* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Test 4 Review - Ralph C. Mahar
Survey
Document related concepts
Rate equation wikipedia , lookup
Detailed balance wikipedia , lookup
Ionic compound wikipedia , lookup
History of electrochemistry wikipedia , lookup
Transition state theory wikipedia , lookup
Sulfuric acid wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
Transcript
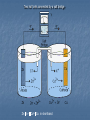
Test 4 Review Advanced Chemistry Equilibrium mA + nB g sP + rQ Keq = [P]s[Q]r [A]m[B]n Ksp = [A+][B-] for dissolving of a solid AB g A+ + BKa = [H+][A-] for the dissociation of an acid [HA] Ka is small for a weak acid, large for a strong acid. Never include solids or pure liquids in a Keq. Solubility Product Constant AB(s) D A+(aq) + B-(aq) Ksp = [A+][B-] At 25°C the solubility product constant for strontium sulfate, SrSO4, is 7.6 x 10-7. What is the concentration of Sr2+ at 25°C? [Sr2+][SO42-] = 7.6 x 10-7 [Sr2+]=[SO42-] [Sr2+] = 8.7 x 10-4M The solubility product constant for strontium fluoride is 7.9 x 10-10. What is the molar solubility of SrF2 at 25°C? SrF2 D Sr+2 + 2F[Sr2+][F-]2=7.9 x 10-10 [Sr2+]= x [F-]= 2x (x)(2x)2=7.9 x 10-10 x = 5.8 x 10-4M Strong bases: hydroxides of groups 1 & 2 (except Be) Strong acids: HCl, HBr, HI HClO4, H2SO4, HNO3 Ionization constant, Ka, for a weak acid HA D H+ + AKa = [H+][A-] [HA] [CH 3 NH 3 ][OH-] Kb = [CH 3 NH 2 ] Ka ∙Kb= Kw = 1.00 x 10-14 What is the [H+] in 0.100M formic acid? Ka for formic acid is 1.77 x 10-4 HCOOH D H+ + COOHSince this is a weak acid, [HCOOH] ͌ 0.100M Ka = [H+][COOH-] = 1.77 x 10-4 [HCOOH] Let x = [H+] = [COOH-] x2 = 1.77 x 10-4 0.100 X = 4.21 x 10-3M Percent ionization [amount ionized] [original acid] What is the percent ionization of [H+] from the previous problem? [H+] = 4.21 x 10-3M, [HCOOH] = 0.100M 4.21 x 10-3 = 4.21% 0.100 hydrolysis The reaction of a salt with water to form an acidic or basic solution. Example: FeCl3 D Fe3+(aq) + Cl-(aq) Fe3+ + 3OH- D Fe(OH)3 H2O D H+ + OH shifts right, creating more H+ cation anion solution Strong base Strong base Weak base Weak base Strong acid Weak acid Strong acid Weak acid Neutral Basic Acidic Neutral [H+] ∙ [OH-] = 10-14 pH + pOH = 14.0 pH = -log[H3O+] [H3O+] = antilog(-pH) Find the pH of a solution with [H3O+] of 9.85 x 10-8M. pH = -log (9.85 x 10-8) = 7.01 What is the [H3O+] in a solution with pH 7.01? [H3O+] = antilog(-7.01)= 9.85 x 10-8M What is the pH of the following solutions? [H ] = 10 , pH = 1 0.1M HCl [OH ]=10 , pOH=1, pH=13 0.1M NaOH K = 4.4 x 10 = x 0.1M H2CO3 0.1 + -1 - a X = 2.1x10-4 -1 -7 2 pH = 3.68 Titration Standard solution- one whose concentration is known Endpoint- the point at which equivalent amounts of reactants are present. M∙V = moles MaVa=MbVb Titration curves Calculating pH of a buffer What is the pH of a buffer that is 0.12M lactic acid (HC3H5O3) and 0.10M sodium lactate? Ka = 1.4 x 10-4 HC3H5O3 Initial Change equilibrium 0.12M -x .12-x H+ C3H5O3- 0 0.10M +x +x x .10+x Henderson-Hasselbalch pH = pKa + log [base] [acid] = -log(1.4x10-4) + log .10 .12 = 3.85 + (-.079) = 3.77 Conjugate base of the acid Common Ion (Buffers) Calculate the pH of a 0.100M solution of formic acid and 0.020M sodium formate. Ka= 1.77 x 10-4 HCOOH g H+ + COOH0.100 x 0.020 Ka = [H+][COOH-] = .020x X = 8.85 x 10 M pH = 3.05 [HCOOH] 0.100 -4 Redox reactions involve a change in oxidation number Oxidation Loss of electrons Reduction Gain electrons 3Cu2+ + 2Fe g 3Cu + 2Fe3+ copper gains 2 electrons (reduced) (oxidized) iron loses 3 electrons Reducing agent- is oxidized (iron) Oxidizing agent- is reduced (copper) Steps for balancing redox reactions using the half-reaction method Write ionic equation for half reactions Balance chemically 1. 2. a) b) c) 3. 4. 5. 6. Balance non- O and H atoms Add H2O to balance O’s Add H+ to balance H’s (in a basic solution add OH-) Balance electrically- add e-’s to the more + side Check for balanced charges on both sides Combine half reactions and cancel common items Add spectator ions and balance Activity Series More active Less active lithium potassium magnesium aluminum zinc iron nickel lead HYDROGREN copper silver platinum gold Oxidizes easily The metal must be above (more active than) the ion for it to be a spontaneous reaction. Reduces easily Voltaic Cell Anode attracts anions where oxidation occurs Cathode attracts cations where reduction occurs Salt bridge connects the two half cells contains a strong electrolyte Two half cells connected by a salt bridge Zn Zn+ Cu2+ Cu in shorthand Reduction half reactions F2 is the strongest oxidizing agent Li is the strongest reducing agent Reduction Potentials (E) If E is positive, the reaction is spontaneous. If E is negative, the reverse reaction is spontaneous. Eo is the standard electrode potential all ions are 1M and gases are 1 atm The net Eo is the sum of the Eo of the half reactions The stronger oxidizing agent reduces. Reverse the sign of the substance oxidized. What is the voltage produced from the reaction of Zn metal with Cu2+ ions? Zn(s) + Cu2+(aq) g Zn2+(aq) + Cu(s) Zn2+ + 2e- g Zn -0.7628 Cu2+ + 2e- g Cu 0.3402 -(-0.7628) + 0.3402 = 1.103 volts Will happen spontaneously Zn2+ + Ni(s) g Zn(s) + Ni2+ -(-0.23) + (-0.7628) = -.53 Will not occur spontaneously Faraday’s Law Coulombs = amperes x seconds 1 C = 1amp·1sec 96,485 coulombs = 1 mole e- What mass of copper will be deposited by a current of 7.89 amps flowing for 1200 seconds? Cu2+ + 2e- g Cu at the cathode 7.89A x 1200s x 1C x 1 mol e- = .0981 mol eA·s 96,485C .0981 mol e- x 1 mol Cu x 63.5g Cu = 3.1g Cu 2 mol e- 1 mole Cu Nernst Equation Eo Voltage under standard conditions (1M solutions at 25°C and 101.3kPa) At non-standard conditions, use Nernst equation E = E° - 0.05916 log [products] n [reactants] n = no. of electrons transferred Coefficients in front of reactants or products are used as powers of their concentrations.