* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Swim Initiation Neurons in Tritonia diomedea1
Holonomic brain theory wikipedia , lookup
Artificial general intelligence wikipedia , lookup
Electrophysiology wikipedia , lookup
Environmental enrichment wikipedia , lookup
Microneurography wikipedia , lookup
Neuroplasticity wikipedia , lookup
Multielectrode array wikipedia , lookup
Synaptogenesis wikipedia , lookup
Neurotransmitter wikipedia , lookup
Neural oscillation wikipedia , lookup
Neuromuscular junction wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Single-unit recording wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Metastability in the brain wikipedia , lookup
Muscle memory wikipedia , lookup
Mirror neuron wikipedia , lookup
Development of the nervous system wikipedia , lookup
Embodied language processing wikipedia , lookup
Nonsynaptic plasticity wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Circumventricular organs wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Chemical synapse wikipedia , lookup
Neural coding wikipedia , lookup
Neuroanatomy wikipedia , lookup
Biological neuron model wikipedia , lookup
Optogenetics wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Nervous system network models wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
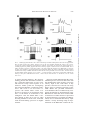
AMER. ZOOL., 41:952–961 (2001) Swim Initiation Neurons in Tritonia diomedea1 W. N. FROST,2,* T. A. HOPPE,† J. WANG,* AND L.-M. TIAN† *Department of Cell Biology and Anatomy, Finch University of Health Sciences/ The Chicago Medical School, 3333 Green Bay Road, North Chicago, Illinois 60064 †Department of Neurobiology and Anatomy, The University of Texas Medical School at Houston, P.O. Box 20708 Houston, Texas 77225 INTRODUCTION The marine mollusc Tritonia diomedea initiates a rhythmic escape swim upon contact with a sufficiently aversive stimulus, such as the seastar Pycnopodia helianthoides (Fig. 1a). The neural program underlying this behavior can be elicited by briefly stimulating any one of a number of peripheral nerves in the isolated brain preparation, indicating that the motor program is centrally generated, with little involvement of sensory feedback ((Dorsett et al., 1969; Frost, 1999) and Fig. 5). Previous work has described much of the circuitry mediating the Tritonia escape swim response, including the afferent neurons (S-cells) (Getting, 1976; Slawsky, 1979), the central pattern generator (CPG) neurons (Getting, 1983), and the swim flexion neurons (Willows et al., 1973; Hume et al., 1982) (Fig. 1b). While these findings made it possible to study pattern generation, From the Symposium Swimming in Opisthobranch Mollusks: Contributions to Control of Motor Behavior presented at the Annual Meeting of the Society for Integrative and Comparative Biology, 4–8 January 2000, at Atlanta, Georgia. 2 E-mail: [email protected] 1 motor program initiation could not readily be addressed due to a gap in our knowledge of the circuit elements between the afferent neurons and the swim CPG. Here we fill-in this gap, by describing two recently discovered pre-CPG interneurons, Tr1 (Triggertype 1) and DRI (Dorsal Ramp Interneuron), that convey the output of the afferent neurons to the CPG. An initial description of DRI has been published previously (Frost and Katz, 1996); Tr1 is described here for the first time. METHODS Animals Tritonia diomedea were collected from the waters of Puget Sound, Washington, and were maintained in natural running seawater (10–128C) at the University of Washington’s Friday Harbor Laboratories and in artificial seawater aquaria (Instant Ocean, 10–128C) in Houston and Chicago. Recording preparations For experiments where skin stimulation was used to elicit the swim motor program, animals were first anesthetized by injecting 60 ml of a solution composed of half 350 mM MgCl2 and half artificial seawater (In- 952 Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 SYNOPSIS. Two groups of interneurons, Tr1 and DRI, have been identified in the escape swim circuit of the marine mollusc Tritonia diomedea that have important roles in behavioral initiation. DRI functions as a command neuron, receiving direct excitatory input from the afferent neurons, and in turn directly exciting the DSI neurons of the central pattern generator. DRI fires throughout the swim motor program, and activity in DRI is both necessary and sufficient for sensory input to elicit the swim motor program. Tr1 is an excitatory interneuron that fires briefly in response to sensory input and then remains silent during the motor program. Tr1 excites DRI with an excitatory connection that has fast and slow components and thus appears to have a role in converting brief afferent neuron activity to long-lasting firing in downstream circuit elements. These neurons complete the description of a continuous synaptic pathway from afferent to flexion neurons in the Tritonia swim circuit. Their identification should facilitate studies of motor program initiation, as well as of how various forms of experience, including simple forms of learning, act to influence neuronal decision-making processes. SWIM INITATION NEURONS IN TRITONIA 953 stant Ocean, Aquarium Systems). The ventral foot was then cut along the midline from the tail to the buccal mass. After removing the internal organs, an opening was cut in the dorsal skin, above the brain. The animal was then positioned dorsal side up in the recording chamber, with the brain (cerebral, pleural and pedal ganglia) pinned out on the Sylgard surface of a 1-cm-diameter post rising from the chamber floor. A thin cylindrical sleeve, containing slits to allow the nerves passage, was raised around the brain, the slits were closed with Vaseline, and the brain and body chambers were perfused separately with normal saline. All other experiments utilized an isolated brain preparation. For both preparations, the brain was perfused at 28C while pinning the brain out and dissecting away the thin sheath enclosing the dorsal, cerebral and pleural ganglia. Suction electrodes were then placed on left and right pedal nerve 3, and the preparation was warmed to 10–118C for the duration of the experiment. Electrophysiology Neurons were impaled with glass microelectrodes (20–40 MV) filled with 3 M Kacetate and inserted into chucks containing 3 M KCl. Swim neurons were identified based on their location, size, color, synaptic connections with other identified neurons, and their activity during the swim motor program (Getting, 1976; Getting et al., 1980; Frost and Katz, 1996). Normal saline composition for both preparations was (in mM): 420 NaCl, 10 KCl, 10 CaCl2, 50 MgCl2, 11 D-glucose and 10 HEPES, pH 7.6. High divalent cation saline composition was (in mM): 285 NaCl, 10 KCl, 25 CaCl2, 125 MgCl2, 11 D-glucose, and 10 HEPES, pH 7.6. Data were initially stored on videotape using a Vetter 3000A PCM recording adapter, and were then digitized and analyzed offline using a Biopac MP100 data acquisition system. Tr1 was iontophoretically injected with 5, 6-carboxyfluorescein and photographed in the live preparation using an Optronics DEI-750D CCD camera Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 FIG. 1. The Tritonia escape swim and its underlying circuit. A. Upon contact with a suitably aversive stimulus, such as the tube feet of the seastar Pycnopodia helianthoides, Tritonia respond with an escape swim consisting of a series of alternating ventral and dorsal whole-body flexions. The photograph shows an animal at a moment of maximum dorsal flexion. B. The known swim circuit. Solid lines represent direct, monosynaptic connections, broken lines represent indirect connections, or connections not yet confirmed to be monosynaptic. Synaptic symbols: lines 5 excitatory, black circles 5 inhibitory, lines and circles 5 multiple component monosynaptic connections. ‘‘VSI’’ represents both VSI-A and VSI-B; the exact connectivity shown is for VSI-B only. The known number of neurons of each type on each side of the brain are: S-cells, ;80; Tr1, 1; DRI, 1; DSI, 3; C2, 1; VSI, 2; FNs, ;55 (Getting, 1983; Frost and Katz, 1996). 954 W. N. FROST ET AL. and a Kramer Scientific fluorescence illumination module attached to a Zeiss SV6 stereomicroscope. RESULTS DRI FIG. 2. The swim motor program elicited via DRI stimulation. DRI and three CPG interneurons (C2 and two DSIs) were impaled with intracellular electrodes. DRI was stimulated to fire at 10 Hz for 50 sec, resulting in a 6 cycle swim motor program. Calibration bars: All 20 mV, 8 sec. S-cells produced EPSPs onto DRI in high divalent cation saline. As reported previously (Frost and Katz, 1996), directly activating DRI using intracellular stimulation elicits the swim motor program (Fig. 2), while hyperpolarizing a single DRI prevents the nerve shock-elicited motor program. These findings satisfy the most stringent criteria for inclusion of DRI as a command neuron (see Discussion). In our prior work we only tested the ability of DRI hyperpolarization to block motor programs elicited by stimulation of Pedal Nerve 3. Here we report that hyperpolarizing DRI also blocks the initiation of motor programs elicited by salt stimuli applied to the skin (Fig. 3, n 5 3 preparations)—the stimulus used in many of our behavioral studies (Frost et al., 1996; Mongeluzi and Frost, 2000). Although DRI satisfies the formal criteria for assignment as a command neuron, the fact that it receives feedback from the CPG and its firing oscillates with the motor program raised the issue of whether it might actually function as a CPG member. Previous work had shown that injecting sufficient hyperpolarizing current to suppress firing in one of the two DRIs completely Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 For many years, a key missing element in the Tritonia swim network was the interneuronal population responsible for producing the long-lasting, declining ‘‘ramp’’ of depolarization in the dorsal swim interneurons (DSIs) of the CPG in response to a swim-eliciting stimulus (Lennard et al., 1980). Prior work (Getting and Dekin, 1985) concluded that this depolarization has an important role in initiating the swim motor program. Using a dye filled electrode, we searched for the missing ramp interneurons among the axons in the central commissure. This procedure led to the discovery of DRI, an interneuron that receives direct excitatory synaptic input from the afferent neurons (see below), and in turn makes strong, direct excitatory connections onto the DSIs of the CPG (Frost and Katz, 1996). The available data are consistent with there being just one DRI on each side of the brain (Frost and Katz, 1996). The discovery of DRI completed the description of a continuous synaptic pathway from the afferent to the efferent neurons of the swim circuit (Fig. 1b). An early report (Getting, 1977) suggested that the S-cells made a monosynaptic connection to CPG neuron Cerebral Cell 2 (C2). (3 of 8 cells were reported to do so.) In the present experiments we observed several indirect, but no direct connections between 58 S-cell–C2 pairs tested in 11 preparations using either saline or a high divalent cation saline solution effective at blocking polysynaptic pathways in Tritonia; (Katz and Frost, 1995b). We therefore suggest that the connections between the Scells and C2 are indirect. In contrast to their connections to C2, we found that the majority of S-cells made direct excitatory connections to DRI (Hoppe, 1998). In 10 preparations, 44 of 62 S-cells made 1-for-1, constant latency EPSPs onto DRI. In one of these preparations, 9 of 11 SWIM INITATION NEURONS IN TRITONIA 955 blocks the ability of a nerve stimulus to elicit the motor program (Frost and Katz, 1996). We reasoned that if DRI functions as a CPG member, suppressing its firing via hyperpolarization should also block oscillation initiated from within the CPG, such as can be elicited via intracellular stimulation of one or more DSI neurons (Fickbohm and Katz, 2000). On the other hand, if DRI functions more as a gating-command neuron that drives the CPG and also receives feedback from it, then suppressing DRI firing should not prevent DSI-driven CPG activity. We found that hyperpolarizing DRI failed to prevent the DSI-driven motor program (Fig. 4, n 5 5 preparations). This result supports our categorization of DRI as a pre-CPG command neuron that functions more to initiate and sustain the motor program than as part of the mechanism generating the cycle-by-cycle mechanics of the oscillation itself. Tr1 Although the discovery of DRI established a continuous afferent-to-flexion neuron synaptic pathway, the firing patterns of the cells suggested the existence of an asyet unidentified circuit element. Brief sen- sory stimulation elicits a correspondingly brief S-cell firing response, yet all previously identified downstream neurons in the swim circuit typically fire for tens of seconds (Figs. 3, 5). Since the S-cells excite DRI with fast EPSPs, something else seemed needed to mediate this brief-to-long firing transformation. We describe here a new neuron that appears to contribute to this process. The Tr1 neuron (n 5 20 preparations, Figs. 1B, 5) is located below the surface in the dorsal pleural ganglion, near DRI and the statocyst (Fig. 6A). Its defining features are: 1) the location of its cell body and its characteristic axonal morphology (Fig. 6A), 2) it receives direct EPSPs from the S-cells (28 of 28 cells tested in 10 preparations, Fig. 6B), 3) it fires briefly, like the S-cells, at the onset of the swim motor program (Fig. 5), 4) it makes an indirect excitatory connection to DSI (Fig. 6C), and 5) direct intracellular stimulation of a single Tr1 neuron is often sufficient to activate the motor program (Fig. 6D). Such Tr1-elicted motor programs typically continue 1 to 3 cycles beyond the end of intracellular stimulation (Avg. 1.5 6 0.3 extra cycles, n 5 8 preparations). This latter property suggests that Tr1 may con- Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 FIG. 3. DRI firing is necessary for the salt-elicited swim motor program. DSI and DRI were impaled with intracellular electrodes in the semi-intact preparation. Three consecutive salt stimuli (0.1 ml, 4 M NaCl) were applied to the tail 3–5 min apart (arrows). The first (left) and third (right) trials elicited swim motor programs. On the second (middle) trial, DRI was hyperpolarized to block its firing response to the salt. This prevented the swim motor program. The dotted line indicates the DRI resting potential before the hyperpolarization. Calibration bars: All 20 mV, 10 sec. 956 W. N. FROST tribute to the mechanism by which brief Scell discharges trigger long-lasting circuit excitation. To date, only one Tr1 neuron has been encountered on each side of the brain. However, hyperpolarizing Tr1 does not block the ability of nerve stimulation to elicit the swim motor program (n 5 2 preparations), nor does Tr1 stimulation elicit motor program activity as long lasting as that elicited by nerve stimulation, suggesting that there may be additional Tr1 neurons, or an additional class of Tr-like neurons on each side of the brain. We next tested whether Tr1’s indirect excitation of DSI (Fig. 6C) occurred via recruitment of DRI, the only known excitatory input to DSI originating from outside the CPG. This was a difficult experiment because Tr1 and DRI are both located out of FIG. 5. The S-cells and Tr1 fire briefly at the onset of the swim motor program. A brief stimulus (10 Hz, 1 sec) applied via suction electrode to Pedal Nerve 3 (arrow) elicited a brief firing train in the S-cell and Tr1 cell, followed by a three cycle swim motor program in CPG neurons DSI and C2. Calibration bars: All 20 mV, 10 sec. sight, below the surface of the pleural ganglion. We were successful in penetrating both DRI and Tr1 in two preparations, and found that Tr1 produced 1-for-1, constant latency EPSPs onto DRI (Fig. 7A). Significantly, brief Tr1 trains produced long-lasting firing in DRI (Fig. 7B). Hyperpolarizing DRI in normal saline revealed that the connection had both fast and slow components (Fig. 7A, C). These preliminary observations suggest that a key location for the brief-to-long firing transformation in the swim circuit may be a dual component fast/ slow excitatory synaptic connection between Tr1 and DRI. However further work is needed to test this hypothesis. Such dual component fast/slow synaptic connections mediate brief-to-long lasting firing transitions in other circuits as well (Brodfuehrer and Friesen, 1986b; Cleary and Byrne, 1993; Lieb and Frost, 1997). DISCUSSION While significant progress has been made in elucidating the neural basis of rhythmic pattern generation (Marder and Calabrese, 1996), less is known about the mechanisms Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 FIG. 4. DRI does not appear to function as a CPG element. DRI, C2 and 2 DSIs were impaled with intracellular electrodes. The two DSIs were stimulated to fire at 20 Hz for 20 sec (hatched bar) while DRI was hyperpolarized to prevent its spiking. This procedure failed to prevent the swim motor program, consistent with the view that DRI is not part of the cycle generating mechanism, but instead functions as a preCPG command neuron. During the swim motor program, C2, DSI and DRI received inhibition during the ventral phase, which was sufficient to phasically block firing in one of the two DSIs (DSI-1), leaving just the stimulus artifacts between the bursts. Calibration bars: All 20 mV, 10 sec. ET AL. SWIM INITATION NEURONS IN TRITONIA 957 of motor program initiation—the decisionmaking process that determines whether or not a particular behavioral act will occur. The Tritonia escape swim response is an attractive model system for investigating the neural mechanisms of behavioral initiation. The swim is an all-or-none response—the animal either swims or it doesn’t in response to an appropriate stimulus—so the nature of its decision is unambiguous. Also, the animal takes a variable amount of time (up to 20 sec) when deciding whether or not to swim, consistent with decision-making processes in higher animals. Progress toward understanding the swiminitiation process in Tritonia has come with the identification of the circuitry conveying sensory input to the swim CPG. A key question concerns how brief S-cell firing brings about a sustained activation of the CPG. Before the pre-CPG interneurons were identified, this was addressed by comparing the duration of depolarization in the DSI neurons when the nerve-elicited CPG oscillation was either allowed or prevented by hyperpolarizing C2 (Getting and Dekin, 1985). It was concluded that sensory input initiates a slowly declining ramp of depolarization in the DSIs that sustains the ini- Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 FIG. 6. Identifying characteristics of Tr1. A. Tr1 neuron injected with Carboxyfluorescein in the live preparation. The cell body is below the surface, and the axon can be seen travelling beneath the surface, out of focus, crossing the central commissure and entering the right side of the brain. S: statocyst; Pd: pedal ganglion; Ce: cerebral ganglion; Pl: pleural ganglion. B. S-cell excitatory synaptic connection to Tr1 in normal saline. C. Tr1 makes an indirect excitatory connection to DSI. A Tr1 train caused the tonically active DSI (DSI-1) to increase its firing rate. Holding a second DSI (DSI-2) hyperpolarized during the train revealed that the DSI excitation resulted from a flurry of EPSPs that did not follow the Tr1 action potentials one-for-one. D. A Tr1 train elicited by intracellular current injection (10 sec, mean 5 15.2 Hz) elicited a swim motor program that outlasted the train by 2 full cycles. Calibration bars: B. Tr1, 1 mV; S-cell, 20 mV; 500 msec. C. All 20 mV, 2 sec. D. All 20 mV, 10 sec. 958 W. N. FROST ET AL. tial 10–15 sec of oscillation. After that, excitatory interactions among the CPG neurons were suggested to sustain the remaining duration of the motor program. DRI was recently identified as the major source of the excitatory synaptic input to the DSIs (Frost and Katz, 1996). When CPG oscillation is suppressed by hyperpolarizing both C2 neurons, a brief nerve stimulus elicits a gradually slowing train of action potentials in DRI, and a corresponding declining ramp of depolarization in the DSIs (Frost and Katz, 1996). The excitatory ramp in the DSIs was found to be reduced or eliminated when DRI was hyperpolarized to prevent its firing. DRI was furthermore found to be individually necessary and sufficient for afferent input to activate the swim motor program (Figs. 3, 4 and (Frost and Katz, 1996)). These features satisfy the most stringent criteria for command neuron status—a cell that fires in response to sensory input and whose activity is both necessary and sufficient for the activation of a motor program by that input (Kupfermann and Weiss, 1978). A number of individual neurons have now been identified that satisfy these criteria (Rock et al., 1981; Fredman and Jahan-Parwar, 1983; Nolen and Hoy, 1984; Frost and Katz, 1996; Xin et al., 1996; Edwards et al., 1999; Jing and Gillette, 1999; Hedwig, 2000). In practice, however, the term ‘‘command neuron’’ has frequently been applied to neurons whose activity is simply sufficient to elicit a motor program, Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 FIG. 7. Tr1 to DRI connection. A. Tr1 connects to DRI with 1-for-1 constant latency EPSPs. B. A Tr1 train (2.5 sec, mean 5 22.6 Hz) produced several seconds of firing in DRI. C. Hyperpolarizing DRI during a Tr1 train revealed a dual component fast/slow EPSP in DRI. Calibration bars: A. DRI, 5 mV; Tr1, 20 mV; 1 sec. B. DRI, 10 mV; Tr1, 20 mV; 10 sec. C. DRI, 4 mV; Tr1, 20 mV; 1 sec. All records in normal saline. SWIM INITATION NEURONS TRITONIA 959 feedback connection from the CPG to its own command neuron may have an important role in initiating and sustaining the swim rhythm (Katz et al., 1994; Katz and Frost, 1997). The overall organization of the swim initiation circuitry in Tritonia appears to be similar to that described for swimming in the leech (Brodfuehrer et al., 1995). Both species direct sensory input to trigger-type neurons that in turn excite gating command neurons that activate and sustain CPG activity. In both cases, as in others (Gillette et al., 1978; Fredman and Jahan-Parwar, 1983; McCrohan, 1984; Nolen and Hoy, 1984; Delaney and Gelperin, 1990; Jing and Gillette, 1999) the gating neurons also receive synaptic feedback from the pattern generator during the rhythm. These similarities across species reinforce the idea that this circuit structure for the control of motor program initiation may be commonly employed for the control of many stimuluselicited, rhythmic motor programs, such as those mediating escape responses. ACKNOWLEDGMENTS We wish to thank Richard Satterlie for organizing the symposium, and Lise Eliot and Ion Popescu for comments on the manuscript. This work was supported by NS36500. The symposium was supported by National Science Foundation grant BN 9905990. REFERENCES Brodfuehrer, P. D., E. A. Debski, B. A. O’Gara, and W. O. Friesen. 1995. Neuronal control of leech swimming. J. Neurobiol. 27:403–418. Brodfuehrer, P. D. and W. O. Friesen. 1986a. From stimulation to undulation: A neuronal pathway for the control of swimming in the leech. Science 234:1002–1004. Brodfuehrer, P. D. and W. O. Friesen. 1986b. Initiation of swimming activity by trigger neurons in the leech subesophageal ganglion. II. Role of segmental swim-initiating interneurons. J. Comp. Physiol. [A] 159:503–510. Cleary, L. J. and J. H. Byrne. 1993. Identification and characterization of a multifunction neuron contributing to defensive arousal in Aplysia. J. Neurophysiol. 70:1767–1776. Delaney, K. and A. Gelperin. 1990. Cerebral interneurons controlling fictive feeding in Limax maximus: II. Initiation and modulation of fictive feeding. J. Comp. Physiol. [A] 166:311–326. Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 without the requirement of individual necessity. Stein divided command neurons into two functional classes, trigger and gating neurons (Stein, 1978). Trigger neurons fire at the outset, and are then silent during the sensory-elicited motor program. Their role is therefore to initiate motor program activity, often by exciting gating command neurons. Gating command neurons, such as DRI, fire throughout the motor program and are important for sustaining rhythmic motor program generation. When driven directly, these neurons elicit a motor program that ceases shortly after the end of the eliciting train (see Fig. 2). Tr1 has several properties common to trigger command neurons, such as those described in the leech (Brodfuehrer and Friesen, 1986a) and crayfish (Murchison and Larimer, 1990). It fires briefly at motor program onset, due to direct, highly convergent, fast excitatory connections from the S-cells, and is then silent during the program itself. When driven directly, a single Tr1 can often elicit motor program activity, and this activity continues for a period of time after the end of the train. Unlike DRI, however, Tr1 fails the test of individual necessity—hyperpolarizing Tr1 does not block motor program activation by sensory input (nerve stimulation). Tr1’s ability to elicit a motor program that outlasts the Tr1 train is also modest, unlike the trigger neurons that initiate leech swimming (Brodfuehrer and Friesen, 1986a). These results suggest that there are additional, as yet unidentified Tr1-type neurons that work together and reinforce one another in initiating the swim, perhaps functioning as a trigger ‘‘command system.’’ Tr1 appears to have a role in converting brief S-cell firing into long lasting DRI firing, which in turn strongly excites all 6 DSI neurons of the swim CPG. The DSI neurons contain 5HT and produce neuromodulatory enhancement of the excitability and synaptic efficacy of CPG neuron C2 (Katz et al., 1994; Katz and Frost, 1995a, b, 1997). The synaptic enhancement involves all chemical synaptic connections made by C2, including those within the CPG and an excitatory feedback connection from C2 to DRI. The modulatory enhancement of this positive IN 960 W. N. FROST for escape swimming in the notaspid sea slug Pleurobranchaea californica. J. Neurophysiol. 81: 654–667. Katz, P. S. and W. N. Frost. 1995a. Intrinsic neuromodulation in the Tritonia swim CPG: Serotonin mediates both neuromodulation and neurotransmission by the dorsal swim interneurons. J. Neurophysiol. 74:2281–2294. Katz, P. S. and W. N. Frost. 1995b. Intrinsic neuromodulation in the Tritonia swim CPG: The serotonergic dorsal swim interneurons act presynaptically to enhance transmitter release from interneuron C2. J. Neurosci. 15:6035–6045. Katz, P. S. and W. N. Frost. 1997. Removal of spike frequency adaptation via neuromodulation intrinsic to the Tritonia escape swim central pattern generator. J. Neurosci. 17:7703–7713. Katz, P. S., P. A. Getting, and W. N. Frost. 1994. Dynamic neuromodulation of synaptic strength intrinsic to a central pattern generator circuit. Nature (London) 367:729–731. Kupfermann, I. and K. R. Weiss. 1978. The command neuron concept. Behav. Brain Sci. 1:3–39. Lennard, P. R., P. A. Getting, and R. I. Hume. 1980. Central pattern generator mediating swimming in Tritonia. II. Initiation, maintenance, and termination. J. Neurophysiol. 44:165–173. Lieb, J. R., Jr. and W. N. Frost. 1997. Realistic simulation of the Aplysia siphon-withdrawal reflex circuit: Roles of circuit elements in producing motor output. J. Neurophysiol. 77:1249–1268. Marder, E. and R. L. Calabrese. 1996. Principles of rhythmic motor pattern generation. Physiol. Rev. 76:687–717. McCrohan, C. R. 1984. Initiation of feeding motor output by an identified interneurone in the snail Lymnaea stagnalis. J. Exp. Biol. 113:351–366. Mongeluzi, D. L. and W. N. Frost. 2000. Dishabituation of the Tritonia escape swim. Learn Mem 7: 43–47. Murchison, D. and J. L. Larimer. 1990. Dual motor output interneurons in the abdominal ganglia of the crayfish Procambarus clarkii: Synaptic activation of motor outputs in both the swimmeret and abdominal positioning systems by single interneurons. J. Exp. Biol. 150:269–293. Nolen, T. G. and R. R. Hoy. 1984. Initiation of behavior by single neurons: The role of behavioral context. Science 226:992–994. Rock, M. K., J. T. Hackett, and D. L. Brown. 1981. Does the Mauthner cell conform to the criteria of the command neuron concept? Brain Research 204:21–27. Slawsky, M. T. 1979. Presynaptic inhibition in the marine mollusk, Tritonia diomedea. Ph.D. Diss., Stanford University. Stein, P. S. G. 1978. Motor systems, with specific reference to the control of locomotion. Ann. Rev. Neurosci. 1:61–81. Willows, A. O. D., D. A. Dorsett, and G. Hoyle. 1973. The neuronal basis of behavior in Tritonia. I. Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014 Dorsett, D. A., A. O. D. Willows, and G. Hoyle. 1969. Centrally generated nerve impulse sequences determining swimming behavior in Tritonia. Nature (London) 224:711–712. Edwards, D. H., W. J. Heitler, and F. B. Krasne. 1999. Fifty years of a command neuron: The neurobiology of escape behavior in the crayfish. Trends Neurosci. 22:153–161. Fickbohm, D. J. and P. S. Katz. 2000. Paradoxical actions of the serotonin precursor 5-hydroxytryptophan on the activity of identified serotonergic neurons in a simple motor circuit. J. Neurosci. 20: 1622–1634. Fredman, S. M. and B. Jahan-Parwar. 1983. Command neurons for locomotion in Aplysia. J. Neurophysiol. 49:1092–1117. Frost, W. N. 1999. Use of intact animal preparations to evaluate the role of afferent neuron firing in habituation and swim pattern generation in Tritonia. Soc. Neurosci. Abstr. 25:1643. Frost, W. N., G. Brown, and P. A. Getting. 1996. Parametric features of habituation of swim cycle number in the marine mollusc Tritonia diomedea. Neurobiol. of Learning and Memory 65:125–134. Frost, W. N. and P. S. Katz. 1996. Single neuron control over a complex motor program. Proc. Nat. Acad. Sci. U.S.A. 93:422–426. Getting, P. A. 1976. Afferent neurons mediating escape swimming of the marine mollusc, Tritonia. J. Comp. Physiol. [A] 110:271–286. Getting, P. A. 1977. Neuronal organization of escape swimming in Tritonia. J. Comp. Physiol. [A] 121: 325–342. Getting, P. A. 1983. Neural control of swimming in Tritonia. Symp. Soc. Exp. Biol. 37:89–128. Getting, P. A. and M. S. Dekin. 1985. Mechanisms of pattern generation underlying swimming in Tritonia. IV. Gating of central pattern generator. J. Neurophysiol. 53:466–480. Getting, P. A., P. R. Lennard, and R. I. Hume. 1980. Central pattern generator mediating swimming in Tritonia. I. Identification and synaptic interactions. J. Neurophysiol. 44:151–164. Gillette, R., M. P. Kovac, and W. J. Davis. 1978. Command neurons in Pleurobranchaea receive synaptic feedback from the motor network they excite. Science 199:798–801. Hedwig, B. 2000. Control of cricket stridulation by a command neuron: Efficacy depends on the behavioral state. J. Neurophysiol. 83:712–722. Hoppe, T. A. 1998. An evaluation of the role of synaptic depression at afferent synapses in habituation of the escape swim response of Tritonia diomedea. Masters Thesis, The University of Texas Health Science Center at Houston. Hume, R. I., P. A. Getting, and M. A. Del Beccaro. 1982. Motor organization of Tritonia swimming. I. Quantitative analysis of swim behavior and flexion neuron firing patterns. J. Neurophysiol. 47:60– 74. Jing, J. and R. Gillette. 1999. Central pattern generator ET AL. SWIM INITATION NEURONS Functional organization of the central nervous system. J. Neurobiol. 4:207–237. Xin, Y., K. R. Weiss, and I. Kupfermann. 1996. A pair of identified interneurons in Aplysia that are in- IN TRITONIA 961 volved in multiple behaviors are necessary and sufficient for the arterial-shortening component of a local withdrawal reflex. J. Neurosci. 16:4518– 4528. Downloaded from http://icb.oxfordjournals.org/ at University of Texas at Austin on September 15, 2014