AP Chemistry Predicting Products Tutorial
... 1. A solution of sulfuric acid is added to a solution of barium hydroxide until the same number of moles of each compound has been added. 4H+ + SO42- + Ba2+ + 2OH- BaSO4 + 2H2O 2. A solution of sodium hydroxide is added to a solution of sodium dihydrogen phosphate until the same number of moles of ...
... 1. A solution of sulfuric acid is added to a solution of barium hydroxide until the same number of moles of each compound has been added. 4H+ + SO42- + Ba2+ + 2OH- BaSO4 + 2H2O 2. A solution of sodium hydroxide is added to a solution of sodium dihydrogen phosphate until the same number of moles of ...
The term “Chromic Acid” actually refers to a collection of compounds
... contain the chromate ion, CrO42-, and have an intense yellow color. Dichromate salts contain the dichromate ion, Cr2O72−, and have an intense orange color. Potassium dichromate, K2Cr2O7, is a common inorganic chemical reagent, most commonly used as an oxidizing agent in various laboratory and indust ...
... contain the chromate ion, CrO42-, and have an intense yellow color. Dichromate salts contain the dichromate ion, Cr2O72−, and have an intense orange color. Potassium dichromate, K2Cr2O7, is a common inorganic chemical reagent, most commonly used as an oxidizing agent in various laboratory and indust ...
Word Pro
... give a precipitate of zinc hydroxide. (b) Copper(II) oxide dissolves in dilute hydrochloric acid. (c) Potassium chromate reacts with lead(II) nitrate solution to give a yellow precipitate. (d) Magnesium ribbon reacts with dilute hydrochloric acid. (e) Solid calcium carbonate reacts with dilute nitri ...
... give a precipitate of zinc hydroxide. (b) Copper(II) oxide dissolves in dilute hydrochloric acid. (c) Potassium chromate reacts with lead(II) nitrate solution to give a yellow precipitate. (d) Magnesium ribbon reacts with dilute hydrochloric acid. (e) Solid calcium carbonate reacts with dilute nitri ...
The general reaction for the esterification of an organic acid with an
... The general reaction for the esterification of an organic acid with an alcohol is R-COOH + HO-R’ R-CO-OR’ + H2O Esterification reactions are a kind of elimination or condensation reaction. In this reaction, R and R’ represent hydrocarbon chains, which may be the same or different. Unlike many orga ...
... The general reaction for the esterification of an organic acid with an alcohol is R-COOH + HO-R’ R-CO-OR’ + H2O Esterification reactions are a kind of elimination or condensation reaction. In this reaction, R and R’ represent hydrocarbon chains, which may be the same or different. Unlike many orga ...
PRACTICE TEST for EXAM 10
... the pH of a solution if the concentration of hydronium ion increases? What if it decreases? 7. Compare and contrast a strong acid and a weak acid, in terms of neutralization titration results, pH, and conductivity, and explain their differences in terms of their reaction with water. 8. A weak acid i ...
... the pH of a solution if the concentration of hydronium ion increases? What if it decreases? 7. Compare and contrast a strong acid and a weak acid, in terms of neutralization titration results, pH, and conductivity, and explain their differences in terms of their reaction with water. 8. A weak acid i ...
cr-0009 - sulfuric acid .12n
... Avoid contact with skin, eyes and clothing. Launder contaminated clothing prior to reuse. Storage requirements: Keep out of direct sunlight. Store in a cool and dry place. Special shipping information: See transportation information. Section 8: FIRST AID MEASURES Skin contact: Remove contaminated cl ...
... Avoid contact with skin, eyes and clothing. Launder contaminated clothing prior to reuse. Storage requirements: Keep out of direct sunlight. Store in a cool and dry place. Special shipping information: See transportation information. Section 8: FIRST AID MEASURES Skin contact: Remove contaminated cl ...
safe disposal of waste containing nitric acid
... a reminder of the hazards of working with nitric acid. Nitric acid is an extremely corrosive acid. It is also a strong oxidizer, which reacts violently with many materials including organic compounds (alcohols, acetone, acetic acid), reducing agents (metal hydrides, formic acid, phosphorus acid) and ...
... a reminder of the hazards of working with nitric acid. Nitric acid is an extremely corrosive acid. It is also a strong oxidizer, which reacts violently with many materials including organic compounds (alcohols, acetone, acetic acid), reducing agents (metal hydrides, formic acid, phosphorus acid) and ...
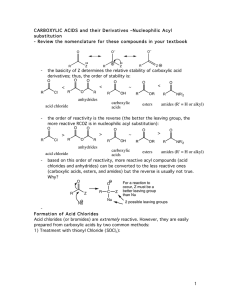
carboxylic acids esters amides (R
... dehydrating conditions. For the most part, this is a difficult reaction to perform, and since the reactivity of anhydrides is so similar to that of acid chlorides, anhydrides are not commonly used in synthesis. ...
... dehydrating conditions. For the most part, this is a difficult reaction to perform, and since the reactivity of anhydrides is so similar to that of acid chlorides, anhydrides are not commonly used in synthesis. ...
Learning Check
... Aspirin stored for a long time may undergo hydrolysis in the presence of water and heat. What will be the products of this reaction? To write the hydrolysis products, separate the compound at the ester bond. Complete the formula of the carboxylic acid by adding –OH (from water) to the carbonyl group ...
... Aspirin stored for a long time may undergo hydrolysis in the presence of water and heat. What will be the products of this reaction? To write the hydrolysis products, separate the compound at the ester bond. Complete the formula of the carboxylic acid by adding –OH (from water) to the carbonyl group ...
Buffers and Acid/Base
... We do not use QCK tables for SA/SB calculations b/c they completely dissociate, no equilibrium so… [ACID] = [H+] Hydrolysis of Salts **Conjugates of weak acids/bases react with water to form acidic/basic solutions** ...
... We do not use QCK tables for SA/SB calculations b/c they completely dissociate, no equilibrium so… [ACID] = [H+] Hydrolysis of Salts **Conjugates of weak acids/bases react with water to form acidic/basic solutions** ...
Unit Seven Worksheet – 2
... Ratio of the concentration of the dissociated (or ionized) form of an acid to the concentration of the undissociated acid; symbolized Ka Base that dissociates completely into metal ions and hydroxide ions in aqueous solution Acid that completely ionizes in aqueous solution Base that does not dissoci ...
... Ratio of the concentration of the dissociated (or ionized) form of an acid to the concentration of the undissociated acid; symbolized Ka Base that dissociates completely into metal ions and hydroxide ions in aqueous solution Acid that completely ionizes in aqueous solution Base that does not dissoci ...
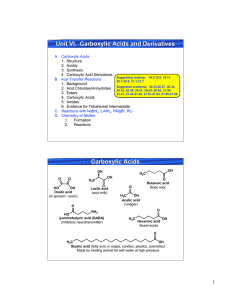
Un it VI. Carboxylic Acids and Derivatives U
... OH Stearic acid (fatty acid, in soaps, candles, plastics, cosmetics) Made by treating animal fat with water at high pressure ...
... OH Stearic acid (fatty acid, in soaps, candles, plastics, cosmetics) Made by treating animal fat with water at high pressure ...
ESTERIFICATION Ester Formation Important Vocabulary (review) O
... Methanol is particularly dangerous as it has such weak intermolecular forces, between its molecules, that it easily and rapidly evaporates, mixing with the air. This makes for a hazardous fire ready to happen. Great care should be used when handling these materials. Goggles, gloves, and aprons/lab c ...
... Methanol is particularly dangerous as it has such weak intermolecular forces, between its molecules, that it easily and rapidly evaporates, mixing with the air. This makes for a hazardous fire ready to happen. Great care should be used when handling these materials. Goggles, gloves, and aprons/lab c ...
Acids and Bases
... Most solutions of both acids and bases are clear and colourless. We need an indicator to tell them apart. An indicator is a chemical which changes colour as the concentration of H+ (aq) and OH- (aq) changes. Two common indicators are litmus and phenolpthalein ...
... Most solutions of both acids and bases are clear and colourless. We need an indicator to tell them apart. An indicator is a chemical which changes colour as the concentration of H+ (aq) and OH- (aq) changes. Two common indicators are litmus and phenolpthalein ...
Problem Set 12-2: Organic Chemistry
... Alcohol, carboxylic acids, amides, amines, aldehydes and ketones, esters. Solubility decreases with the length of the carbon chain. 9. Give 3 factors that determine solubility in water. Polarity of molecule Ability to form hydrogen bonds Length of non-polar carbon chains 10. Rank in terms of boiling ...
... Alcohol, carboxylic acids, amides, amines, aldehydes and ketones, esters. Solubility decreases with the length of the carbon chain. 9. Give 3 factors that determine solubility in water. Polarity of molecule Ability to form hydrogen bonds Length of non-polar carbon chains 10. Rank in terms of boiling ...
Going Bananas Over Isoamyl Acetate
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
Going Bananas Over Isoamyl Acetate
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
... acid, as noted above. If either one is doubled, the theoretical yield increases to 85%. When one is tripled, it goes to 90%. But note that in the example cited the boiling point of the relatively nonpolar ester is only about 8° C higher than the boiling points of the polar acetic acid and 1-butanol, ...
Benzocaine Synthesis via Esterification
... as they are formed. This can be accomplished by physical (e.g., azeotropic distillation of water) or chemical means. The position of equilibrium is determined by the equilibrium constant (K), as defined by the following mass-action expression: ...
... as they are formed. This can be accomplished by physical (e.g., azeotropic distillation of water) or chemical means. The position of equilibrium is determined by the equilibrium constant (K), as defined by the following mass-action expression: ...
Carboxylic Acids General formula R C O OH C O OH carboxyl group
... β-methylbutyric acid isovaleric acid very unpleasant smell like that of dirty socks ...
... β-methylbutyric acid isovaleric acid very unpleasant smell like that of dirty socks ...
Safety Data Sheet - Fisher Scientific
... appropriate fire suppression agents for adjacent combustible materials or sources of ignition. Use dry Chemical, foam, or carbon dioxide to extinguish fire. For safety reasons unsuitable extinguishing agents: Do not use water directly on sulfuric acid Special hazards arising from the substance or mi ...
... appropriate fire suppression agents for adjacent combustible materials or sources of ignition. Use dry Chemical, foam, or carbon dioxide to extinguish fire. For safety reasons unsuitable extinguishing agents: Do not use water directly on sulfuric acid Special hazards arising from the substance or mi ...
Test 4 Review - Ralph C. Mahar
... Check for balanced charges on both sides Combine half reactions and cancel common items Add spectator ions and balance ...
... Check for balanced charges on both sides Combine half reactions and cancel common items Add spectator ions and balance ...
Experiment 7
... (acting as keys) bind to specific receptors (acting as locks) to excite a neuron and trigger the sensation of particular smells. ...
... (acting as keys) bind to specific receptors (acting as locks) to excite a neuron and trigger the sensation of particular smells. ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.