Physical and Chemical Changes
... b. some mass is converted to energy c. some form of light is given off d. a new material is formed ...
... b. some mass is converted to energy c. some form of light is given off d. a new material is formed ...
Chapter 14 Chemical Reactions
... reacted in a closed container, you can show that the mass before and after the reaction is the same. ...
... reacted in a closed container, you can show that the mass before and after the reaction is the same. ...
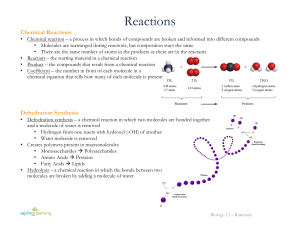
Reactions
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
Classification of Matter
... A subscript is placed to the lower right of the chemical symbol It gives the number of atoms of the element ...
... A subscript is placed to the lower right of the chemical symbol It gives the number of atoms of the element ...
Classifying Chemical Reactions 9-3
... Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
... Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
CHEMICAL EQUATIONS NAME PERIOD_______ DATE________
... reaction. In a chemical equation, the substances on the left side of the arrow are the starting substances. These substances are called ______________. The substances on the right side of the arrow are the substances that result from the reaction. These substances are called ____________________. Th ...
... reaction. In a chemical equation, the substances on the left side of the arrow are the starting substances. These substances are called ______________. The substances on the right side of the arrow are the substances that result from the reaction. These substances are called ____________________. Th ...
Nothing Lost, Nothing Gained
... change so that they look very different, but the amount of stuff stays the same. A chemical reaction is what happens when one kind of thing changes into another kind of thing. The same number of atoms are there, but they are changed into something new. If I burn a book, all the parts that made up th ...
... change so that they look very different, but the amount of stuff stays the same. A chemical reaction is what happens when one kind of thing changes into another kind of thing. The same number of atoms are there, but they are changed into something new. If I burn a book, all the parts that made up th ...
FHN - Chemical and Physical Changes
... change, but the substances in the material stay the same. Change in state Solid melting to a liquid Liquid evaporating to a gas Gas condensing to a liquid Liquid freezing into a solid Usually occur with a change in temperature Can also be when a substance dissolves in a liquid, but doe ...
... change, but the substances in the material stay the same. Change in state Solid melting to a liquid Liquid evaporating to a gas Gas condensing to a liquid Liquid freezing into a solid Usually occur with a change in temperature Can also be when a substance dissolves in a liquid, but doe ...
Notes for Types of Reactions:
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
*6th Grade Science-Chapter 5 Study Guide Lesson 5.1: Observing
... A precipitate is a solid that forms from liquids that undergo chemical changes in a chemical reaction. A gas can form from a solid or liquid as a result of chemical changes. A color change can occur as a result of chemical changes. Exothermic reaction- net energy is released from a chemical reaction ...
... A precipitate is a solid that forms from liquids that undergo chemical changes in a chemical reaction. A gas can form from a solid or liquid as a result of chemical changes. A color change can occur as a result of chemical changes. Exothermic reaction- net energy is released from a chemical reaction ...
File - Flipped Out Science with Mrs. Thomas!
... used to classify elements 2.2 Chemical Equations 5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances 5F recognize whether a chemical equation containing coefficients is balanced or not and ho ...
... used to classify elements 2.2 Chemical Equations 5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances 5F recognize whether a chemical equation containing coefficients is balanced or not and ho ...
File - Flipped Out Science with Mrs. Thomas!
... used to classify elements 2.2 Chemical Equations 5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances 5F recognize whether a chemical equation containing coefficients is balanced or not and ho ...
... used to classify elements 2.2 Chemical Equations 5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances 5F recognize whether a chemical equation containing coefficients is balanced or not and ho ...
Chemical Building Blocks Chapter One
... Characteristic Property: a quality of a substance that never changes and can be used to identify the substances (pg. 15) Boiling Point: the temperature at which a substance changes from a liquid to a gas (pg. 16) Physical Change: a change in a substance that does not change its identity; for example ...
... Characteristic Property: a quality of a substance that never changes and can be used to identify the substances (pg. 15) Boiling Point: the temperature at which a substance changes from a liquid to a gas (pg. 16) Physical Change: a change in a substance that does not change its identity; for example ...
Topic2890 Thermodynamics and Kinetics A given system at
... reaction. In fact the link between the rate of chemical reaction (dξ / dt ) and the affinity for spontaneous change A is intuitively attractive. However while one may monitor the dependence of composition on time, dξ/dt, it is not immediately obvious ∂A how one might estimate the affinity A and ...
... reaction. In fact the link between the rate of chemical reaction (dξ / dt ) and the affinity for spontaneous change A is intuitively attractive. However while one may monitor the dependence of composition on time, dξ/dt, it is not immediately obvious ∂A how one might estimate the affinity A and ...
Introductory Chemistry Test Review
... 25. In the laboratory, potassium chlorate will decompose when heated to form potassium chloride and oxygen gas according to the following equation. Calculate how much oxygen in grams is produced when 35.0 grams of potassium chlorate decomposes. 2 KClO3(s) ...
... 25. In the laboratory, potassium chlorate will decompose when heated to form potassium chloride and oxygen gas according to the following equation. Calculate how much oxygen in grams is produced when 35.0 grams of potassium chlorate decomposes. 2 KClO3(s) ...
Reading-Chem v Phys
... remains the same. A change in temperature may also bring about a physical change. The melting of ice, the freezing of water, the conversion of water to steam, and the condensation of steam to water are all examples of physical changes. We know that these physical properties of water are the same for ...
... remains the same. A change in temperature may also bring about a physical change. The melting of ice, the freezing of water, the conversion of water to steam, and the condensation of steam to water are all examples of physical changes. We know that these physical properties of water are the same for ...
Atoms, Elements, Compounds File
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
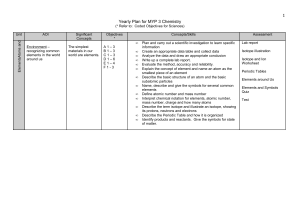
Yearly Plan for MYP 1 Science
... environment and that we cannot survive without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
... environment and that we cannot survive without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
Chemistry Review: Unit2 - Menno Simons Christian School
... What is the difference between a molecule and diatomic molecule? Diatomic molecules have to be made up of the same element whereas molecules do not. Topic 7: 19) Express the following chemical reaction in words: ...
... What is the difference between a molecule and diatomic molecule? Diatomic molecules have to be made up of the same element whereas molecules do not. Topic 7: 19) Express the following chemical reaction in words: ...
Chemistry! - Duplin County Schools
... – Ductile – can be stretched into a wire without breaking – Conductor – can conduct heat or electricity ...
... – Ductile – can be stretched into a wire without breaking – Conductor – can conduct heat or electricity ...
673 lab three
... A) DISCUSS CHEMICAL REACTIONS: start with a definition and apply the law of conservation of mass in a chemical reaction to the reaction in this lab. Discuss balanced reactions and give three example reactions and SHOW that they are balanced. Clearly indicate the role of COEFFICIENTS., B) DISCUSS CHE ...
... A) DISCUSS CHEMICAL REACTIONS: start with a definition and apply the law of conservation of mass in a chemical reaction to the reaction in this lab. Discuss balanced reactions and give three example reactions and SHOW that they are balanced. Clearly indicate the role of COEFFICIENTS., B) DISCUSS CHE ...
Chapter 15 - cloudfront.net
... Water boiling or freezing Color changes Cutting paper into smaller pieces • Stretching a rubber band • Grass growing ...
... Water boiling or freezing Color changes Cutting paper into smaller pieces • Stretching a rubber band • Grass growing ...
Chemistry Unit Study Guide Key
... 6) difference between physical and chemical changes – In a physical change, nothing new is formed. In a chemical change, a new substance is formed. 7) what it means if elements are in the same family/group – They have similar properties. 8) how to find elements on the periodic table – You can use th ...
... 6) difference between physical and chemical changes – In a physical change, nothing new is formed. In a chemical change, a new substance is formed. 7) what it means if elements are in the same family/group – They have similar properties. 8) how to find elements on the periodic table – You can use th ...
CHEMICAL REACTION
... precipitate (solid forming from solutions) reaction goes both ways, forward and reverse ...
... precipitate (solid forming from solutions) reaction goes both ways, forward and reverse ...
Chemical Corps

The Chemical Corps is the branch of the United States Army tasked with defending against chemical, biological, radiological, and nuclear (CBRN) weapons. The corps was founded as the Chemical Warfare Service (CWS) during World War I. Its name was changed to the Chemical Corps in 1946.