* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Plant Tissue Culture
Plant tolerance to herbivory wikipedia , lookup
Gartons Agricultural Plant Breeders wikipedia , lookup
History of herbalism wikipedia , lookup
Evolutionary history of plants wikipedia , lookup
Ornamental bulbous plant wikipedia , lookup
Plant stress measurement wikipedia , lookup
Historia Plantarum (Theophrastus) wikipedia , lookup
Flowering plant wikipedia , lookup
Venus flytrap wikipedia , lookup
History of botany wikipedia , lookup
Plant nutrition wikipedia , lookup
Plant use of endophytic fungi in defense wikipedia , lookup
Plant defense against herbivory wikipedia , lookup
Plant secondary metabolism wikipedia , lookup
Plant evolutionary developmental biology wikipedia , lookup
Plant reproduction wikipedia , lookup
Plant breeding wikipedia , lookup
Plant physiology wikipedia , lookup
Plant morphology wikipedia , lookup
Plant ecology wikipedia , lookup
Sustainable landscaping wikipedia , lookup
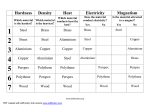
Plant Tissue Culture Experiment 1: Effect of pH on Nutrition and Growth of Tissue Cultured Ginger Introduction Plant tissue culture is the cultivation of plant cells or tissues on specially-formulated nutrient media. In appropriate conditions, an entire plant can be regenerated from each single cell, permitting the rapid production of many identical plants. Tissue culture is an essential tool in modern plant breeding. Since it was first developed in the early 1960s, plant tissue culture has become the basis of a major industry, providing high-value plants for nurseries. Where a crop (e.g., banana) does not produce seeds, plants derived from tissue culture are sometimes planted directly in farmers' fields. However this is rarely done for seed-bearing species - more often tissue-cultured plants are used to produce the seeds from which crops. Cell culture is also used for the conservation of those plant varieties that cannot be maintained in a normal seed bank. The chemical compounds that make up plants are mainly carbohydrates, lipids, proteins and nucleic acids. These are all organic compounds and contain the elements carbon, hydrogen and oxygen. Proteins and nucleic acids also contain nitrogen and phosphorus. Proteins can also contain other elements (eg chlorophyll contains magnesium). These elements are usually incorporated into tissue culture media as nutrient salts according to the recipe of Murashige and Skoog. Their availability is affected by pH, which is known to change over time in culture. Students will work in groups of 2-3 and will aseptically transfer ginger tissue culture plants to media containing MS nutrients at 3 pH levels (3, 6 and 9) Aim To investigate the effect pH has on nutrient availability on tissue cultured plants as measured by changes in the plant. Materials 18 cultured ginger plants Perspex box Scalpel Alcohol burner Ethanol Bunsen burner Swabs (cotton balls) Ceramic tile Auto-claved paper Cling wrap Tweezers Specific media (pH 3, 6 and 9) Lab coat Face mask Hair net Gloves Permanent markers 18 Plastic tubes with sealable lids Method Contamination considerations The environment used when carrying out the experiment needs to be completely sterile. Making sure the area, equipment and the students are totally aseptic is essential to the success of the experiment. These aseptic techniques ensure there is no contamination and prevents bacterial or germ growth on the experiment. Using products like Ethanol and heat from the Bunsen burner assists in sterilizing all utensils preventing contamination. Step 1. Swab interior of Perspex box liberally with ethanol and cotton buds Step 2. Sterilize hands with lathering ethanol over them. Step 3. Sterilize each utensil to be used in the Perspex box by holding it over an open flame. All utensils that come into contact with the plant are heated by the flame. 1. Working within the Perspex box (laminar flow cabinet) use sterile tweezers to remove the sealed auto-claved paper and place it on the tile inside the Perspex box. 2. Collect the Ginger plants of similar size and colour. Inside the Perspex box remove the ginger plantlets with tweezers and place them one at a time on the auto-claved paper. 3. Using a sterile scalpel and tweezers measure the plantlet to the desired length and remove the leaves and roots cutting the plantlet to the desired length. Ensure that the rhizome remains as part of the plantlet. 4. Using the tweezers place the cut plantlet into a corresponding tube with the correct media. Ensure the plant is placed securely in the agar and seal the tube. Label the tube correctly. 6 x pH 3 media 6 x pH 6 media 6 x pH 9 media 5. Repeat the steps 1-5 for each of the ginger plantlets. 6. Place the plants under lights in a controlled environment. 7. Each week plants are to be measured and data recorded including contamination, height colour etc 8. After 7-8 weeks of growth a deconstructive harvest will take place. This involves collecting the final set of data and measurements by removing each plant from the agar tube. These results collected should include weighing the plants (whole plant, leaves, rhizome etc), final height measurements and photos may be taken. 9. After each plant is removed and measured discard the plant and remove the agar from each tube. Plant Tissue Culture Experiment 2: Effect of Light and Sucrose on Growth of Tissue Cultured Ginger Introduction Plant tissue culture is the cultivation of plant cells or tissues on specially-formulated nutrient media. In appropriate conditions, an entire plant can be regenerated from each single cell, permitting the rapid production of many identical plants. Tissue culture is an essential tool in modern plant breeding. Since it was first developed in the early 1960s, plant tissue culture has become the basis of a major industry, providing high-value plants for nurseries. Where a crop (e.g., banana) does not produce seeds, plants derived from tissue culture are sometimes planted directly in farmers' fields. However this is rarely done for seed-bearing species - more often tissue-cultured plants are used to produce the seeds from which crops. Cell culture is also used for the conservation of those plant varieties that cannot be maintained in a normal seed bank. Plants are autotrophic; via photosynthesis they use carbon dioxide and water to make carbohydrates and lipids. Plants in tissue culture are unable to photosynthesize effectively so tissue culture media almost always has sucrose added as a readily available source to stimulate growth and development. Tissue cultured plants can therefore be grown as heterotrophic organisms. Students will work in groups of 2-3 and will aseptically transfer ginger tissue culture plants to media containing MS nutrients and some media with added sucrose. Aim To investigate the effect of light and sucrose on the growth and development of tissue cultured plants as measured by changes in the plant. Materials 24 cultured ginger plants Perspex box Tweezers Scalpel Alcohol burner Ethanol Bunsen burner Swabs (cotton balls) Ceramic tile Auto-claved paper Cling wrap Foil 24 Plastic tubes with sealable lids Specific media (MS media and MS media + sucrose) Lab coat Face mask Hair net Gloves Permanent markers Method Contamination considerations The environment used when carrying out the experiment needs to be completely sterile. Making sure the area, equipment and the students are totally aseptic is essential to the success of the experiment. These aseptic techniques ensure there is no contamination and prevents bacterial or germ growth on the experiment. Using products like Ethanol and heat from the Bunsen burner assists in sterilizing all utensils preventing contamination. Step 1. Swab interior of Perspex box liberally with ethanol and cotton buds Step 2. Sterilize hands with lathering ethanol over them. Step 3. Sterilize each utensil to be used in the Perspex box by holding it over an open flame. All utensils that come into contact with the plant are heated by the flame. 1. Working within the Perspex box (laminar flow cabinet) use sterile tweezers to remove the sealed auto-claved paper and place it on the tile inside the Perspex box. 2. Collect the Ginger plants of similar size and colour. Inside the Perspex box remove the ginger plantlets with tweezers and place them one at a time on the auto-claved paper. 3. Using a sterile scalpel and tweezers measure the plantlet to the desired length and remove the leaves and roots cutting the plantlet to the desired length. Ensure that the rhizome remains as part of the plantlet. 4. Using the tweezers place the cut plantlet into a corresponding tube with the correct media. Ensure the plant is placed securely in the agar and seal the tube. Label the tube correctly. 12 x MS media (6 to be kept in light, 6 in dark) 12 x MS + sucrose media (6 to be kept in light, 6 in dark) 5. Repeat the steps 1-5 for each of the ginger plantlets. 6. Those plants that are to be kept in full light are to be placed under lights in a controlled environment and those that are to be kept in the dark are to be carefully wrapped in alfoil and placed in the same controlled environment. 7. Each week plants are to be measured and data recorded including contamination, height colour etc 8. After 7-8 weeks of growth a deconstructive harvest will take place. This involves collecting the final set of data and measurements by removing each plant from the agar tube. These results collected should include weighing the plants (whole plant, leaves, rhizome etc), final height measurements and photos may be taken. 9. After each plant is removed and measured discard the plant and remove the agar from each tube. Plant Tissue Culture Experiment 3: Effect of Hormones on Growth of Tissue Cultured Ginger Introduction Plant tissue culture is the cultivation of plant cells or tissues on specially-formulated nutrient media. In appropriate conditions, an entire plant can be regenerated from each single cell, permitting the rapid production of many identical plants. Tissue culture is an essential tool in modern plant breeding. Since it was first developed in the early 1960s, plant tissue culture has become the basis of a major industry, providing high-value plants for nurseries. Where a crop (e.g., banana) does not produce seeds, plants derived from tissue culture are sometimes planted directly in farmers' fields. However this is rarely done for seed-bearing species - more often tissue-cultured plants are used to produce the seeds from which crops. Cell culture is also used for the conservation of those plant varieties that cannot be maintained in a normal seed bank. Natural plant hormones and their synthetic relatives have enormous benefits for tissue culture of plants. Plant hormones increase productivity and control of stages of development during culture. There are two broad classes of hormone used in culture; cytokinins (eg BAP) to control shoot proliferation and multiplication and auxins (eg IBA) to control root development. Students will work in groups of 2-3 and will aseptically transfer ginger tissue culture plants to media containing MS nutrients and some media with high or low levels of the hormone cytokinin. Aim To investigate the effect of cytokinin on the growth and development of tissue cultured plants as measured by changes in the plant. Materials 18 cultured ginger plants Tweezers Scalpel Alcohol burner Ethanol Bunsen burner Swabs (cotton balls) Ceramic tile Auto-claved paper Cling wrap 18 Plastic tubes with sealable lids Specific media (MS media, MS media + low level cytokinin and MS media + high level cytokinin) Lab coat Face mask Hair net Gloves Permanent markers Perspex box Method Contamination considerations The environment used when carrying out the experiment needs to be completely sterile. Making sure the area, equipment and the students are totally aseptic is essential to the success of the experiment. These aseptic techniques ensure there is no contamination and prevents bacterial or germ growth on the experiment. Using products like Ethanol and heat from the Bunsen burner assists in sterilizing all utensils preventing contamination. Step 1. Swab interior of Perspex box liberally with ethanol and cotton buds Step 2. Sterilize hands with lathering ethanol over them. Step 3. Sterilize each utensil to be used in the Perspex box by holding it over an open flame. All utensils that come into contact with the plant are heated by the flame. 1. Working within the Perspex box (laminar flow cabinet) use sterile tweezers to remove the sealed auto-claved paper and place it on the tile inside the Perspex box. 2. Collect the Ginger plants of similar size and colour. Inside the Perspex box remove the ginger plantlets with tweezers and place them one at a time on the auto-claved paper. 3. Using a sterile scalpel and tweezers measure the plantlet to the desired length and remove the leaves and roots cutting the plantlet to the desired length. Ensure that the rhizome remains as part of the plantlet. 4. Using the tweezers place the cut plantlet into a corresponding tube with the correct media. Ensure the plant is placed securely in the agar and seal the tube. Label the tube correctly. 6 x MS media 6 x MS media + low level cytokinin 6 x MS media + high level cytokinin 5. Repeat the steps 1-5 for each of the ginger plantlets. 6. Place the plants under lights in a controlled environment. 7. Each week plants are to be measured and data recorded including contamination, height colour etc 8. After 7-8 weeks of growth a deconstructive harvest will take place. This involves collecting the final set of data and measurements by removing each plant from the agar tube. These results collected should include weighing the plants (whole plant, leaves, rhizome etc), final height measurements and photos may be taken. 9. After each plant is removed and measured discard the plant and remove the agar from each tube.