labeling wikipedia , lookup
Photosynthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Transcript
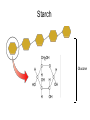
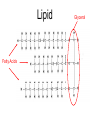
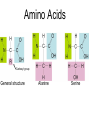
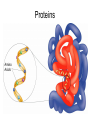
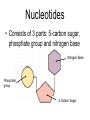
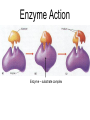
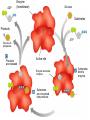
Unit 3:CELLS Cellular Energy Carbon Compounds Objective • What are the functions of each group of organic compounds? Interest Grabber Section 2-3 Life’s backbone • Most of the compounds that make up living things contain carbon. In fact, carbon makes up the basic structure, or “backbone,” of these compounds. Each atom of carbon has four electrons in its outer energy level, which makes it possible for each carbon atom to form four bonds with other atoms. • As a result, carbon atoms can form long chains. A huge number of different carbon compounds exist. Each compound has a different structure. For example, carbon chains can be straight or branching. Also, other kinds of atoms can be attached to the carbon chain. Methane Acetylene Butadiene Benzene Isooctane Macromolecules “giant molecules” • Formed by a process called polymerization Monomers • Smaller units Polymers • Linked up monomers Carbohydrates • Compounds made up of carbon, hydrogen, and oxygen atoms usually in a ratio of 1:2:1 • Main source of energy • The monomers of starch are sugars • Single sugar molecules are called monosaccharides • The large macromolecules formed from monosaccharides are known as polysaccharides Starch Glucose Lipids • Made mostly from carbon and hydrogen atoms • Used to store energy Lipid Fatty Acids Glycerol Proteins • Macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen • Proteins are polymers of molecules called amino acids Amino Acids Carboxyl group General structure Alanine Serine • More than 20 different amino acids, can join to any other amino acid • The instructions for arranging amino acids into many different proteins are stored in DNA • Each protein has a specific role • The shape of proteins can be very important Proteins Amino Acids Nucleic Acids • Macromolecules containing hydrogen, oxygen, nitrogen, carbon, and phosphorus Double Helix Nucleotides • Consists of 3 parts: 5-carbon sugar, phosphate group and nitrogen base Nitrogen Base Phosphate group 5-Carbon Sugar 2 kinds of nucleic acids • RNA (ribonucleic acids) – contains sugar ribose • DNA (deoxyribonucleic acid) – contains sugar deoxyribose Homework 1. Name four groups of organic compounds found in living things 2. Describe at least one function of each group of organic compounds 3. Compare the structures and functions of lipids and starches Chemical Reactions and Enzymes Objectives • What happens to chemical bonds during chemical reactions? • How do energy changes affect whether a chemical reaction will occur? • Why are enzymes important to living things? The Big Idea • Living things are made up of chemical compounds • Everything that happens to an organism is based on chemical reactions Chemical Reactions • A process that changes or transforms one set of chemicals into another Reactants • Elements or compounds that enter into a reaction Products • Elements or compounds produced by a chemical reaction Example Reaction: Getting rid of carbon dioxide • In the blood CO2 + H20 H2CO3 (carbonic acid) • In the lungs H2CO3 CO2 + H2O Released as you breathe Energy in reactions Energy-Absorbing Reaction Energy-Releasing Reaction Activation energy Products Activation energy Reactants Reactants Products Activation Energy • The energy that is needed to get a reaction started Enzymes • Some chemical reactions are too slow or have activation energies that are too high to make them practical for living tissue • These chemical reactions are made possible by catalysts Catalyst • Substance that speeds up the rate of chemical reactions • Work by lowering a reactions activation energy Enzyme • • • • • Biological catalysts Speed up reactions in cells Very specific Named for the reaction is catylzes Enzyme names always end in ase Reaction pathway without enzyme Activation energy without enzyme Reactants Reaction pathway with enzyme Activation energy with enzyme Products Substrates • The reactants of enzyme catalyzed reactions • The active site of the enzyme and the substrate have complementary shapes • Fit like a lock and key Enzyme Action Enzyme – substrate complex ADP Enzyme (hexokinase) Glucose Substrates Products ATP Glucose-6phosphate Products are released Active site Enzyme-substrate complex Substrates are converted into products Substrates bind to enzyme Regulation of Enzyme Activity • Enzymes are affected by any variable that affects chemical reactions 1. pH 2. Temperature 3. Concentration of enzyme Homework 1. What happens to chemical bonds during chemical reactions 2. Describe the role of energy in chemical reactions 3. What are enzymes, and how are they important to living things? 4. Describe how enzymes work, including the role of the enzyme substrate complex 5. A change in pH can change the protein. How might a change in pH affect the function of an enzyme such as hexokinase (hint: think about the analogy of the lock and key)