Tunneling through a Barrier
... the walls, where it decays exponentially towards zero, and oscillates within the well. The wavefunctions and their slopes are continuous at the edges of the ...
... the walls, where it decays exponentially towards zero, and oscillates within the well. The wavefunctions and their slopes are continuous at the edges of the ...
17-Sept
... •Mass number (A) = total number of nucleons in the nucleus (i.e., protons and neutrons). •One nucleon has a mass of 1 amu (Atomic Atomic Mass Unit) Dalton” Unit a.k.a “Dalton ...
... •Mass number (A) = total number of nucleons in the nucleus (i.e., protons and neutrons). •One nucleon has a mass of 1 amu (Atomic Atomic Mass Unit) Dalton” Unit a.k.a “Dalton ...
Crystal structure and spectroscopic properties of [Zn(2-qmpe)Cl ] containing diethyl (quinolin-2-ylmethyl)phosphonate ligand (2-qmpe)
... ppm with respect to TMS (1%) as an internal standard and H3PO4 (85%) as the external standard. Determination of the crystal structure of [Zn(2-qmpe)Cl2]: X-Ray data were collected on a Kuma KM4CCD diffractometer (MoKα radiation, λ = 0.71073 Å). X-Ray data were collected at 100 K using an Oxford Cryo ...
... ppm with respect to TMS (1%) as an internal standard and H3PO4 (85%) as the external standard. Determination of the crystal structure of [Zn(2-qmpe)Cl2]: X-Ray data were collected on a Kuma KM4CCD diffractometer (MoKα radiation, λ = 0.71073 Å). X-Ray data were collected at 100 K using an Oxford Cryo ...
Chapter 3 Atomic mass
... At present carbon-12 is used as the standard of comparison because It is solid and can be easily handled, Its mass can be more easily measured with a mass spectrometer. Carbon-12 is easily found as its compounds ...
... At present carbon-12 is used as the standard of comparison because It is solid and can be easily handled, Its mass can be more easily measured with a mass spectrometer. Carbon-12 is easily found as its compounds ...
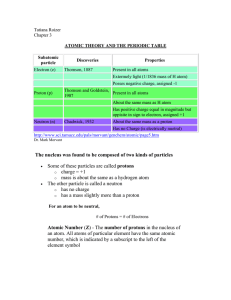
atomic theory and the periodic table
... the electron is further from the nucleus - this is an orbital at the second energy level. If you look carefully, you will notice that there is another region of slightly higher electron density (where the dots are thicker) nearer the nucleus. ("Electron density" is another way of talking about how l ...
... the electron is further from the nucleus - this is an orbital at the second energy level. If you look carefully, you will notice that there is another region of slightly higher electron density (where the dots are thicker) nearer the nucleus. ("Electron density" is another way of talking about how l ...
A STUDY ON STRUCTURAL ASPECTS AND MICROBIAL ACTIVITY OF (E)-4- PYRIDINECARBOXALDEHYDE-3-HYDROXY-5-(HYDROXYMETHYL)-2-METHYL-OXIME
... and Pseudomonas aeruginosa (Gram negative) bacteria. The comparison of biological activity with different strains of bacteria revealed that the compound under investigation is highly active against both gram positive and negative bacteria. The results are tabulated in table-3. The studies revealed t ...
... and Pseudomonas aeruginosa (Gram negative) bacteria. The comparison of biological activity with different strains of bacteria revealed that the compound under investigation is highly active against both gram positive and negative bacteria. The results are tabulated in table-3. The studies revealed t ...
Analyzing ITC Data for the Enthalpy of Binding Metal Ions to Ligands
... All ITC measurements were performed in the TA Instruments® standard volume nanoITC with a gold cell and carried out at 25ºC while stirring at 350 rpm. The 100 mM buffer solutions of Tris, HEPES, and imidazole were adjusted to have identical ionic strength and pH (µ = 0.1 M and pH =7.4). A background ...
... All ITC measurements were performed in the TA Instruments® standard volume nanoITC with a gold cell and carried out at 25ºC while stirring at 350 rpm. The 100 mM buffer solutions of Tris, HEPES, and imidazole were adjusted to have identical ionic strength and pH (µ = 0.1 M and pH =7.4). A background ...
atoms and molecules
... Answer: In all chemical reactions, there is only exchange of atoms of reactants taking place when products are formed. Since there is no loss or gain of mass ,the chemical reactions are balanced according to law of conservation of mass. Question What is basic difference between atoms and molecules? ...
... Answer: In all chemical reactions, there is only exchange of atoms of reactants taking place when products are formed. Since there is no loss or gain of mass ,the chemical reactions are balanced according to law of conservation of mass. Question What is basic difference between atoms and molecules? ...
Polarizability
... Dispersion Influence The strength of a dispersion force depends on the ease with which the charge distribution in a molecule can be distorted. ...
... Dispersion Influence The strength of a dispersion force depends on the ease with which the charge distribution in a molecule can be distorted. ...
Balancing Chemical Equations
... Similarly, we can state that 6 moles of H2O are produced for every two moles of C2H6SO consumed. Thus, the stoichiometric ratio is 3 for H2O relative to C2H6SO. 2 C2H6SO + 9 O2 = 6 H2O + 4 CO2 + 2 SO3 ...
... Similarly, we can state that 6 moles of H2O are produced for every two moles of C2H6SO consumed. Thus, the stoichiometric ratio is 3 for H2O relative to C2H6SO. 2 C2H6SO + 9 O2 = 6 H2O + 4 CO2 + 2 SO3 ...
Word - chemmybear.com
... 17. 1. Bases are _______________. 2. Bases cause indicators to turn a characteristic ...
... 17. 1. Bases are _______________. 2. Bases cause indicators to turn a characteristic ...
CHEM 210 Ch06
... • Any ion that has a net positive charge at that pH will migrate toward the negatively charged cathode. • Any ion having a net negative charge will migrate toward the positively charged anode. • If the net charge is zero, the species will not move. • An amino acid’s isoelectric pH, or isoelectric po ...
... • Any ion that has a net positive charge at that pH will migrate toward the negatively charged cathode. • Any ion having a net negative charge will migrate toward the positively charged anode. • If the net charge is zero, the species will not move. • An amino acid’s isoelectric pH, or isoelectric po ...
Atomic mass unit - Liceo Mascheroni
... The unit is convenient because one hydrogen atom has a mass of approximately 1 u, and more generally an atom or molecule that contains n protons and neutrons will have a mass approximately equal to n u. (The reason is that a 12C atom contains 6 protons, 6 neutrons and 6 electrons, with the protons a ...
... The unit is convenient because one hydrogen atom has a mass of approximately 1 u, and more generally an atom or molecule that contains n protons and neutrons will have a mass approximately equal to n u. (The reason is that a 12C atom contains 6 protons, 6 neutrons and 6 electrons, with the protons a ...
Structure of atoms
... ‘Strong’ and weak’ when applied to acids and bases have a different meaning from ‘concentrated’ and ‘dilute’. In a given volume of water: • a concentrated acid or base solution has many molecules of acid or base present and/or dissolved • a dilute solution has fewer acid or base molecules present ...
... ‘Strong’ and weak’ when applied to acids and bases have a different meaning from ‘concentrated’ and ‘dilute’. In a given volume of water: • a concentrated acid or base solution has many molecules of acid or base present and/or dissolved • a dilute solution has fewer acid or base molecules present ...
a level chemistry - some definitions to learn
... The number of protons in the nucleus of an atom The sum of the protons and neutrons in the nucleus of an atom The mass of an atom relative to that of the carbon 12 isotope having a value of 12.000 The simplest, whole number, ratio of elements in a compound The exact number of atoms of each element i ...
... The number of protons in the nucleus of an atom The sum of the protons and neutrons in the nucleus of an atom The mass of an atom relative to that of the carbon 12 isotope having a value of 12.000 The simplest, whole number, ratio of elements in a compound The exact number of atoms of each element i ...
C2.3 Atomic Structure, Analysis and Quantitative Chemistry
... for higher tier with calculations on empirical formula as well as reacting mass calculations and percentage yield All candidates need to know that there are reasons why not all reactants are converted to products as they could be lost in transfer, bi- products can be produced or that the reaction is ...
... for higher tier with calculations on empirical formula as well as reacting mass calculations and percentage yield All candidates need to know that there are reasons why not all reactants are converted to products as they could be lost in transfer, bi- products can be produced or that the reaction is ...
Midterm Study Guide with Answers
... of a sugar sample you knew to have a mass of 1 g. How would you know whether or not the measurements were accurate? How would you know whether or not they were precise? Could the three measurements be precise, but not accurate? Explain. ...
... of a sugar sample you knew to have a mass of 1 g. How would you know whether or not the measurements were accurate? How would you know whether or not they were precise? Could the three measurements be precise, but not accurate? Explain. ...
ELEMENTS AND ATOMIC MASSES INTRODUCTION An element is
... ELEMENTS AND ATOMIC MASSES INTRODUCTION An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Elements are the building blocks of all matter. Currently 106 different elements are known. Of these only 88 are found naturally. Some of the other elem ...
... ELEMENTS AND ATOMIC MASSES INTRODUCTION An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Elements are the building blocks of all matter. Currently 106 different elements are known. Of these only 88 are found naturally. Some of the other elem ...
Moseley`s law refuted
... A is the number of hydrogen building blocks of an element and its isotopes. X-ray radiation of elements can be explained due to the specific atomic architecture of hydrogen and helium building blocks. According to Moseley’s law, isotopes of elements (for example Ag-107 and Ag-109) would cause equal ...
... A is the number of hydrogen building blocks of an element and its isotopes. X-ray radiation of elements can be explained due to the specific atomic architecture of hydrogen and helium building blocks. According to Moseley’s law, isotopes of elements (for example Ag-107 and Ag-109) would cause equal ...
June 2011 review
... Explain, in terms of electronegativity difference, why the bond between hydrogen and oxygen in a water molecule is more polar than the bond between hydrogen and nitrogen in an ammonia molecule. [1] 9. Base your answer on the information below. In 1864, the Solvay process was developed to make soda ...
... Explain, in terms of electronegativity difference, why the bond between hydrogen and oxygen in a water molecule is more polar than the bond between hydrogen and nitrogen in an ammonia molecule. [1] 9. Base your answer on the information below. In 1864, the Solvay process was developed to make soda ...
First-Sample_Exam-1-Key
... 2. Consider an atom of deuterium (2H = D). Assuming a nuclear radius of 10-4 Å, estimate the density of the deuterium nucleus. a. 8 x 1014 kg/m3 b. 5 x 10-14 kg/m3 e. none of the above ...
... 2. Consider an atom of deuterium (2H = D). Assuming a nuclear radius of 10-4 Å, estimate the density of the deuterium nucleus. a. 8 x 1014 kg/m3 b. 5 x 10-14 kg/m3 e. none of the above ...
Proton
The proton is an elementary subatomic particle, symbol p or p+, with a positive electric charge of +1e elementary charge and mass slightly less than that of a neutron. Protons and neutrons, each with mass approximately one atomic mass unit, are collectively referred to as ""nucleons"". One or more protons are present in the nucleus of an atom. The number of protons in the nucleus is referred to as its atomic number. Since each element has a unique number of protons, each element has its own unique atomic number. The word proton is Greek for ""first"", and this name was given to the hydrogen nucleus by Ernest Rutherford in 1920. In previous years Rutherford had discovered that the hydrogen nucleus (known to be the lightest nucleus) could be extracted from the nuclei of nitrogen by collision. The proton was therefore a candidate to be a fundamental particle and a building block of nitrogen and all other heavier atomic nuclei.In the modern Standard Model of particle physics, the proton is a hadron, and like the neutron, the other nucleon (particle present in atomic nuclei), is composed of three quarks. Although the proton was originally considered a fundamental particle, it is composed of three valence quarks: two up quarks and one down quark. The rest masses of the quarks contribute only about 1% of the proton's mass, however. The remainder of the proton mass is due to the kinetic energy of the quarks and to the energy of the gluon fields that bind the quarks together. Because the proton is not a fundamental particle, it possesses a physical size; the radius of the proton is about 0.84–0.87 fm.At sufficiently low temperatures, free protons will bind to electrons. However, the character of such bound protons does not change, and they remain protons. A fast proton moving through matter will slow by interactions with electrons and nuclei, until it is captured by the electron cloud of an atom. The result is a protonated atom, which is a chemical compound of hydrogen. In vacuum, when free electrons are present, a sufficiently slow proton may pick up a single free electron, becoming a neutral hydrogen atom, which is chemically a free radical. Such ""free hydrogen atoms"" tend to react chemically with many other types of atoms at sufficiently low energies. When free hydrogen atoms react with each other, they form neutral hydrogen molecules (H2), which are the most common molecular component of molecular clouds in interstellar space. Such molecules of hydrogen on Earth may then serve (among many other uses) as a convenient source of protons for accelerators (as used in proton therapy) and other hadron particle physics experiments that require protons to accelerate, with the most powerful and noted example being the Large Hadron Collider.

![Crystal structure and spectroscopic properties of [Zn(2-qmpe)Cl ] containing diethyl (quinolin-2-ylmethyl)phosphonate ligand (2-qmpe)](http://s1.studyres.com/store/data/008844838_1-bf4cef9539c1ba0a885c25bcd900772d-300x300.png)