LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034 PART-A
... 12. State and explain the Hammond postulate to the bromination of n-propane. 13. How will you determine the reaction mechanism of hydrolysis of an ester using isotoping labeling method? 14. Write and explain the Steven’s rearrangement. 15. Predict the product and explain the mechanism of the followi ...
... 12. State and explain the Hammond postulate to the bromination of n-propane. 13. How will you determine the reaction mechanism of hydrolysis of an ester using isotoping labeling method? 14. Write and explain the Steven’s rearrangement. 15. Predict the product and explain the mechanism of the followi ...
Alkene Addition Reactions
... The shifting group migrates with its pair of electrons therefore the name hydride (H-‐) or methide (CH3-‐). The order of migrating groups is H > CH3. Alcohols can be produced by addition of ...
... The shifting group migrates with its pair of electrons therefore the name hydride (H-‐) or methide (CH3-‐). The order of migrating groups is H > CH3. Alcohols can be produced by addition of ...
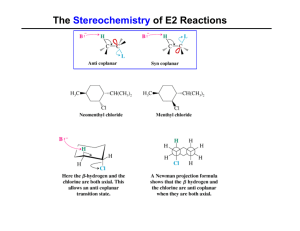
The Stereochemistry of E2 Reactions
... Dehydration of Alcohols: E1 Mechanism Rate Limiting Step ...
... Dehydration of Alcohols: E1 Mechanism Rate Limiting Step ...
Workshop 9
... reaction occurs and is based on the known experimental data about the reaction. Some reaction mechanisms are well established. In other cases they may be speculative and are likely to change as more data become available. Mechanisms map the path by which the reactants change into products and the mo ...
... reaction occurs and is based on the known experimental data about the reaction. Some reaction mechanisms are well established. In other cases they may be speculative and are likely to change as more data become available. Mechanisms map the path by which the reactants change into products and the mo ...
Senior Science topics Programme
... Teachers may discuss with students how scientists combine basic chemical principles, understanding of the reactivity of different chemical species with experimental observations to deduce the most likely sequence of elementary steps and thus the mechanism of a particular reaction. Knowledge of react ...
... Teachers may discuss with students how scientists combine basic chemical principles, understanding of the reactivity of different chemical species with experimental observations to deduce the most likely sequence of elementary steps and thus the mechanism of a particular reaction. Knowledge of react ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 14. a) Write the mechanism for the reduction of CH3COOC2H5 by LAH. b) How will you prepare isobutyl alcohol using a Grignard reagent? 15. An amide(A) having molecular formula C3H7ON on hydrolysis gives an acid C3H6O2(B) which upon chlorination in the presence of red phosphorus produces a chloroacid( ...
... 14. a) Write the mechanism for the reduction of CH3COOC2H5 by LAH. b) How will you prepare isobutyl alcohol using a Grignard reagent? 15. An amide(A) having molecular formula C3H7ON on hydrolysis gives an acid C3H6O2(B) which upon chlorination in the presence of red phosphorus produces a chloroacid( ...
- EdShare - University of Southampton
... The questions in this resource are based on past exam questions, and are designed to be challenging. The purpose of this resource is as a self-assessment exercise, which you can then look back over using specially made talking mark schemes, explaining the answers to the problems, the process in answ ...
... The questions in this resource are based on past exam questions, and are designed to be challenging. The purpose of this resource is as a self-assessment exercise, which you can then look back over using specially made talking mark schemes, explaining the answers to the problems, the process in answ ...
- EdShare - University of Southampton
... The questions in this resource are based on past exam questions, and are designed to be challenging. The purpose of this resource is as a self-assessment exercise, which you can then look back over using specially made talking mark schemes, explaining the answers to the problems, the process in answ ...
... The questions in this resource are based on past exam questions, and are designed to be challenging. The purpose of this resource is as a self-assessment exercise, which you can then look back over using specially made talking mark schemes, explaining the answers to the problems, the process in answ ...
Chapter 9. Addition Reactions of Alkenes
... Biosynthesis of a prostaglandin from arachidonic acid: intermediate intramolecular radical addition ...
... Biosynthesis of a prostaglandin from arachidonic acid: intermediate intramolecular radical addition ...
Elimination reactions under acidic conditions
... product is produced more, which is the left most product due to the maximum amount of sigma bonds. ...
... product is produced more, which is the left most product due to the maximum amount of sigma bonds. ...
Microsoft Word - Final Exam Study Guide
... stability, elimination reactions, Zaitsev’s rule, E1 mechanism, E2 mechanism, antiperiplanar, comparing substitution and elimination mechanisms, synthesis of ethers, alcohols, and epoxides, dehydration of alcohols, carbocation rearrangements, reactions of alcohols/ethers/epoxides, multistep synthesi ...
... stability, elimination reactions, Zaitsev’s rule, E1 mechanism, E2 mechanism, antiperiplanar, comparing substitution and elimination mechanisms, synthesis of ethers, alcohols, and epoxides, dehydration of alcohols, carbocation rearrangements, reactions of alcohols/ethers/epoxides, multistep synthesi ...
Elimination Reactions
... mechanisms Describe how to shift equilibrium in favor of elimination or addition Predict the major product according to alkene stability Daily Problems 1. Provide a mechanism for these elimination reactions of alcohols under acidic conditions. ...
... mechanisms Describe how to shift equilibrium in favor of elimination or addition Predict the major product according to alkene stability Daily Problems 1. Provide a mechanism for these elimination reactions of alcohols under acidic conditions. ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... a) When nitrobenzene is nitrated m-dinitrobenzene is formed as the major product whereas anisole on nitration forms a mixture of o- and p-nitro compounds. Explain. b) How are the following conversions done? i) phenol to cyclohexane ii) cyclopropane to butanoic acid. ...
... a) When nitrobenzene is nitrated m-dinitrobenzene is formed as the major product whereas anisole on nitration forms a mixture of o- and p-nitro compounds. Explain. b) How are the following conversions done? i) phenol to cyclohexane ii) cyclopropane to butanoic acid. ...
Dehydration of 3,3-dimethyl-2-butanol to make alkenes March 1 & 3
... – Drain lower aqueous layer into a beaker ...
... – Drain lower aqueous layer into a beaker ...
2015 CH 420 Take Home Quiz 3 March 24
... 4) Cassava root is extensively cultivated and consumed in tropical and subtropical regions, though it must be properly prepared to avoid toxicity due to breakdown of cyanogenic glucosides such as linamarin. Enzymatic hydrolysis of linamarin releases glucose and a cyanohydrin. The cyanohydrin further ...
... 4) Cassava root is extensively cultivated and consumed in tropical and subtropical regions, though it must be properly prepared to avoid toxicity due to breakdown of cyanogenic glucosides such as linamarin. Enzymatic hydrolysis of linamarin releases glucose and a cyanohydrin. The cyanohydrin further ...
Synthesis of a Family of Chiral Asymmetric Schiff - Blogs at H-SC
... Condensation reactions of carbonyl compounds are an important class of such reactions. Chiral organometallic compounds have been shown in some cases to act as catalysts to give condensation products in high yields and with high ...
... Condensation reactions of carbonyl compounds are an important class of such reactions. Chiral organometallic compounds have been shown in some cases to act as catalysts to give condensation products in high yields and with high ...
Document
... Diazomethane forms methylene, which converts alkenes into cyclopropanes. The highly reactive species methylene, H2C: (the simplest carbene) can be produced from the decomposition of diazomethane: ...
... Diazomethane forms methylene, which converts alkenes into cyclopropanes. The highly reactive species methylene, H2C: (the simplest carbene) can be produced from the decomposition of diazomethane: ...
Microsoft Word - Final Exam Study Guide
... synthesis of ethers, alcohols, and epoxides, dehydration of alcohols, carbocation rearrangements, reactions of alcohols/ethers/epoxides, multistep synthesis, protecting groups, redox reactions, reagents for redox reactions, Grignard reaction ...
... synthesis of ethers, alcohols, and epoxides, dehydration of alcohols, carbocation rearrangements, reactions of alcohols/ethers/epoxides, multistep synthesis, protecting groups, redox reactions, reagents for redox reactions, Grignard reaction ...
1. Rank the following compounds in order of decreasing acidity (1
... 1. Rank the following compounds in order of decreasing acidity (1 = most acidic à 5 = least acidic): O ...
... 1. Rank the following compounds in order of decreasing acidity (1 = most acidic à 5 = least acidic): O ...
Chem 2641 Chapter 5 Understanding Organic Reactions I. Writing
... B. Reactions can be one step or more than one step. 1. Reactions that are one step are called concerted reactions. The reactant is converted directly to product. 2. Reactions that require more than one step contain species ...
... B. Reactions can be one step or more than one step. 1. Reactions that are one step are called concerted reactions. The reactant is converted directly to product. 2. Reactions that require more than one step contain species ...
Chapter 7: Structure and Synthesis of Alkenes
... Ú Alkenes are hydrocarbons with C=C in their structure. They are also known as “olefins”. Ú We have already covered the basics of alkenes nomenclature, but here are a few important groups with common names that you can use as well. ...
... Ú Alkenes are hydrocarbons with C=C in their structure. They are also known as “olefins”. Ú We have already covered the basics of alkenes nomenclature, but here are a few important groups with common names that you can use as well. ...
C:\Users\mrh70950\Documents\My Files\WordPerfect
... i. anti-addition stereochemistry yields 1-alkenes from terminal alkynes and (E)-alkenes from internal alkynes 2. double hydrogenation: addition of 2 mol of H2 to yield alkanes a. noble metal catalyst + excess H2 3. electrophilic additions (all by very similar mechanisms) a. hydrohalogenation: addit ...
... i. anti-addition stereochemistry yields 1-alkenes from terminal alkynes and (E)-alkenes from internal alkynes 2. double hydrogenation: addition of 2 mol of H2 to yield alkanes a. noble metal catalyst + excess H2 3. electrophilic additions (all by very similar mechanisms) a. hydrohalogenation: addit ...
Vinylcyclopropane rearrangement

The vinylcyclopropane rearrangement or vinylcyclopropane-cyclopentene rearrangement is a ring expansion reaction, converting a vinyl-substituted cyclopropane ring into a cyclopentene ring.Intense experimental as well as computational investigations have revealed that mechanistically, the vinylcyclopropane rearrangement can be thought of as either a diradical-mediated two-step and/or orbital-symmetry-controlled pericyclic process. The amount by which each of the two mechanisms is operative is highly dependent on the substrate.Due to its ability to form cyclopentene rings the vinylcyclopropane rearrangement has served several times as a key reaction in complex natural product synthesis.