o Positive charge • Electrons
... Atomic mass (u) = average mass of atoms of isotopes as the occur naturally To calculate: o Divide percentages by 100 to find natural abundances o Atomic mass = (abundance 1 x mass 1) + (abundance 2 x mass 2) ...
... Atomic mass (u) = average mass of atoms of isotopes as the occur naturally To calculate: o Divide percentages by 100 to find natural abundances o Atomic mass = (abundance 1 x mass 1) + (abundance 2 x mass 2) ...
atoms
... - Most of the mass of the atom is in the center - The positive charged particle (proton) must be in the center of the atom ...
... - Most of the mass of the atom is in the center - The positive charged particle (proton) must be in the center of the atom ...
Chemistry 150 - CSUB Home Page
... 1. What is the difference between atomic number and mass number? (2 points) The atomic number is equal to the number of protons while the mass number is the sum of the number of protons plus the number of neutrons. 2. Which of the following list the elements K, Li, Be, C, and F in order of increasin ...
... 1. What is the difference between atomic number and mass number? (2 points) The atomic number is equal to the number of protons while the mass number is the sum of the number of protons plus the number of neutrons. 2. Which of the following list the elements K, Li, Be, C, and F in order of increasin ...
Atomic Theory (Or a quick Chemistry Review)
... Atomic Theory Q: What does science study? A: The natural world, the physical universe Q: What are the components of the P.U? A: matter, energy, forces ...
... Atomic Theory Q: What does science study? A: The natural world, the physical universe Q: What are the components of the P.U? A: matter, energy, forces ...
Atomic structure and bonding I can name group 1, 7 and 0 of the
... I can state the names of the seven diatomic elements. I can label a diagram of an atom. I can state the mass, charge and position of a proton, neutron and electron within an atom. I can state the definition of an isotope. I can state the meaning of atomic number and mass number. I can use the atomic ...
... I can state the names of the seven diatomic elements. I can label a diagram of an atom. I can state the mass, charge and position of a proton, neutron and electron within an atom. I can state the definition of an isotope. I can state the meaning of atomic number and mass number. I can use the atomic ...
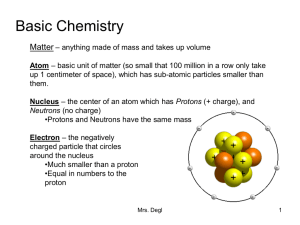
Basic Chemistry
... around the nucleus •Much smaller than a proton •Equal in numbers to the proton ...
... around the nucleus •Much smaller than a proton •Equal in numbers to the proton ...
Atomic Structure Mini Lab
... To understand how the number of protons, neutrons and electrons determine the properties of atoms Procedure: Obtain a baggie from your instructor Baggies contain the following: White bead represents a proton Purple bead represents a neutron Blue bead represents an electron Count the number of proton ...
... To understand how the number of protons, neutrons and electrons determine the properties of atoms Procedure: Obtain a baggie from your instructor Baggies contain the following: White bead represents a proton Purple bead represents a neutron Blue bead represents an electron Count the number of proton ...
Chemistry I Unit Review: The Atom Text Chapters 2 and 7 1. The
... Determine the number of valence electrons and draw the dot diagram for the following atoms: ...
... Determine the number of valence electrons and draw the dot diagram for the following atoms: ...
Atomic Theory Worksheet
... 4. Atoms are very small, what unit are they measured in? 5. What holds an atom together? 6. Label the parts on the following diagram of an atom: ...
... 4. Atoms are very small, what unit are they measured in? 5. What holds an atom together? 6. Label the parts on the following diagram of an atom: ...
Proton
The proton is an elementary subatomic particle, symbol p or p+, with a positive electric charge of +1e elementary charge and mass slightly less than that of a neutron. Protons and neutrons, each with mass approximately one atomic mass unit, are collectively referred to as ""nucleons"". One or more protons are present in the nucleus of an atom. The number of protons in the nucleus is referred to as its atomic number. Since each element has a unique number of protons, each element has its own unique atomic number. The word proton is Greek for ""first"", and this name was given to the hydrogen nucleus by Ernest Rutherford in 1920. In previous years Rutherford had discovered that the hydrogen nucleus (known to be the lightest nucleus) could be extracted from the nuclei of nitrogen by collision. The proton was therefore a candidate to be a fundamental particle and a building block of nitrogen and all other heavier atomic nuclei.In the modern Standard Model of particle physics, the proton is a hadron, and like the neutron, the other nucleon (particle present in atomic nuclei), is composed of three quarks. Although the proton was originally considered a fundamental particle, it is composed of three valence quarks: two up quarks and one down quark. The rest masses of the quarks contribute only about 1% of the proton's mass, however. The remainder of the proton mass is due to the kinetic energy of the quarks and to the energy of the gluon fields that bind the quarks together. Because the proton is not a fundamental particle, it possesses a physical size; the radius of the proton is about 0.84–0.87 fm.At sufficiently low temperatures, free protons will bind to electrons. However, the character of such bound protons does not change, and they remain protons. A fast proton moving through matter will slow by interactions with electrons and nuclei, until it is captured by the electron cloud of an atom. The result is a protonated atom, which is a chemical compound of hydrogen. In vacuum, when free electrons are present, a sufficiently slow proton may pick up a single free electron, becoming a neutral hydrogen atom, which is chemically a free radical. Such ""free hydrogen atoms"" tend to react chemically with many other types of atoms at sufficiently low energies. When free hydrogen atoms react with each other, they form neutral hydrogen molecules (H2), which are the most common molecular component of molecular clouds in interstellar space. Such molecules of hydrogen on Earth may then serve (among many other uses) as a convenient source of protons for accelerators (as used in proton therapy) and other hadron particle physics experiments that require protons to accelerate, with the most powerful and noted example being the Large Hadron Collider.