* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Biochemistry PowerPoint 1
Survey
Document related concepts
Radical (chemistry) wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Genetic code wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Photosynthesis wikipedia , lookup
Peptide synthesis wikipedia , lookup
Drug discovery wikipedia , lookup
Protein structure prediction wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Metalloprotein wikipedia , lookup
Biosynthesis wikipedia , lookup
Transcript
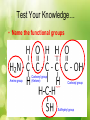
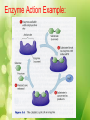
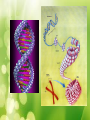
Unit #1 The Chemistry of Life Organic Chemistry Organic Compounds • Organic compounds are compounds that contain carbon (with the exception of CO2 and a few others). Organic Compounds • Make up most of living organisms • Carbon can easily bond with up to 4 other elements 4 valence electrons = 4 covalent bonds Organic Compounds • Carbon atoms form the “backbone” of long chains or rings • Organic molecules can be extremely large and complex; these are called macromolecules (or polymers) Functional Groups • Various elements attach to the hydrocarbon backbone to form different types of compounds. • These reactive clusters of atoms are called functional groups. • Elements include: H, O, S & P Functional Groups Hydroxyl group • -OH • Found in alcohols • E.g. Ethanol • Polar Functional Groups Carboxyl group • -COOH • Found in acids • E.g. Vinegar • Polar Functional Groups Amino group • -NH2 • Found in bases • E.g. Ammonia Functional Groups Sulfhydryl group • -SH • Found in Rubber Functional Groups Phosphate group • -PO4 • Found in ATP Functional Groups TEST YOUR KNOWLEDGE • What functional groups are in this molecule? Test Your Knowledge… • Name the functional groups Test Your Knowledge… • Name the functional groups Amino group Carbonyl group (Ketone) Carboxyl group Sulfhydryl group Organic Compounds The 4 main types of organic macromolecules: Carbohydrates Lipids Proteins Nucleic Acids Making & Breaking Organic Compounds Anabolic Reactions Condensation Reactions (Dehydration synthesis Reaction) • The removal of a –H from the functional group of one unit and a –OH from another unit to form a water molecule (H2O). • Energy absorbed Making & Breaking Organic Compounds Catabolic Reactions Hydrolysis Reactions • A water molecule (H2O) is used to break a covalent bond holding subunits together. • A –H from is given to one unit and a –OH to the another • Energy released Enzymes • Enzymes are biological catalysts. • They speed up reactions without actually being consumed in the reaction. • They are needed for condensation & hydrolysis reactions. Enzyme Action Example: End Part I Get ready for Carbohydrates! Carbohydrates • Made of C, H, & O • Main energy source for living things • Breakdown of sugars supplies immediate energy for cell activities • Plants store extra sugar as complex carbohydrates called starches Carbohydrates • Single sugar molecules are called monosaccharides Examples: glucose – in many plant and animal tissues, most common monosaccharide fructose – in many fruits galactose – component of milk Carbohydrates • Large molecules of many monosaccharide are polysaccharides • Examples: • glycogen – animals use to store excess sugar • plant starch – plants use to store excess sugar • cellulose – fibers that give plants their rigidity & strength Lipids • Store more energy than CHOs because the chains are longer • Ex: Fats, oils, waxes • Won’t dissolve in water Lipids • Important parts of biological membranes and waterproof coverings • Steroids are lipids that act as chemical messengers Lipids • Many lipids are made from a glycerol combined with fatty acids – If all carbons have single bonds, lipid is saturated – Ex: butter, lard, animal fat (usually solid at room temperature) – If any carbons have double or triple bonds, lipid is unsaturated – Ex: vegetable oil, fish oil, peanut oil room temperature) (usually liquid at Proteins • Contain C, H, O, plus nitrogen • Formed from amino acids joined together • More than 20 amino acids can be joined in any order or number to make countless proteins (think of how many words can be made from 26 letters!) Proteins • Chains are folded and twisted giving each protein a unique shape • Van der Waals forces and hydrogen bonds help maintain protein’s shape • Shape of protein is important to its function! Proteins • Provide structure – Ex: Collagen- makes up your skin, muscles & bones • Aid chemical activities in your body – Ex: Enzymes- work to speed up rxns in your body • Transport substances into or out of cells • Help fight diseases Nucleic Acids • Contain C, H, O, N plus phosphorus • Formed by bonding of individual units called nucleotides nucleotide Nucleic Acid Nucleic Acids • Store and transmit hereditary information –Ex: DNA (deoxyribonucleic acid) RNA (ribonucleic acid)