* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Naming Aldehydes & Ketones
Survey
Document related concepts
Physical organic chemistry wikipedia , lookup
Metal carbonyl wikipedia , lookup
Discodermolide wikipedia , lookup
Kinetic resolution wikipedia , lookup
Hydrogenation wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
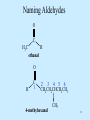
Polythiophene wikipedia , lookup
Ene reaction wikipedia , lookup
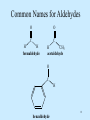
Wolff rearrangement wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
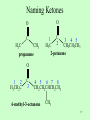
Carbohydrate wikipedia , lookup
Aldol reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Transcript
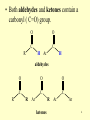
Aldehydes and Ketones Chapter 23 Hein * Best * Pattison * Arena Version 1.0 Colleen Kelley Chemistry Department 1 College Pima Community © John Wiley and Sons, Inc. Chapter Outline 23.1 Structures of Aldehydes and Ketones 23.4 Chemical and Physical Properties of Aldehydes and Ketones 23.2 Naming Aldehydes and 23.5 Common Aldehydes and Ketones Ketones 23.3 Bonding and Physical Properties 23.6 Condensation Polymers 2 Structures of Aldehydes & Ketones 3 • Both aldehydes and ketones contain a carbonyl ( C=O) group. O O C C R Ar H H aldehydes R O O O C C C R Ar R ketones Ar Ar 4 • The general formula for the saturated homologous series of aldehydes and ketones is: CnH2nO 5 •In a linear expression, the aldehyde group is often written as: CHO O C H3C H is equivalent to CH3CHO 6 •In the linear expression of a ketone, the carbonyl group is written as: CO O C H3C CH3 is equivalent to CH3COCH3 7 Naming Aldehydes & Ketones 8 IUPAC Rules for Naming Aldehydes 1. To establish the parent name, select the longest continuous chain of carbon atoms that contains the aldehyde group. 2. The carbons of the parent chain are numbered starting with the aldehyde group. Since the aldehyde group is at the beginning (or end) of a chain, it is understood to be number 1. 9 IUPAC Rules for Naming Aldehydes 3. Form the parent aldehyde name by dropping the –e from the corresponding alkane name and adding the suffix –al. 4. Other groups attached to the parent chain are named and numbered as we have done before. 10 Naming Aldehydes O C H3C H ethanal O H C 1 2 3 4 5 6 CH2CH2CHCH2CH3 4-methyhexanal CH3 11 12 Common Names for Aldehydes O O C C H H formaldehyde H CH3 acetaldehyde O C H 13 benzaldehyde Dialdehydes • In dialdehydes, the suffix –dial is added to the corresponding hydrocarbon name. O O HCCH2CH2CH butanedial 14 IUPAC Rules for Naming Ketones 1. To establish the parent name, select the longest continuous chain of carbon atoms that contain the ketone group. 2. Form the parent name by dropping the –e from the corresponding alkane name and add the suffix –one. 15 IUPAC Rules for Naming Ketones 3. If the chain is longer than four carbons, it is numbered so that the carbonyl group has the smallest number possible; this number is prefixed to the parent name of the ketone. 4. Other groups attached to the parent chain are named and numbered as we have done before. 16 Naming Ketones O O C H3C CH3 propanone C 1 3 4 5 2 H3C CH2CH2CH3 2-petanone O 1 2 H3CH2C C 3 4 5 6 7 8 CH2CH2CHCH2CH3 6-methyl-3-octanone CH3 17 Common Names for Ketones O O C C H3C CH3 propanone acetone H3C CH2CH3 butanone methyl ethyl ketone, MEK 18 Bonding and Physical Properties 19 Bonding • The carbon atom of the carbonyl group is sp2-hybridized and is joined to three other atoms by sigma bonds. • The fourth bond is made by overlapping p electrons of carbon and oxygen to form a pi bond between the carbon and oxygen atoms. 20 Bonding • Because the oxygen atom is considerably more electronegative than carbon, the C=O group is polar. • Many of the chemical reactions of aldehydes and ketones are due to this polarity. C + O 21 Properties • Unlike alcohols, aldehydes and ketones cannot hydrogen-bond to themselves, because no hydrogen atom is attached to the oxygen atom of the carbonyl group. • Aldehydes and ketones, therefore, have lower boiling points than alcohols of comparable molar mass. 22 23 Chemical Properties of Aldehydes & Ketones 24 Reactions of Aldehydes & Ketones • Oxidation – aldehydes only • Reduction – aldehydes and ketones • Addition – aldehydes and ketones 25 Oxidation of Aldehydes • Aldehydes are easily oxidized to carboxylic acids by a variety of oxidizing agents, including (under some conditions) oxygen of the air. O O 3 3 + C R H Cr2O72- + + 8H + 3 Cr3+ + 4H2O C R OH 26 Tollens test • The Tollens test (silver-mirror test) for aldehydes is based on the ability of silver ions to oxidize aldehydes. O C R O + 2 Ag+ H NH3 H2O + 2 Ag (s) C R O-NH4+ 27 Fehling and Benedict Tests • Fehling and Benedict solutions contain Cu2+ ions in an alkaline medium. • In these tests, the aldehyde group is oxidized to an acid by Cu2+ ions. O C R O + 2 Cu+2 NaOH H2O H blue R + 2 Cu2O (s) C O-Na+ brick red 28 Tollens, Fehling & Benedict Tests • Because most ketones do not give a positive with Tollens, Fehling, or Benedict solutions, these tests are used to distinguish between aldehydes and ketones. O C R + 2 Cu+2 NaOH no reaction H2O R O C R + 2 Ag+ R NH3 H2O no reaction 29 Biochemical Oxidation of Aldehydes • When our cells ‘burn’ carbohydrates, they take advantage of the aldehyde reactivity. • The aldehyde is oxidized to a carboxylic acid and is eventually converted to carbon dioxide, which is then exhaled. • This stepwise oxidation provides some of the energy necessary to sustain life. 30 Reduction of Aldehydes & Ketones • Aldehydes and ketones are easily reduced to alcohols. O H2/Ni heat C R RCH2OH primary alcohol H O H2/Ni heat C R R OH RCHR secondary alcohol 31 Addition Reactions of Aldehydes & Ketones • Common addition reactions: – Addition of alcohols • hemiacetal, hemiketal, acetal, ketal – Addition of hydrogen cyanide (HCN) • cyanohydrin – Aldol Condensation (self-addition) 32 Addition of Alcohols • Aldehydes react with alcohols in the presence of a trace of acid to form hemiacetals: OH O + CH3OH C H CH2CH3 propanal H+ HC CH2CH3 OCH3 1-methoxy-1-propanol (propionaldehyde methyl hemiacetal) 33 Addition of Alcohols • In the presence of excess alcohol and strong acid such as dry HCl, aldehydes or hemiacetals react with a second molecule of the alcohol to give an acetal: OH HC CH2CH3 + CH3OH OCH3 OCH3 dry HCl HC CH2CH3 +H2O OCH3 1,1-dimethoxypropane (propionaldehyde dimethyl acetal) 34 Addition of Alcohols to Aldehydes and Ketones OH C R OR' H hemiacetal OH C R OR' R hemiketal OR' R H OR' C OR' acetal R R C OR' ketal 35 Addition of Hydrogen Cyanide • The addition of HCN to aldehydes and ketones forms a class of compounds known as O cyanohydrins: OH + HCN C H CH3 acetaldehyde O + HCN C H3C CH3 acetone HO - HO - HC CH3 CN acetaldehyde cyanohydrin OH H3CC CH3 CN acetone cyanohydrin 36 Aldol Condensation (Self-Addition) • In a carbonyl compound, the carbon atoms are labeled alpha (), beta (), gamma (), delta (), and so on, according to their positions with respect to the carbonyl group. -C-C-C-C=O • The hydrogen atoms attached to the -carbon have the unique ability to be more easily released as protons than other hydrogens within the molecule. 37 Aldol Condensation (Self-Addition) • An aldehyde or ketone that contains hydrogens may add to itself or to another hydrogen containing aldehyde or ketone. O C H H CH3 O dilute NaOH H C H C H OH H O CH H3C C H2C H aldol (3-hydroxybutanal) 38 Common Aldehydes & Ketones 39 Formaldehyde (Methanal) • Formaldehyde is made from methanol by reaction with oxygen (air) in the presence of a silver or copper catalyst. Ag • 2 CH3OH + O2 2H2C=O + 2H2O heat • Formaldehyde is widely used in the synthesis of polymers. 40 Acetaldehyde (Ethanal) • Its principal use is as an intermediate in the manufacture of other chemicals, such as acetic acid and 1-butanol. 41 Acetone and Methyl Ethyl Ketone • • Acetone is used as a solvent in the manufacture of drugs, chemicals, and explosives. It is also used as a solvent. Methyl ethyl ketone (MEK) is also widely used as a solvent, especially for lacquers. 42 Condensation Polymers 43 Phenol-Formaldehyde Polymers (Bakelite) • Each formaldehyde molecule reacts with two phenol molecules to eliminate water. The polymer is then OH formed. OH O + + C H H OH OH H2 C + H2O 44 Phenol-Formaldehyde Polymers (Bakelite) • Polymers of this type are still used, especially in electrical equipment, because of the insulating and fire-resistant properties. 45 46