* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Introduction to the study of cell biology
Magnesium transporter wikipedia , lookup
Magnesium in biology wikipedia , lookup
Butyric acid wikipedia , lookup
Lipid signaling wikipedia , lookup
Biosynthesis wikipedia , lookup
Signal transduction wikipedia , lookup
Western blot wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Microbial metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Mitochondrial replacement therapy wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Phosphorylation wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Biochemistry wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Electron transport chain wikipedia , lookup
Mitochondrion wikipedia , lookup
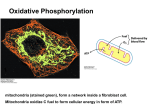
Chapter 6 Mitochondria : Energy Conversion Mitochondria: in all eukaryotic cells Mit: Oxidative phosphorylation → ATP ZHOU Yong Department of Biology XinJiang Medical University 1 Teaching Requirements • 1. Mastering: ultrastructure of mitochondria; function of mitochondria: oxidative phosphorylation. • 2. Comprehending: relationship between structure and function of mitochondria. • 3. Understanding: genomes of mitochondria; proliferation of Mitochondria 2 I. Distribution of Mitochondria The size and number of mitochondria reflect the energy requirements of the cell. Figure. Relationship between mitochondria and microtubules. 3 4 Figure. Localization of mitochondria near sites of high ATP utilization in cardiac muscle and a sperm tail. 5 Figure. Mitochondrial plasticity. Rapid changes of shape are observed when a mitochondrion is visualized in a living cell. 6 Figure. Fractionation of purified mitochondria into separate components. 7 II. Mitochondrial ultrastructure (1) Inner membrane (2) Out membrane (3) Intermembrane space (4) Matrix Inner and outer mitochondrial membranes enclose two spaces: the matrix and intermembrane space. 8 Electron micrograph of a mitochondrion 9 10 1.Ribosome 2.Cristae 3.DNA 4.ATP synthase particles 11 The inner membrane is folded into Cristae 12 Some morphology of mitochondrial cristae 13 ATP synthase particles Elementary particles F0-F1 ATPase complex F0-F1 coupling factor 1. Head sector 2. Stalk sector 3. Membrane sector 14 The structure of the ATP synthase particle Molecular basis of phosphorylation F1 particle is the catalytic subunit; The F0 particle attaches to F1 and is embedded in the inner membrane. F1: 5 subunits in the ratio 3:3:1:1:1 F0: 1a:2b:12c 15 16 Outer membrane: Contains channel-forming protein, called Porin. Permeable to all molecules of 5000 daltons or less. Inner membrane (Impermeability): Contains proteins with three types of functions: (1) Electron-transport chain: Carry out oxidation reactions; (2) ATP synthase: Makes ATP in the matrix; (3) Transport proteins: Allow the passage of metabolites Intermembrane space: Contains several enzymes use ATP to phosphorylate other nucleotides. Matrix: Enzymes; Mit DNA, Ribosomes, etc. 17 18 III. Oxidative phosphorylation Fig. Three stages of cellular catabolism that via controlled “burning” conserve energy for use in heterotrophic cells. Food is hydrolysed into small molecules in the cytoplasm. Glycolysis is also cytoplasmic. Pyruvate and other substrates are taken up by mitochondria under aerobic conditions and through TCA - Krebs cycle and electron transport converted into waste molecules and products ATP and NADH. 19 Localization of metabolic functions within the mitochondrion Outer membrane: Phospholipid synthesis Fatty acid desaturation Fatty acid elongation Matrix Pyruvate oxidation TCA cycle ß oxidation of fats Inner membrane: Electron transport Oxidative phosphorylation Metabolite transport Intermembrane space Nucleotide phosphorylation DNA replication, RNA transcription, Protein translation 20 Complete lysis of glucose can be divided into four steps: 1. Glycolysis 2. Formation of the acetyl CoA 3. Tricarboxylic acid cycle (TCA) 4. Oxidative phosphorylation 21 ATP molecule: energy currency or energy carrier 22 23 A. Glycolysis 24 Fig. Summary of glycolysis. • glycolysis can provide sufficient energy for growth of anaerobic organisms and tissues, or autotrophic cells in the dark. • the reactions only partially oxidize glucose to ethanol or pyruvate • occur in cytoplasm. 25 B. Formation of the acetyl CoA 1. Pyruvate enter into mitochondrial matrix from cytoplasm. 2. Catalyzed by pyruvate dehydrogenase 26 C. Tricarboxylic acid cycle (TCA) Krebs cycle Citric acid cycle 1. Occur in mitochondrial matrix. 2. Require oxygen. 27 Fig. TCA (tricarboxylic acid) cycle. MAIN FUNCTIONS: •oxidation of substrates •reduction of cofactors •substrate level phosphorylation •regeneration of acceptor •release of 3CO2 per turn 28 The main points to remember: 1.The cycle uses acetyl CoA as the immediate substrate - this can come from beta oxidation of fatty acids OR from pyruvate via glycolysis. 2.The products are reducing molecules NADH and FADH2; GTP; CO2 and a molecule of oxaloacetate is regenerated. 3.One way of describing the stoichiometry of the TCA cycle is as follows: Glucose + 6H2O + 10NAD+ + 2FAD + 4ADP + 4Pi ——> 6CO2 + 10NADH +10H+ + 2FADH2 + 4ATP + 4H2O 29 Net result of the glycolytic pathway and the citric acid cycle 30 D. Oxidative phosphorylation 1. Electron carriers (cofactors) (1) nicotinamide adenine dinucleotide (NAD) R=H 31 (2) Flavoproteins a. flavin mononucleotide (FMN) 32 b. flavin adenine dinucleotide (FAD) Oxidized form Reduced form 33 (3) Ubiquinone 34 (4) Cytochromes 35 (5) Iron-sulfur proteins 36 2. Electron-transport chain (respiratory chain) Electron-carrying prosthetic groups in the respiratory chain 37 38 Mitochondrial electron transport chain shown in the context of redox potential (I.e. free energy content per electron) of the components. Most of the energy is CONSERVED in the proton gradient and membrane potential this energy is harvested in the next step, by the ATPase complex. 39 Primary and secondary electron-transport chains NADHO2: 3ATP/2e; FADH2 O2 : 2ATP/2e 40 3. ATP synthesis Energy contained in the reduced molecules formed in TCA cycle is converted into high energy of ATP by: 1) ELECTRON TRANSPORT CHAIN (forming a proton gradient and membrane potential) and B) proton gradient dissipating ATPase which SYNTHESIZES ATP. 2) SUBSTRATE LEVEL 41 PHOSPHORYLATION F1 particles have ATP synthase activity 42 Mithchell’s Chemiosmotic theory (1961) The electrochemical gradient resulting from transport of protons links to oxidative phosphorylation. When electrons are transported along the chain, the H+ is translocated across the inner membrane. The mitochondrial inner membrane is impermeable to H+ . When protons flow in the reverse direction through the F1-F0 coupling factor complex, the potential energy is released. It drive ATP synthesis. 43 44 45 Inhibitors affect the respiratory chain and ATP synthesis in mitochondia 46 Summary of the major activities during aerobic respiration in a mitochondrion 47 IV. Mitochondrial semi-independence 48 IV. Mitochondrial proliferation Direct division of Mitochondria in mouse liver cell 49 REVIEW QUESTIONS • 1. Describe the main opinions briefly about Mithchell’s Chemiosmotic theory. • 2. Describe the ultrastructure of Mitochondrion. • 3. Compare the permeability of outer membrane and inner membrane of Mitochondrion. 50