CHAPTER 1 Practice Exercises 1.1 12.3 g Cd 1.3 26.9814 u 1.5
... Silver and gold are in the same periodic table group as copper, so they might well be expected to occur together in nature, because of their similar properties and tendencies to form similar compounds. ...
... Silver and gold are in the same periodic table group as copper, so they might well be expected to occur together in nature, because of their similar properties and tendencies to form similar compounds. ...
Chapter #3
... There are N of the above equations, one for each element (atom type) in the reaction. Generally there are M coefficients to find using the N equations. Unfortunately, in most chemical equations, M > N. Usually, we have the case that M = N+1. Thus, we need to find one additional equation. One simple ...
... There are N of the above equations, one for each element (atom type) in the reaction. Generally there are M coefficients to find using the N equations. Unfortunately, in most chemical equations, M > N. Usually, we have the case that M = N+1. Thus, we need to find one additional equation. One simple ...
13.2 Chemical Formulas
... Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elements hydrogen (H) and oxygen ( ...
... Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elements hydrogen (H) and oxygen ( ...
classification of chemical reactions
... A new substance is formed Na + Cl NaCl Gas production (bubbles) 2H2O2 H2O + O2 Color change Temperature change (endothermic vs. exothermic) Precipitate forms – solid substance that forms when two substances mix ...
... A new substance is formed Na + Cl NaCl Gas production (bubbles) 2H2O2 H2O + O2 Color change Temperature change (endothermic vs. exothermic) Precipitate forms – solid substance that forms when two substances mix ...
File
... Atoms can’t be broken into smaller pieces. Atoms are indivisible. In any element, all the atoms are exactly alike. Atoms of different elements are different. Atoms of two or more elements can combine to form compounds. Atoms of each element have a unique mass. The masses of the elements in a compoun ...
... Atoms can’t be broken into smaller pieces. Atoms are indivisible. In any element, all the atoms are exactly alike. Atoms of different elements are different. Atoms of two or more elements can combine to form compounds. Atoms of each element have a unique mass. The masses of the elements in a compoun ...
ap chemistry – 2013-2014
... taking observations and data manipulation. Students are to communicate and collaborate in lab groups (ideally working in pairs); however, each student writes a laboratory report in a lab notebook for every lab they perform. It is important that students do not procrastinate when doing pre-lab and po ...
... taking observations and data manipulation. Students are to communicate and collaborate in lab groups (ideally working in pairs); however, each student writes a laboratory report in a lab notebook for every lab they perform. It is important that students do not procrastinate when doing pre-lab and po ...
Chapter 2 Outline
... G. A change in the number of electrons results in an ion IV. Compounds and mixtures A. When two or more atoms bond covalently, they form a molecule B. A compound is formed when two or more different atoms bond chemically C. A mixture occurs when compounds can be separated by non-chemical means NO CH ...
... G. A change in the number of electrons results in an ion IV. Compounds and mixtures A. When two or more atoms bond covalently, they form a molecule B. A compound is formed when two or more different atoms bond chemically C. A mixture occurs when compounds can be separated by non-chemical means NO CH ...
1 - mvhs-fuhsd.org
... the changes it undergoes. 4. What branch of chemistry is most concerned with the study of carbon compounds? Organic Chemistry 5. What does the word chemical, as used by scientists ,mean? 6. Briefly describe the difference between basic research, applied research, and technological development. Provi ...
... the changes it undergoes. 4. What branch of chemistry is most concerned with the study of carbon compounds? Organic Chemistry 5. What does the word chemical, as used by scientists ,mean? 6. Briefly describe the difference between basic research, applied research, and technological development. Provi ...
Reactive Materials - NC State University
... The reactivity of inorganic compounds may be frequently correlated with their “family” in the periodic table. Within a given family, similar types of behavior are observed with changes in the magnitude of reactivity varying consistently with atomic weight. The reactivity of chemical compounds shows ...
... The reactivity of inorganic compounds may be frequently correlated with their “family” in the periodic table. Within a given family, similar types of behavior are observed with changes in the magnitude of reactivity varying consistently with atomic weight. The reactivity of chemical compounds shows ...
A Study of Matter
... • Boiling point- liquid turns to a gas (water to water vapor) • Condensation- where a gas turns to a liquid (the sweating on a glass that is colder than it’s environment) • Sublimation point- temperature at which a solid changes directly to a gas without first changing into a liquid. (dry ice) ...
... • Boiling point- liquid turns to a gas (water to water vapor) • Condensation- where a gas turns to a liquid (the sweating on a glass that is colder than it’s environment) • Sublimation point- temperature at which a solid changes directly to a gas without first changing into a liquid. (dry ice) ...
The retrospect of the science and the thermodynamics
... Using a number to tell us how free of a problems. ...
... Using a number to tell us how free of a problems. ...
chemical reaction
... Write a balanced chemical equation for the reaction described below. When solid copper metal is placed in a solution of silver (I) nitrate, silver metal precipitates out and a solution of copper (II) nitrate is formed. Chemical Equation: Cu(s) + AgNO3(aq) → Ag(s) + Cu(NO3)2 Balanced Chemical Equatio ...
... Write a balanced chemical equation for the reaction described below. When solid copper metal is placed in a solution of silver (I) nitrate, silver metal precipitates out and a solution of copper (II) nitrate is formed. Chemical Equation: Cu(s) + AgNO3(aq) → Ag(s) + Cu(NO3)2 Balanced Chemical Equatio ...
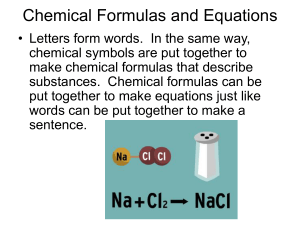
Chemical Formulas and Equations
... The Importance of Accuracy • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
... The Importance of Accuracy • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
Balancing chemical equations notes
... Balancing Chemical Equations Chemical equations can be viewed as recipes for chemical reactions. They give a description of what chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, ...
... Balancing Chemical Equations Chemical equations can be viewed as recipes for chemical reactions. They give a description of what chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, ...
Standard Operating Procedure Template
... This Standard Operating Procedure must be placed in the Chemical Hygiene Plan and the SDS must be accessible. Also, all laboratory personnel must be familiar with safe handling practices (i.e., training with documentation of training) when working with these chemicals. This must be incorporated int ...
... This Standard Operating Procedure must be placed in the Chemical Hygiene Plan and the SDS must be accessible. Also, all laboratory personnel must be familiar with safe handling practices (i.e., training with documentation of training) when working with these chemicals. This must be incorporated int ...
Docking
... Small Molecule/Ligand (Similarity) Predictive Methods (Kernel Methods) Why it is not hopeless ...
... Small Molecule/Ligand (Similarity) Predictive Methods (Kernel Methods) Why it is not hopeless ...
Introduction to Chemical Reactions
... Chemical Reactions are represented by Chemical Equations. Chemical Equations are balanced to show the same number of atoms of each element on each side. The Law of Conservation of Mass says that atoms won’t be created or destroyed in a chemical reaction. That is why you have to balance chemical equa ...
... Chemical Reactions are represented by Chemical Equations. Chemical Equations are balanced to show the same number of atoms of each element on each side. The Law of Conservation of Mass says that atoms won’t be created or destroyed in a chemical reaction. That is why you have to balance chemical equa ...
Balancing Equations
... the formulas of the reactants (on the left) are connected by an arrow with the formulas for the products (on the right). • Example: Reactants Products ...
... the formulas of the reactants (on the left) are connected by an arrow with the formulas for the products (on the right). • Example: Reactants Products ...
Name__________________________________ Block______
... 3. (Breaking, Melting, Burning) is an example of a chemical change. 4. (Lemonade, Iced tea, Water) is not a mixture. 5. (Density, Burning, Color) is not a physical property. 6. Iron exists as a (solid, liquid, gas) at room temperature. 7. At room temperature, helium is usually a (liquid, gas, solid) ...
... 3. (Breaking, Melting, Burning) is an example of a chemical change. 4. (Lemonade, Iced tea, Water) is not a mixture. 5. (Density, Burning, Color) is not a physical property. 6. Iron exists as a (solid, liquid, gas) at room temperature. 7. At room temperature, helium is usually a (liquid, gas, solid) ...
Discover Chemical Changes - gk-12
... with before and after chemical changes so that students can make the chemical changes happen themselves or at least make observations of chemical changes that have happened at each station. I have listed above 9 possible chemical changes that can be used. Students should move from station to station ...
... with before and after chemical changes so that students can make the chemical changes happen themselves or at least make observations of chemical changes that have happened at each station. I have listed above 9 possible chemical changes that can be used. Students should move from station to station ...
Matter Change
... Carbon – black, tasteless solid Oxygen – colorless, tasteless gas Hydrogen – colorless, tasteless gas ...
... Carbon – black, tasteless solid Oxygen – colorless, tasteless gas Hydrogen – colorless, tasteless gas ...
SCIENCE 9
... has its own distinct properties and cannot be broken down into simpler substances by means of a chemical change. COMPOUNDS- are pure substances that are made up of two or more elements chemically combined together. Compounds can be broken down into elements again by chemical means ...
... has its own distinct properties and cannot be broken down into simpler substances by means of a chemical change. COMPOUNDS- are pure substances that are made up of two or more elements chemically combined together. Compounds can be broken down into elements again by chemical means ...
Cosmetology Learning Module 12
... matter and how matter changes under different chemical conditions Organic Chemistry – is the study of substances that contain carbon All living things are made up of compounds that contain carbon Organic compounds will burn ...
... matter and how matter changes under different chemical conditions Organic Chemistry – is the study of substances that contain carbon All living things are made up of compounds that contain carbon Organic compounds will burn ...
Chemical Corps

The Chemical Corps is the branch of the United States Army tasked with defending against chemical, biological, radiological, and nuclear (CBRN) weapons. The corps was founded as the Chemical Warfare Service (CWS) during World War I. Its name was changed to the Chemical Corps in 1946.