* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Microbial Desulfurization
Survey
Document related concepts
Transcript
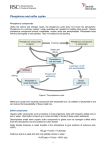
Microbial Desulfurization CHBE446: Process Economics and Design 2 6 February 2014 Heather Cook Savannah Green Dave Weglein Mike Wellen Outline • • • • • Introduction & History Mechanisms Current Uses in Industry Major Challenges & Advantages Current Research Introduction: What is Microbial Desulfurization? ● Also known as Biodesulfurization (BDS) ● Biological desulfurization process where microbial catalysts are used to oxidize sulfur in crude oils Introduction: Why is BDS important? ● Combustion of sulfur compounds leads to production of sulfur oxides ● High concentrations of sulfur oxides in the atmosphere can lead to health issues such as asthma, bronchial irritation, and lung cancer ● Ability to “desulfurize compounds that are recalcitrant to the current standard technology in the oil industry” (Abin-Fuentes, et al) New Regulations • Sulfur content in crude oil ranges from 0.03% - 7.89% • • Many crude oils are increasing in sulfur content Clean Air Act Amendments introduced by EPA in 1990 to restrict sulfur concentrations in fuels • Reduce annual SO2 emissions BDS Overview ● Increased interest over last 20 years ○ Desulfurizes wider range of compounds then conventional hydrodesulfurization (HDS) ● Three pathways ○ Kodama (destructive) ○ Anaerobic (selective) ○ 4S (specific oxidative) ● 4S is the most popular/effective Mechanisms Kodama Pathway ● Sulfur not selectively cleaved from dibenzothiopene (DBT) Carbon-carbon bonds broken Metabolize DBT’s & convert to water soluble compounds Water soluble products inhibits further microbial growth & DBT oxidation • • • Anaerobic pathway • • • • Anaerobic strain can degrade some of DBT Products: biphenyl & H2S o Makes this a selective pathway Advantage: o Oxidation of hydrocarbons to undesired compounds is minimal Disadvantage: o Reduced caloric content in fuel o Specific activity for most isolated strains are insignificant for alkylated DBTs 4S Pathway ● Carbon-sulfur bond selectively cleaved Bacteria Used 4S Pathway Enzymes • • • Reaction is energy-intensive and needs cellular metabolism The 4S pathway involves sequential oxidation of the sulfur part and cleaving of the C–S bonds Four main enzymes used in the 4S pathway DszC Enzyme • • • 45 kDa protein Catalyzes DBT->DBTO->DBTO2 Step uses oxygen, NADH, and FMNH2 for activity DszA Enzyme • • 50 kDa protein Transforms sulfone into sulfinate o • Uses FMH2 as cosubstrate Step requires oxygen and NADH as well o Oxygen from molecular oxygen DszB Enzyme • • 40 kDa protein Final step in the reaction • Rate limiting step o Present in cells in smaller amount in cytoplasm DszD Enzyme • • • Uses FMN as a substrate Couples the oxidation of NADH to substrate oxidation Produces FMNH2 to allow DszC and DszA to work Currents Uses in Industry Thiopaq • Biogas • • • • • • • • Vent air Refinery Gas Hydrogen Sulphide 120 Installations World-wide Reduces to under 25 ppm Fluctuating Gas Flows Low maintenance Ambient Pressures an Temperatures • Produces Elemental Sulfur Benefits of System • • • • • • Deep H2S removal and recovery as elemental S, extremely low SO2 emissions are achieved Special costly equipment such as burners and reboilers are not required. The regeneration and sulphur recovery section always operate at atmospheric pressure and ambient temperature Reliability of a natural process coupled with the efficiency of dedicated engineering Simple process configuration- and control with stable operation Broad and flexible operating range with short system start-up times Expensive chemicals such as those required for liquid redox processes are not required. Only sodium hydroxide and nutrients are needed More Benefits ● Limited utility requirements ● Ease of operation. Produced biosulphur is hydrophilic and behaves like a relatively stable suspension without clogging or other nuisances ● Environmentally friendly process based on naturally occurring bacteria ● Inherently safe operation: ○ no free H2S downstream absorber ○ ambient temperatures for the whole system (solution temperatures of 25 – 40 °C) ○ bioreactor and sulphur recovery at atmospheric pressure. ● Produced biosulphur is the basis for a range of new agricultural products designed to act as (ingredients for) liquid fertilizers and liquid fungicides Steps of Process • • • • • • • • • Sulfide rich solution loaded to flash drum Loaded to bioreactor Lean solution returned to absorber Lean solution returned to absorber Elemental Sulfur seperated out Bioreactor contents are recycled over settler Concentrated slurry dewatered in centrifuge Filtrate is cycled back Small slipstream of clear solvent Industrie Eerbek • Netherlands treats water from three neighboring paper mills • Biogas used to produce electricity • 1% to 25 ppm • Thiopaq system was installed in 1993 Ben & Jerry’s • • • • • Hellendoorn, Netherlands Ice cream waste products converted into electricity Desulfurized with Thiopaq 40% of factory's energy requirements Operational 2011 Cargill • • Starch processing company Sulfate rich water treated with anaerobic bioreactor Lenzig Ag • Viscose Fiber Production • 2009 produced 568,600 tonnes • Produces range of secondary compounds • Some streams need to be discharged. • SULFATEQ system installed in 2002 McCain • • • • Potato processing company Receives biogas from anaerobic water treatment and solids digester To prevent corrosion of gas engine, Thiopaq converts hydrogen sulfide to elemental sulfur Longer life for gas engine. Other Examples • • • • • • WaterStromen Hulshof Royal Dutch Tanneries Weltec BioPowr GmbH Tempec Nine Dragons Smurfit Kappa Challenges, Advantages & Current Research 5 Step Process • Production of active resting cells with high • • • • specific activity Preparation of biphasic system containing oil fraction, aqueous phase and biocatalyst BDS of wide range of sulfur compounds at acceptable rate Separation of desulfurized oil fraction, recovery of biocatalyst and return to bioreactor Efficient wastewater treatment Major Challenges • Biocatalyst activity improvement • Biocatalyst longevity improvement • Phase contact and separation • Process engineering research Current Research • • • • Reduction in biocatalyst activity associated with the generation of the end product (2hydroxybiphenyl) Increase of bacterial desulfurization rate through identification of certain genes Overexpression of FMN reductase Change in host strain for dsz genes Advantages of BDS • • • • • Requires less energy and hydrogen Operates at ambient temperature and pressure with high selectivity Decreased energy costs Low emissions No generation of undesired products Questions? References Abin-Fuentes, A., M. E.-S. Mohamed, D. I. C. Wang, and K. L. J. Prather. "Exploring the Mechanism of Biocatalyst Inhibition in Microbial Desulfurization." Applied and Environmental Microbiology 79.24 (2013): 7807-817. Web. 6 Feb. 2014. Mohebali, G., and A. S. Ball. "Biocatalytic Desulfurization (BDS) of Petrodiesel Fuels."Microbiology 154.8 (2008): 2169-183. Web. 6 Feb. 2014. Ohshiro, Takashi, and Yoshikazu Izumi. "Microbial Desulfurization of Organic Sulfur Compounds in Petroleum." Bioscience, Biotechnology, and Biochemistry 63.1 (1999): 1-9. Web. 6 Feb. 2014. Paqell | THIOPAQ O&G - Biological Gas Desulpherisation and Sulphur Recovery | Paqell."Paqell | THIOPAQ O&G - Biological Gas Desulpherisation and Sulphur Recovery | Paqell. Paqell BV, n.d. Web . 05 Feb. 2014. Soleimani, M., Bassi, A., and Margaritis, A. 2007. Biodesulfurization of refractory organic sulfur compounds in fossil fuels. Biotechnol. Adv. 25(6):570-96.