amino acids
... enzymes (speed up reactions in the body) structure (keratin, collagen) carriers & transport (hemoglobin, aquaporin) ...
... enzymes (speed up reactions in the body) structure (keratin, collagen) carriers & transport (hemoglobin, aquaporin) ...
Proteins
... 6. Label the type of bond used to make proteins. 7. Draw arrows to identify these bonds in your model 8. Label the N-terminus and C-terminus 9. Put SQUARES around the R groups 10. Use your amino acid chart to identify & label the type of R group (non-polar, polar, charge basic, charged acidic, etc) ...
... 6. Label the type of bond used to make proteins. 7. Draw arrows to identify these bonds in your model 8. Label the N-terminus and C-terminus 9. Put SQUARES around the R groups 10. Use your amino acid chart to identify & label the type of R group (non-polar, polar, charge basic, charged acidic, etc) ...
Proteins 1 - Dr Rob's A
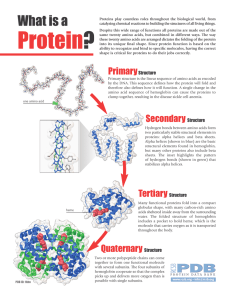
... Most proteins have α-helix regions and β-pleating But folding of the polypeptide chain into a compact, globular shape is called the tertiary structure The bending and folding is irregular Caused by formation of differing bonds between aa residues. ...
... Most proteins have α-helix regions and β-pleating But folding of the polypeptide chain into a compact, globular shape is called the tertiary structure The bending and folding is irregular Caused by formation of differing bonds between aa residues. ...
Interaction Site Evolution
... DNA is the blueprint for generating strings of amino acids which fold into proteins. The interactions these proteins form with each other are primary components of organismal physiology. Proteins assume very specific shapes, and the amino acids on their surface involved in protein interactions are s ...
... DNA is the blueprint for generating strings of amino acids which fold into proteins. The interactions these proteins form with each other are primary components of organismal physiology. Proteins assume very specific shapes, and the amino acids on their surface involved in protein interactions are s ...
Hands-on Exercise: Locating Protein Information
... Find the homologous mouse and fruit fly orthologs of this human protein and report the % protein shared identity. ...
... Find the homologous mouse and fruit fly orthologs of this human protein and report the % protein shared identity. ...
Describe the relationship between genes, nucleic acids, amino
... serve as enzymes that catalyze numerous and necessary chemical reactions that involve very stable molecules. DNA’s structure is critical to its function. It is organized in a manner that there are 30,000 different locations along the length of the molecule that specifically direct the production of ...
... serve as enzymes that catalyze numerous and necessary chemical reactions that involve very stable molecules. DNA’s structure is critical to its function. It is organized in a manner that there are 30,000 different locations along the length of the molecule that specifically direct the production of ...
Protein?
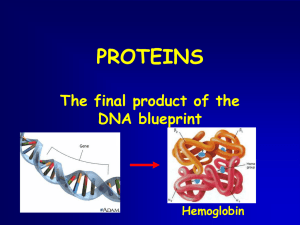
... Alpha helices (shown in blue) are the basic structural elements found in hemoglobin, but many other proteins also include beta sheets. The inset highlights the pattern of hydrogen bonds (shown in green) that stabilizes alpha helices. ...
... Alpha helices (shown in blue) are the basic structural elements found in hemoglobin, but many other proteins also include beta sheets. The inset highlights the pattern of hydrogen bonds (shown in green) that stabilizes alpha helices. ...
Competition
... together There are only 20 types of amino acids, but there are millions of proteins. Therefore the number of amino acids, and their sequence determine the protein. Only 12 of the amino acids can be made by our body, the rest have to come from our food (meat, nuts). These 8 are called Essential Amino ...
... together There are only 20 types of amino acids, but there are millions of proteins. Therefore the number of amino acids, and their sequence determine the protein. Only 12 of the amino acids can be made by our body, the rest have to come from our food (meat, nuts). These 8 are called Essential Amino ...
Amino acid sequence fingerprints in divergent evolution of
... are preserved, the resulting protein function and activity can remain without any observable modifications. The essential amino acid residues are mostly parts of the so-called conserved sequence regions that cover the isolated segments belonging to the active site of the protein. In the case the pro ...
... are preserved, the resulting protein function and activity can remain without any observable modifications. The essential amino acid residues are mostly parts of the so-called conserved sequence regions that cover the isolated segments belonging to the active site of the protein. In the case the pro ...
AS Biology - Everything Protein
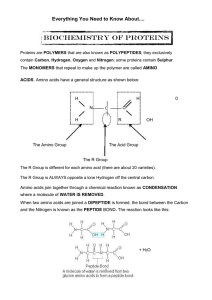
... where a molecule of WATER IS REMOVED. When two amino acids are joined a DIPEPTIDE is formed, the bond between the Carbon and the Nitrogen is known as the PEPTIDE BOND. The reaction looks like this: ...
... where a molecule of WATER IS REMOVED. When two amino acids are joined a DIPEPTIDE is formed, the bond between the Carbon and the Nitrogen is known as the PEPTIDE BOND. The reaction looks like this: ...
The Amino Acid Song
... The Amino Acid Song (to the tune of Old McDonald) Tracey Tripp, Nell Ditch, Julie Milam and Frances Jenkins Amino acids are the building blocks of protein And there are 20 of them. Nine of them we call essential Our body cannot make them. They are histidine, isoleucine, leucine, lysine, methionine, ...
... The Amino Acid Song (to the tune of Old McDonald) Tracey Tripp, Nell Ditch, Julie Milam and Frances Jenkins Amino acids are the building blocks of protein And there are 20 of them. Nine of them we call essential Our body cannot make them. They are histidine, isoleucine, leucine, lysine, methionine, ...
Protein Reading Questions Due Monday File
... 8. Explain the properties of the amino acid groups below, based on their R-group: a. Nonpolar side chains/Hydrophobic: b. Polar side chains/ Hydrophilic: c. Electrically charged side chains/Hydrophilic: 9. What are the bonds between amino acids in a polypeptide called AND what type of bond is it? ...
... 8. Explain the properties of the amino acid groups below, based on their R-group: a. Nonpolar side chains/Hydrophobic: b. Polar side chains/ Hydrophilic: c. Electrically charged side chains/Hydrophilic: 9. What are the bonds between amino acids in a polypeptide called AND what type of bond is it? ...
Let`s Get Pumped Up about Proteins!!!
... • Biochemical compounds that contain nitrogen as well as C, H, O, and sometimes S. (Remember: CHNOS) • consist of 1 or more polypeptide chains folded and coiled into specific conformations • What is the difference between a polypeptide and a protein? ...
... • Biochemical compounds that contain nitrogen as well as C, H, O, and sometimes S. (Remember: CHNOS) • consist of 1 or more polypeptide chains folded and coiled into specific conformations • What is the difference between a polypeptide and a protein? ...
PowerPoint - Center for Biological Sequence Analysis
... Reading direction 4 backbone atoms Amino acid properties ...
... Reading direction 4 backbone atoms Amino acid properties ...
Lectures 1-3: Review of forces and elementary statistical
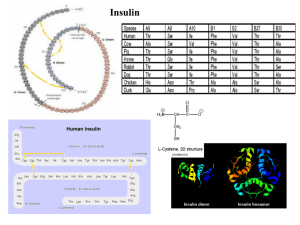
... Studying the three-dimensional structure of insulin As described above, human insulin consists of 51 amino acids, divided into two chains, commonly labeled A and B, with 21 and 30 amino acids respectively. The chains are linked by three disulfide bridges, two forming inter-chain cystine at A7-B7 and ...
... Studying the three-dimensional structure of insulin As described above, human insulin consists of 51 amino acids, divided into two chains, commonly labeled A and B, with 21 and 30 amino acids respectively. The chains are linked by three disulfide bridges, two forming inter-chain cystine at A7-B7 and ...
Proteins have a higher order of folding known as tertiary structure
... Proteins have a higher order of folding known as tertiary structure. The tertiary structure is a description of the way the whole chain (including the secondary structural features) folds itself into a 3D shape. The structure is held in place by interactions between the side chains (R groups) of the ...
... Proteins have a higher order of folding known as tertiary structure. The tertiary structure is a description of the way the whole chain (including the secondary structural features) folds itself into a 3D shape. The structure is held in place by interactions between the side chains (R groups) of the ...
CH 107 SI Summer 2015 Worksheet 13 Answers What are the two
... 1. What are the two major types of secondary protein structure and what bonds are present in each? α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydr ...
... 1. What are the two major types of secondary protein structure and what bonds are present in each? α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydr ...
Chapter 20 Amino acids and proteins
... 1. Draw the ionic form of an amino acid at the pH above the pI, at the pI and below the pI. 2. Briefly, summarize the process of electrophoresis. 3. Given three or four amino acids, their pIs, and the pH of the buffer, determine the movement of the amino acids on an electrophoresis gel. 20.4 formati ...
... 1. Draw the ionic form of an amino acid at the pH above the pI, at the pI and below the pI. 2. Briefly, summarize the process of electrophoresis. 3. Given three or four amino acids, their pIs, and the pH of the buffer, determine the movement of the amino acids on an electrophoresis gel. 20.4 formati ...
Pipe Cleaner Protein
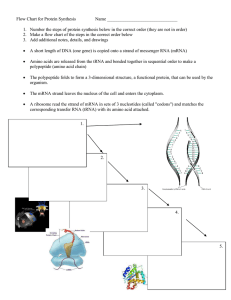
... ◦ DNA sequence written out ◦ mRNA sequence written out ◦ Amino acid sequence written out ...
... ◦ DNA sequence written out ◦ mRNA sequence written out ◦ Amino acid sequence written out ...
Protein structure prediction

Protein structure prediction is the prediction of the three-dimensional structure of a protein from its amino acid sequence — that is, the prediction of its folding and its secondary, tertiary, and quaternary structure from its primary structure. Structure prediction is fundamentally different from the inverse problem of protein design. Protein structure prediction is one of the most important goals pursued by bioinformatics and theoretical chemistry; it is highly important in medicine (for example, in drug design) and biotechnology (for example, in the design of novel enzymes). Every two years, the performance of current methods is assessed in the CASP experiment (Critical Assessment of Techniques for Protein Structure Prediction). A continuous evaluation of protein structure prediction web servers is performed by the community project CAMEO3D.