Document
... i. To make writing names of the elements simple, chemists use abbreviations, or element symbols. ii. Many of these symbols consist of the first letter or the first two letters of the element name. c. 3.3 Dalton’s Atomic Theory i. In the early 1800’s English scientist John Dalton came up with an expl ...
... i. To make writing names of the elements simple, chemists use abbreviations, or element symbols. ii. Many of these symbols consist of the first letter or the first two letters of the element name. c. 3.3 Dalton’s Atomic Theory i. In the early 1800’s English scientist John Dalton came up with an expl ...
chapter 3 notes for power point
... • According to Bohr’s model, electrons can be only certain distances from the nucleus. Each distance corresponds to a certain quantity of energy that an electron can have. • An electron that is as close to the nucleus as it can be is in its lowest energy level. • The farther an electron is from the ...
... • According to Bohr’s model, electrons can be only certain distances from the nucleus. Each distance corresponds to a certain quantity of energy that an electron can have. • An electron that is as close to the nucleus as it can be is in its lowest energy level. • The farther an electron is from the ...
Chapter 1: Fundamental Concepts
... --Matter consists of tiny particles called atoms. --Atoms are indestructible. In chemical reactions, the atoms rearrange but they do not themselves break apart. --In any sample of a pure element, all the atoms are identical in mass and other properties. --The atoms of different elements differ in ma ...
... --Matter consists of tiny particles called atoms. --Atoms are indestructible. In chemical reactions, the atoms rearrange but they do not themselves break apart. --In any sample of a pure element, all the atoms are identical in mass and other properties. --The atoms of different elements differ in ma ...
Matter: properties and characteristics COPY
... A student divides several cubes into two groups, based on whether or not each cube can float in water. Which property is the student using to classify the cubes? a. weight b. density c. conductivity d. mass ...
... A student divides several cubes into two groups, based on whether or not each cube can float in water. Which property is the student using to classify the cubes? a. weight b. density c. conductivity d. mass ...
Atoms and quantum phenomena
... • (starting with Thorium, Neptunium, Uranium, Actinium) • spontaneous nuclear reactions: mother, daughter, radiation emitted • (with time) all 4 series end at Pb (lead) [Pb is nice and stable] ...
... • (starting with Thorium, Neptunium, Uranium, Actinium) • spontaneous nuclear reactions: mother, daughter, radiation emitted • (with time) all 4 series end at Pb (lead) [Pb is nice and stable] ...
PPT
... Example: Nuclear Spin and MRI Magnetic resonance imaging (MRI) depends on the absorption of electromagnetic radiation by the nuclear spin of the hydrogen atoms in our bodies. The nucleus is a proton with spin ½, so in a magnetic field B there are two energy states. The proton’s magnetic moment is m ...
... Example: Nuclear Spin and MRI Magnetic resonance imaging (MRI) depends on the absorption of electromagnetic radiation by the nuclear spin of the hydrogen atoms in our bodies. The nucleus is a proton with spin ½, so in a magnetic field B there are two energy states. The proton’s magnetic moment is m ...
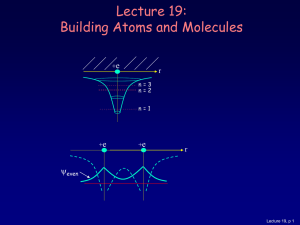
Lecture 19: Building Atoms and Molecules
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
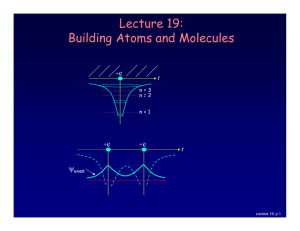
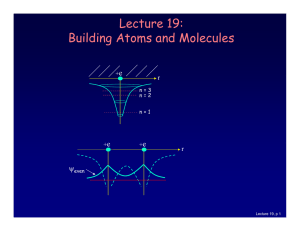
Lecture 19: Building Atoms and Molecules
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
Chapter 1
... A. Protons have an elementary positive charge of 1. B. Protons have one Dalton of mass. C. Protons are always found in the nucleus of the atom. D. Any atom found in nature always has the same number of protons as electrons. 2. Which of the following statements about electron orbitals is FALSE? A. Th ...
... A. Protons have an elementary positive charge of 1. B. Protons have one Dalton of mass. C. Protons are always found in the nucleus of the atom. D. Any atom found in nature always has the same number of protons as electrons. 2. Which of the following statements about electron orbitals is FALSE? A. Th ...
Nuclear Structure - UNLV Radiochemistry
... • Odd-A Nuclei In odd A nucleus of all but one of the nucleons considered to have their angular momenta paired off forming even-even core single odd nucleon moves essentially independently in this core net angular momentum of entire nucleus determined by quantum state of single odd nucleon • Co ...
... • Odd-A Nuclei In odd A nucleus of all but one of the nucleons considered to have their angular momenta paired off forming even-even core single odd nucleon moves essentially independently in this core net angular momentum of entire nucleus determined by quantum state of single odd nucleon • Co ...
File
... B) Two different compounds formed from carbon and oxygen have the following mass ratios: 1.33 g O: 1 g C and 2.66 g O: 1 g C. C) Two different samples of table salt are found to have the same ratio of sodium to chlorine. D) The atomic mass of bromine is found to be 79.90 amu. E) Nitrogen dioxide alw ...
... B) Two different compounds formed from carbon and oxygen have the following mass ratios: 1.33 g O: 1 g C and 2.66 g O: 1 g C. C) Two different samples of table salt are found to have the same ratio of sodium to chlorine. D) The atomic mass of bromine is found to be 79.90 amu. E) Nitrogen dioxide alw ...
chemistry SLO content practice
... a) atomic mass b) mass number c) atomic number d) isotope 9. _____ What is the average mass of atoms of a given element? a)atomic mass b)mass number c)atomic number d)isotope 10. _____ What is the mass of one atom of a given element? a) atomic mass b) mass number c) atomic number d) isotope 11. ____ ...
... a) atomic mass b) mass number c) atomic number d) isotope 9. _____ What is the average mass of atoms of a given element? a)atomic mass b)mass number c)atomic number d)isotope 10. _____ What is the mass of one atom of a given element? a) atomic mass b) mass number c) atomic number d) isotope 11. ____ ...
ψ 2
... The nucleus is the very dense region consisting of nucleons (protons and neutrons) at the center of an atom. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus, with a very small contribution from the orbiting electrons. The diameter of the nucleus is in t ...
... The nucleus is the very dense region consisting of nucleons (protons and neutrons) at the center of an atom. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus, with a very small contribution from the orbiting electrons. The diameter of the nucleus is in t ...
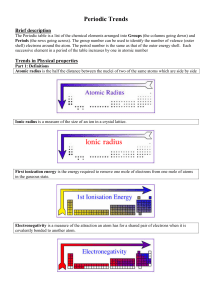
Topic 3 Periodicity notes SL - Chemical Minds
... i) there is a decrease in the electrostatic attraction between the positive protons in the nucleus and the negative electrons in the electron shells orbiting the nucleus and ii) the valence electrons are shielded by the inner electron shells Shielding means that there is an increase in electron-elec ...
... i) there is a decrease in the electrostatic attraction between the positive protons in the nucleus and the negative electrons in the electron shells orbiting the nucleus and ii) the valence electrons are shielded by the inner electron shells Shielding means that there is an increase in electron-elec ...
Activation parameters for ET
... Due to entropy mixing, the slope of the pH-dependence should be negative. Observed (total) activation entropy change The slope of the pH-dependence of the observed (total) entropy change is positive (or in some cases slightly negative) Our data indicate that the magnitude of ΔGET is at least as larg ...
... Due to entropy mixing, the slope of the pH-dependence should be negative. Observed (total) activation entropy change The slope of the pH-dependence of the observed (total) entropy change is positive (or in some cases slightly negative) Our data indicate that the magnitude of ΔGET is at least as larg ...
Physical or Chemical Property?
... compacted in the tiny positively charged nucleus accounting for most of the mass of the atom • The negatively charged electrons are small and have a relatively small mass but occupy a large volume of space outside the nucleus ...
... compacted in the tiny positively charged nucleus accounting for most of the mass of the atom • The negatively charged electrons are small and have a relatively small mass but occupy a large volume of space outside the nucleus ...
Chemistry primer Atom = the smallest unit of an element Element
... Ion: in an ion the number of e- is not equal to the number of P+ Charge = number of P+ minus number of eAtomic weight is equal to the number of protons plus the number of neutrons Atomic number si equal to the number protons Atomic number determines the identity of the atom or ion. In other words at ...
... Ion: in an ion the number of e- is not equal to the number of P+ Charge = number of P+ minus number of eAtomic weight is equal to the number of protons plus the number of neutrons Atomic number si equal to the number protons Atomic number determines the identity of the atom or ion. In other words at ...
File
... because water molecules form hydrogen bonds with each other! 1. Ice floats- water is less dense as a solid than as a liquid! ...
... because water molecules form hydrogen bonds with each other! 1. Ice floats- water is less dense as a solid than as a liquid! ...
Basic Chemistry
... The first energy level holds two electrons The second energy level holds eight electrons The third energy level holds eight electrons Other levels can hold more Octet Rule: Stable atoms have eight electrons in their outer level ...
... The first energy level holds two electrons The second energy level holds eight electrons The third energy level holds eight electrons Other levels can hold more Octet Rule: Stable atoms have eight electrons in their outer level ...
Cold Fusion By Plasma Electrolysis of Water
... When the atomic nucleus of iron (Fig. 1, b) pass into the atomic nucleus of chromium (Fig. 1, a), two protons and two neutrons are released; two atoms of deuterium or one atom of helium can be formed from them. If the neutrons pass into the protons, four atoms of hydrogen are formed. It is easy to ...
... When the atomic nucleus of iron (Fig. 1, b) pass into the atomic nucleus of chromium (Fig. 1, a), two protons and two neutrons are released; two atoms of deuterium or one atom of helium can be formed from them. If the neutrons pass into the protons, four atoms of hydrogen are formed. It is easy to ...
Equivalent weight
... •Equivalent weight (also known as gram equivalent) is a term which has been used in several contexts in chemistry. •In its most general usage, it is the mass of one equivalent, that is the mass of a given substance which will: •supply or react with one mole of hydrogen ions(H+) in an acid–base react ...
... •Equivalent weight (also known as gram equivalent) is a term which has been used in several contexts in chemistry. •In its most general usage, it is the mass of one equivalent, that is the mass of a given substance which will: •supply or react with one mole of hydrogen ions(H+) in an acid–base react ...
Activities 2
... A. Protons have an elementary positive charge of 1. B. Protons have one Dalton of mass. C. Protons are always found in the nucleus of the atom. D. Any atom found in nature always has the same number of protons as electrons. 2. Which of the following statements about electron orbitals is FALSE? A. Th ...
... A. Protons have an elementary positive charge of 1. B. Protons have one Dalton of mass. C. Protons are always found in the nucleus of the atom. D. Any atom found in nature always has the same number of protons as electrons. 2. Which of the following statements about electron orbitals is FALSE? A. Th ...
0_specific - ugc-inno
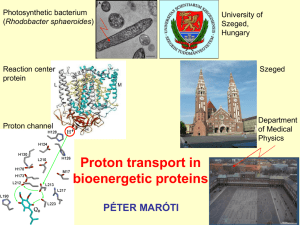
... molecules in the coordination sphere and the relative disposition of these molecules to influence the proton transfers to take place in such a way that proton prefers to get lodged at the ammonium ion region rather than the carboxylate ion region of the amino acid? In the following slide such a envi ...
... molecules in the coordination sphere and the relative disposition of these molecules to influence the proton transfers to take place in such a way that proton prefers to get lodged at the ammonium ion region rather than the carboxylate ion region of the amino acid? In the following slide such a envi ...
Proton
The proton is an elementary subatomic particle, symbol p or p+, with a positive electric charge of +1e elementary charge and mass slightly less than that of a neutron. Protons and neutrons, each with mass approximately one atomic mass unit, are collectively referred to as ""nucleons"". One or more protons are present in the nucleus of an atom. The number of protons in the nucleus is referred to as its atomic number. Since each element has a unique number of protons, each element has its own unique atomic number. The word proton is Greek for ""first"", and this name was given to the hydrogen nucleus by Ernest Rutherford in 1920. In previous years Rutherford had discovered that the hydrogen nucleus (known to be the lightest nucleus) could be extracted from the nuclei of nitrogen by collision. The proton was therefore a candidate to be a fundamental particle and a building block of nitrogen and all other heavier atomic nuclei.In the modern Standard Model of particle physics, the proton is a hadron, and like the neutron, the other nucleon (particle present in atomic nuclei), is composed of three quarks. Although the proton was originally considered a fundamental particle, it is composed of three valence quarks: two up quarks and one down quark. The rest masses of the quarks contribute only about 1% of the proton's mass, however. The remainder of the proton mass is due to the kinetic energy of the quarks and to the energy of the gluon fields that bind the quarks together. Because the proton is not a fundamental particle, it possesses a physical size; the radius of the proton is about 0.84–0.87 fm.At sufficiently low temperatures, free protons will bind to electrons. However, the character of such bound protons does not change, and they remain protons. A fast proton moving through matter will slow by interactions with electrons and nuclei, until it is captured by the electron cloud of an atom. The result is a protonated atom, which is a chemical compound of hydrogen. In vacuum, when free electrons are present, a sufficiently slow proton may pick up a single free electron, becoming a neutral hydrogen atom, which is chemically a free radical. Such ""free hydrogen atoms"" tend to react chemically with many other types of atoms at sufficiently low energies. When free hydrogen atoms react with each other, they form neutral hydrogen molecules (H2), which are the most common molecular component of molecular clouds in interstellar space. Such molecules of hydrogen on Earth may then serve (among many other uses) as a convenient source of protons for accelerators (as used in proton therapy) and other hadron particle physics experiments that require protons to accelerate, with the most powerful and noted example being the Large Hadron Collider.