* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Conservation of Mass
History of electrochemistry wikipedia , lookup
Electromigration wikipedia , lookup
Lorentz force wikipedia , lookup
Electromotive force wikipedia , lookup
Electric charge wikipedia , lookup
Electrostatics wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
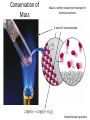
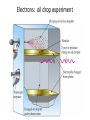
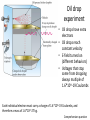
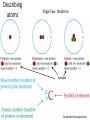
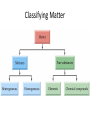
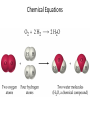
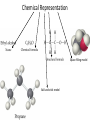
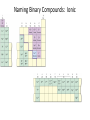
Conservation of Mass Mass is neither created nor destroyed in chemical reactions. 3 levels of representation 2 HgOs 2 Hg l O2 g Comprehension question Law of Definite Proportions Different samples of a pure chemical substance always contain the same proportion of elements by mass. Comprehension question Dalton’s Atomic Theory • Elements are made of tiny particles called atoms • The atoms of each element have the same unique mass • Chemical reactions make new substances by combining atoms in small whole-number ratios • Chemical reactions leave the atoms themselves unchanged. Only the groupings of atoms changes. Dalton’s atomic theory predicts. . . Law of Multiple Proportions Elements can combine in different ways to form different substances, whose mass ratios are small wholenumber ratios of each other. How many ratios are there in the law of multiple proportions? Comprehension question Electrons: Cathode-ray experiment Electrons: oil drop experiment Electric & Magnetic Fields All electrically charged particles are surrounded by an e-field (a field of electrical attraction/repulsion). This field is very analogous to the emanation of gravity from a mass. Other charged particles feel this emanation like mass feels gravity*—charge is accelerated by it. The force depends only on the strength of the field (E) and the strength of the charge (e) A charged particle doesn’t interact at all with a magnetic field unless it is moving. In that case, it is accelerated in a direction that depends upon the sign of the charge, and rotated 90 degrees to the acceleration from an e-field. The force depends upon the strength of the magnetic field (H) and the magnitude of the charge (e) and the velocity of the particle (v). Picture from: home.att.net/~numericana/arms/crt.gif Crookes tube From Reasons to Ratios Deflection Controllers • A. Strength of magnetic field (H) • B. Charge magnitude of electric unit (e) • C. Mass of electric unit (m) • D. Speed of electric unit (v) Experiment: Hev Ee E v H Magnetic force Electric force He knows H and E! He “dialed them in” to his experiment! So he can calculate v (~0.2 c) With knowledge of the velocity, he can solve deflection in the e-field just like a gravity problem to give him an e/m ratio: 1*10^8 Coulombs/gram Comprehension Huge result!!! The race is on! question Oil drop experiment • Oil drops have extra electrons • Oil drops reach constant velocity • E-field turned on (different behaviors) • Voltages that stop some from dropping always multiple of 1.6*10^-19 Coulombs Each individual electron must carry a charge of 1.6*10^-19 Coulombs, and therefore a mass of 1.6*10^-27 kg. Comprehension question Gold foil experiment • If atom is continuous and uniform, alpha particles should show ~0° deflection • 1 out of 100,000 did something different Nuclear Model of the Atom Subatomic Particles Comprehension question Describing atoms Stage One: Protons and Electrons Describing atoms Stage Two: Neutrons Isotopes Comprehension questions Describing elements Atomic Mass mass of isotope 1abundance of isotope 1 Atomic mass of X mass of isotope 2abundance of isotope 2 Actual masses, NOT atomic numbers You are calculating a weighted average. Percent abundances (must sum to 100%) The masses for these calculations are (generally) in amu, not grams. Comprehension question Classifying Matter Particulate Drawings In particulate drawings, atoms are represented by shapes and one focuses on the organization of the shapes rather than what the individual shapes represent. A fully classified particulate drawing has the phase, the state of purity, and the nature of each molecule fully labelled. # of atoms (mono/di/tri) # of elements (homo/hetero) gas phase, monatomic element Gas Liquid Solid Mixture (hetero/homo) Element Compound heterogeneous mixture of two solid phases, one a heteroatomic diatomic compound and the other a heteroatomic triatomic compound Chemical Equations Chemical Bonding: Covalent Electron sharing Only bonding type that makes molecules Generally between nonmetals Chemical Representation Space-filling model Ball-and-stick model Chemical Bonding: Ionic ions Giving and taking of electrons results in no molecules, instead we talk about ionic substances (usually ionic solids) Generally between a metal and non-metal The ratios of positive ions (cations) to negative (ions) makes the total charge on the ionic substance equal to zero. Polyatomic Ions Acids and Bases A compound that dissolves in water and yields H+ ions is an acid. A compound that dissolves in water and yields OH- ions in water is a base. Naming Binary Compounds: Ionic Naming Binary Compounds: Covalent Learning from the name The initial observation of the name should tell you whether the compound is ionic or covalent. How does it do that? •Numerical prefixes? Must be covalent (watch for polyatomic ion exceptions) •Ends in –ite or –ate? Must be ionic •Neither of those two conditions apply? Check metal vs. nonmetal of leading element. Once you’ve got the bonding type, try to produce the formula. •For covalent, use the prefixes and be done. •For ionic, figure out the two ions and then balance the charge. For now, the first element in the formula is the first element in the name. Learning from the formula The initial observation of the formula should tell you whether the compound is ionic or covalent. How does it do that? •More than two elements? Must be ionic •Metal bonded to nonmetal? Must be ionic •Non-metal bonded to non-metal? Must be covalent If its covalent, put in the numerical prefixes and you’re done. (Remember to avoid “ao” or “oo” combinations. If its ionic, determine the two ions involved and write cation then anion and you’re done. (Remember that if the cation is a transition/post-transition metal, the ion is named with the charge in roman numerals inside parentheses (usually). For now, the first element in the formula is the first element in the name.