Synthesis of 1
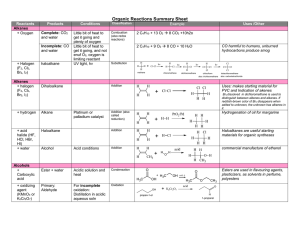
... Alcohols are easily converted into the corresponding alkyl halides by reacting them with concentrated aqueous hydrogen halides. Tertiary alcohols react almost instantly at cool temperatures via an SN1 mechanism since 3o carbocations are relatively easy to form. Primary alcohols, on the other hand, r ...
... Alcohols are easily converted into the corresponding alkyl halides by reacting them with concentrated aqueous hydrogen halides. Tertiary alcohols react almost instantly at cool temperatures via an SN1 mechanism since 3o carbocations are relatively easy to form. Primary alcohols, on the other hand, r ...
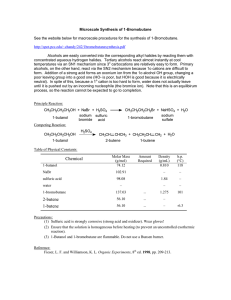
Electrolysis, the Faraday, and Avogadro`s Number
... 1. Determine the exact moles of H2 produced (PV = nRT) under your conditions. 2. Using the elapsed time, determine the amount of charge q in Coulombs that were transferred. 3. Determine the Faraday number F and calculate a percent error. 4. Determine a value for Avogadro’s number using your Faraday ...
... 1. Determine the exact moles of H2 produced (PV = nRT) under your conditions. 2. Using the elapsed time, determine the amount of charge q in Coulombs that were transferred. 3. Determine the Faraday number F and calculate a percent error. 4. Determine a value for Avogadro’s number using your Faraday ...
CHE 145-381 – TEST #2 SPRING 2009 CHAPTERS 6, 7, 8 NAME
... N2O5 __________________________________ NO ____________________________________ CBr4 ___________________________________ CO ________________________________________ N2O4 _______________________________________ SO3 ________________________________________ IF7 _____________________________________ NH3 ...
... N2O5 __________________________________ NO ____________________________________ CBr4 ___________________________________ CO ________________________________________ N2O4 _______________________________________ SO3 ________________________________________ IF7 _____________________________________ NH3 ...
Diet and Exercise – Healthy Diet
... the chemical industry. The flavours and fragrances of different esters are widely used to produce food flavourings and perfumes. ...
... the chemical industry. The flavours and fragrances of different esters are widely used to produce food flavourings and perfumes. ...
aldehydesketonescarb..
... properties of these compounds? • They are weak acids that will ionize slightly in water. • The H on the –OH is the acidic hydrogen and leaves when the acid is ionized. ...
... properties of these compounds? • They are weak acids that will ionize slightly in water. • The H on the –OH is the acidic hydrogen and leaves when the acid is ionized. ...
Pre-AP Chemistry
... only cheap perfumes use esters, while more expensive ones use natural essential oils that are extracted by alcohol. Esters are not stable to perspiration and usually break down producing the carboxylic acid, generally with an unpleasant odor. Esters are named by first giving the alcohol name, follow ...
... only cheap perfumes use esters, while more expensive ones use natural essential oils that are extracted by alcohol. Esters are not stable to perspiration and usually break down producing the carboxylic acid, generally with an unpleasant odor. Esters are named by first giving the alcohol name, follow ...
Alcohols and ethers
... • Molecules have attractive forces between the molecules called hydrogen bonds, not as strong as covalent bonds. • Higher boiling point than corresponding alkanes. • Hydrogen bonds form between alcohol and water molecules therefore they are miscible / soluble. • Long chain alcohols are less soluble. ...
... • Molecules have attractive forces between the molecules called hydrogen bonds, not as strong as covalent bonds. • Higher boiling point than corresponding alkanes. • Hydrogen bonds form between alcohol and water molecules therefore they are miscible / soluble. • Long chain alcohols are less soluble. ...
877-Alcohols Carboxylic acids and Esters Presentation
... Q Why not write C2H6O? The formula gives us clues about the structure of the molecule The oxygen and hydrogen in the alcohol functional group are bonded and so are written together ...
... Q Why not write C2H6O? The formula gives us clues about the structure of the molecule The oxygen and hydrogen in the alcohol functional group are bonded and so are written together ...
Storage Pattern for Chemicals Where Space is Limited
... Do not store chemical containers above eye level if possible. Separate inorganic compounds from organic compounds. Store solids above and liquids below. Storage cabinets for acids, bases and flammables are meant for liquids, not dry solids. Vent acid cabinets to prevent vapor build-up. Store concent ...
... Do not store chemical containers above eye level if possible. Separate inorganic compounds from organic compounds. Store solids above and liquids below. Storage cabinets for acids, bases and flammables are meant for liquids, not dry solids. Vent acid cabinets to prevent vapor build-up. Store concent ...
Derivatives of Carboxylic Acids
... salicylate ester of acetic acid. • Salicylic acid is a bifunctional molecule (acid and phenol) which is itself a disinfectant. • Aspirin comes from the willow bark which was used in the Middle Ages by Jesuit missionaries. ...
... salicylate ester of acetic acid. • Salicylic acid is a bifunctional molecule (acid and phenol) which is itself a disinfectant. • Aspirin comes from the willow bark which was used in the Middle Ages by Jesuit missionaries. ...
Alcohols revisited
... • Molecules have attractive forces between the molecules called hydrogen bonds, not as strong as covalent bonds. • Higher boiling point than corresponding alkanes. • Hydrogen bonds form between alcohol and water molecules therefore they are miscible / soluble. • Long chain alcohols are less soluble. ...
... • Molecules have attractive forces between the molecules called hydrogen bonds, not as strong as covalent bonds. • Higher boiling point than corresponding alkanes. • Hydrogen bonds form between alcohol and water molecules therefore they are miscible / soluble. • Long chain alcohols are less soluble. ...
Chapter 20 - people.vcu.edu
... These chains form micelles. Water-soluble with lipophilic interiors. ...
... These chains form micelles. Water-soluble with lipophilic interiors. ...
Homework Set #1
... and ribose form esters with phosphoric acid in DNA and RNA. Can both monosaccharides react to form the same number of ester combinations? Show the reactive sites for esterfication. CH2OH O ...
... and ribose form esters with phosphoric acid in DNA and RNA. Can both monosaccharides react to form the same number of ester combinations? Show the reactive sites for esterfication. CH2OH O ...
Chemistry 3202 Name: Acid-base Theory Problems Assignment 1
... from a hydrochloric acid solution. Label the reactants as acids or bases. ...
... from a hydrochloric acid solution. Label the reactants as acids or bases. ...
Slide 1
... ions act as a catalyst to increase the rate of the reaction and • Secondly it reacts with the water formed to shift the position of the equilibriumm to the right hand side (Le Chatelier’s principle) ensuring a good yield of product. • Note that unlike the acid and alcohol, an ester does not contain ...
... ions act as a catalyst to increase the rate of the reaction and • Secondly it reacts with the water formed to shift the position of the equilibriumm to the right hand side (Le Chatelier’s principle) ensuring a good yield of product. • Note that unlike the acid and alcohol, an ester does not contain ...
Acids and Bases
... Dissociation • In water all ionic compounds dissociate into its ionic parts • So NaCl in water dissociates into Na+ and Cl• So H3PO4 dissociates into 3H+ and PO4-3 • Remembers ionic compounds are formed by metals and nonmetals or by metals and polyatomic ions ...
... Dissociation • In water all ionic compounds dissociate into its ionic parts • So NaCl in water dissociates into Na+ and Cl• So H3PO4 dissociates into 3H+ and PO4-3 • Remembers ionic compounds are formed by metals and nonmetals or by metals and polyatomic ions ...
material safety data sheet
... MATERIAL SAFETY DATA SHEET I - IDENTIFICATION AND USE PRODUCT NAME: FLO-CLENE PRODUCT USE: LIQUID DRAIN OPENER SUPPLIER: ...
... MATERIAL SAFETY DATA SHEET I - IDENTIFICATION AND USE PRODUCT NAME: FLO-CLENE PRODUCT USE: LIQUID DRAIN OPENER SUPPLIER: ...
5 - Polyprotic Acids
... The second proton dissociation is much ______________ and therefore usually only consider the ___________________________ Generally, we only consider the ______ dissociation when calculating [H3O+] and pH Subsequent dissociations will be ________________ by hydronium _____________ from the first dis ...
... The second proton dissociation is much ______________ and therefore usually only consider the ___________________________ Generally, we only consider the ______ dissociation when calculating [H3O+] and pH Subsequent dissociations will be ________________ by hydronium _____________ from the first dis ...
Polyesters are condensation polymers.
... Note that sometimes other molecules (HCl for eg) are lost in other condensation reactions. It is the elimination of a molecule which makes it a condensation reaction, not the loss of water. ...
... Note that sometimes other molecules (HCl for eg) are lost in other condensation reactions. It is the elimination of a molecule which makes it a condensation reaction, not the loss of water. ...
Synthesis of an Ester: Fischer Esterification The ester is synthesised
... - It speeds up the reaction rate, allowing equilibrium to be reached more quickly - It acts as a dehydrating agent, helping to shift the equilibrium towards products (alcohols are weak nucleophiles – protonation of the carbonyl group enhances the reactivity of the acid, meaning it is more readily at ...
... - It speeds up the reaction rate, allowing equilibrium to be reached more quickly - It acts as a dehydrating agent, helping to shift the equilibrium towards products (alcohols are weak nucleophiles – protonation of the carbonyl group enhances the reactivity of the acid, meaning it is more readily at ...
Name / Functional Group
... 6. What kinds of chemical compounds are aspirin (acetyl salicylic acid) and methyl salicylate (i.e., alcohols, aldehydes, ketones, acids, esters, amides, etc)? __________________ 7. How would you describe the aroma of methyl salicylate? _____________________________ 8. If you combine the organic aci ...
... 6. What kinds of chemical compounds are aspirin (acetyl salicylic acid) and methyl salicylate (i.e., alcohols, aldehydes, ketones, acids, esters, amides, etc)? __________________ 7. How would you describe the aroma of methyl salicylate? _____________________________ 8. If you combine the organic aci ...
Answer on Question #44399 – Chemistry – Other HC2O4 − + HOH
... Answer According to the Brønsted–Lowry theory, an acid is a species able to lose, or "donate" a proton (H+) while a base is a species with the ability to gain, or "accept," a proton. The hydrogen oxalate ion can gain a proton acting as a base towards water, while the latter donates proton acting as ...
... Answer According to the Brønsted–Lowry theory, an acid is a species able to lose, or "donate" a proton (H+) while a base is a species with the ability to gain, or "accept," a proton. The hydrogen oxalate ion can gain a proton acting as a base towards water, while the latter donates proton acting as ...
othschem.pbworks.com
... OH group and a carbonyl group. They are quite similar to alcohols, however have the characteristics of acids such as turning litmus paper red (an excellent test for this group only) and neutralizing base. Ethers The structure of an ether is similar to water. Rather than a hydrogen on each side of th ...
... OH group and a carbonyl group. They are quite similar to alcohols, however have the characteristics of acids such as turning litmus paper red (an excellent test for this group only) and neutralizing base. Ethers The structure of an ether is similar to water. Rather than a hydrogen on each side of th ...
Synthesis of Esters Problem: Produce an ester that smells
... compounds. The pharmaceutical, agrochemical, and biotechnology industries use combinatorial chemistry to reduce the time and cost associated with the discovery of new drugs, pesticides, and peptides. Instead of making one compound at a time and testing it for desirable properties, hundreds of compou ...
... compounds. The pharmaceutical, agrochemical, and biotechnology industries use combinatorial chemistry to reduce the time and cost associated with the discovery of new drugs, pesticides, and peptides. Instead of making one compound at a time and testing it for desirable properties, hundreds of compou ...
Sulfuric acid
Sulfuric acid (alternative spelling sulphuric acid) is a highly corrosive strong mineral acid with the molecular formula H2SO4 and molecular weight 98.079 g/mol. It is a pungent-ethereal, colorless to slightly yellow viscous liquid which is soluble in water at all concentrations. Sometimes, it is dyed dark brown during production to alert people to its hazards. The historical name of this acid is oil of vitriol.Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration. It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed. Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.Sulfuric acid has a wide range of applications including domestic acidic drain cleaner, electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.