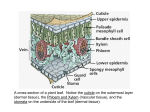
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Monocots vs
Plant use of endophytic fungi in defense wikipedia , lookup
Photosynthesis wikipedia , lookup
Plant stress measurement wikipedia , lookup
History of botany wikipedia , lookup
Plant defense against herbivory wikipedia , lookup
Evolutionary history of plants wikipedia , lookup
Plant nutrition wikipedia , lookup
Plant breeding wikipedia , lookup
Plant secondary metabolism wikipedia , lookup
Plant ecology wikipedia , lookup
Plant physiology wikipedia , lookup
Plant reproduction wikipedia , lookup
Plant morphology wikipedia , lookup
Sustainable landscaping wikipedia , lookup
Perovskia atriplicifolia wikipedia , lookup
Plant evolutionary developmental biology wikipedia , lookup
Monocotyledon wikipedia , lookup