* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 03/08
Survey
Document related concepts
Transcript
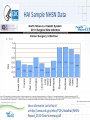
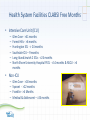
Patient Safety - Infection Prevention Donna Armellino, RN, DNP, CIC Vice President, Infection Prevention North Shore – LIJ Health System Infection Surveillance • Data is collected by staff that has certification by the Certification Board of Infection Control and Epidemiology, Inc. • Definition for healthcare-associated infections are from the Centers for Disease Control and Prevention’s National Healthcare Safety Network (NHSN) Superficial incisional SSI • Information used to screen for cases includes laboratory data, admission diagnosis, readmission data, Emergency Department chief complaint, return to the operating room, etc... Deep incisional SSI Organ/space SSI Healthcare-Acquired Infection (HAIs) Central Line Associated Bacteremias (CLABSI) – Intensive Care Units (ICU) – Non-ICU Ventilator Associated Pneumonias (VAPs) – ICU – Non-ICU Surgical Site Infections (SSIs) Select or all high volume procedures Catheter Associated Urinary Tract Infections (CAUTI) ICU Non-ICU Methicillin Resistant Staphylococcus aureus (MRSA) infections and colonization Facility-wide Clostridium difficile Facility-wide 3 Required HAI Monitoring and Reporting • New York State Department of Health (NYSDOH) and Center for Medicare & Medicaid Services (CMS) Through the National Healthcare Safety (NHSN): – Surgical procedure monitored and SSIs reported based on ICD-9 codes for: • • • • Hip Colon * CMS 01/01/12 Cardiac Hysterectomies *CMS 01/01/12 – Other HAIs: • Central line-associated bacteremias (CLABSI) *CMS 01/01/11 - ICU • Catheter-associated urinary tract infection (CAUTI) *CMS 01/01/12 – ICU only • Clostridium difficile HAI Data Comparison • NHSN: – SSI comparison to other reporting facilities within the United States is with a Standard Infection Ration (SIR): • The SIR adjusts for patients of varying risk within each facility. • An SIR > 1.0 indicates that more SSIs were observed than predicted and a SIR < 1.0 indicates that fewer SSIs were observed than predicted. • New York State Department of Health – Report using upper and lower confidence levels and the average for the NYSDOH – below, average, and higher than the NYS average. HAI Sample NHSN Data More information can be found at:http://www.cdc.gov/nhsn/PDFs/dataStat/NHSN- Report_2010-Data-Summary.pdf HAI Sample NYSDOH Data More information can be found at: http://www.health.ny.gov/statistics/facilities/hospital/hospita l_acquired_infections HAI Impact • Potentially preventable HAIs cause patient harm: – – – – morbidity mortality increased length of stay Increase health care cost Health System Facilities CLABSI Free Months • Intensive Care Unit (ICU) – – – – – – Glen Cove - >41 months Forest Hills - >6 months Huntington ICU - > 24 months Southside ICU – 9 months Long Island Jewish 2 ICUs - >24 months North Shore University Hospital PICU - >14 months & NSCU - >6 months • Non-ICU – – – – Glen Cove - >18 months Syosset - >22 months Franklin - >6 Months Medical & Adolescent – >24 months CLABSI: 2004 - 2011 ICU CLABSI per 1,000 Central Line Days From September 2005 to December 2008, central line insertion bundle compliance increased from 25% to >80%. 3.5 Change: 2005 through 2008 3 2.5 3.25 3.21 2 2.03 1.5 1.8 1 1.23 1.39 1.45 1.09 0.5 0 2004 2005 2006 2007 2008 2009 2010 2011 Standards of Practice: CLABSI • • • • • • • • • • Central line insertion and dressing kit with chlorhexidine/alcohol Standardized evidence-based central line protocol Antiseptic-impregnated catheters for high risk patients Insertion bundle checklist (skin preparation with chlorhexidine, use of barriers when inserting, site selection, daily assessment) Procedure “STOP” when there is a break in insertion technique Antiseptic dressings/impregnated chlorhexidine disk Needless connectors (neutral pressure) Scrub the hub or alcohol cap Daily chlorhexidine baths Simulation to increase competency 11 Journey Toward Zero – Ongoing Learning LINE MAINTENANCE IV tubing not changed on a timely basis Line in for too long Dressing not change using aseptic techniques IV tubing not labeled properly to change Line not manipulated appropriately Injection hub not disinfected TECHNIQUE NOT ADEQUATE Not compliant with hand hygiene Line inserted w/o sterile technique Inadequate use of maximal barrier precautions Inadequate prep before insertion Femoral line chosen instead of subclavian LACK OF EDUCATION Inexperienced residents and clinicians CLABSI Clinicians not knowledgeable about Central Line Bundle Nurses do not properly know how to change dressings MD does not select a catheter with the least number of lumens Assessment: Identification of patterns or trends Clinicians unaware of line maintenance CLABSI CAUTI Process Change = Outcome Change Baseline* (Feb. 2011 – July 2011) Post-intervention* (Aug. 2011 – Feb. 14, 2012) Southside Hospital – device utilization Syosset Hospital – infection LIJ – infection decrease Plainview Hospital – device utilization Standard of Practice: Indwelling Urinary Catheter • Place indwelling urinary catheters only when indicated: – – – – – Urinary tract obstruction Gross hematuria Neurogenic bladder with retention Urologic surgery or studies Hospice, Comfort or Palliative Care (if patient requests) • When inserted adhere to: – Hand hygiene – Aseptic technique when inserting – Maintain indwelling urinary catheter based on center for Disease Control and Prevention guidelines – Review the need for indwelling urinary catheters daily and remove when no longer needed Joint Project Bundle • • • • Use of an alcohol-containing antiseptic agent for preoperative skin preparation. Preoperative bathing or showering for 3 days prior to surgery with: – 2% CHG impregnated wipe, or – 4% Chlorhexidine Gluconate soap Nasal Staphylococcus aureus screening and use of intranasal Mupirocin for 5 days Surgical Care Improvement Project (SCIP) practices: • Appropriate use of prophylactic antibiotics • • • • • dosing selection timing prior to incision re-dosing based on the facility protocol Appropriate hair removal Potential Avoidance: Case Review • Patient: 67 year-old male • Past Medical History: chronic obstructive pulmonary disease, elevated blood pressure, and osteoarthritis • Surgical History: open reduction and internal fixation (ORIF) for a tibia fracture on 08/25/11 following a motor vehicle accident • Post-operatively: Uncomplicated admission and was discharged home • Readmission Chief Complaint: – On 09/13/11 he had drainage, pain, and increased swelling at the surgical site – The patient was evaluated by the surgeon within the office, sent to the Emergency Department and subsequently admitted Potential Avoidance: Case Review • Hospitalization: Continued – Laboratory: • Surgical wound and blood cultures were positive for methicillin resistant Staphylococcus aureus • Patient remained bacteremic for 8 days – Procedures: • Transesophageal echocardiogram (TEE) negative for endocarditis • Return to the operating room for a wound debridement on 09/13/11 – Antibiotic treatment: Treatment with vancomycin for more than 42 days – Additional management: Return to the operating room for removal of hardware Problem: Hand Hygiene Project Aim: Improved and sustained high hand hygiene compliance 3rd Party Remote Video Auditing • Door motion detector triggers audit • Auditors rate activity based on pass/fail criteria • Video camera records activity • • Digital Video Recorders stores footage locally Audits stored in external auditors database • Feedback delivered via on-site light emitting diode boards, daily e-mails, and weekly e-mails • External auditors connect remotely Timeline: 2008 1 4 02/08 Hand hygiene compliance calculated with the use of remote video auditing and real-time feedback Discussion with staff on the use of Cameras for Hand Hygiene Compliance 03/08 2 04/08 05/08 06/08 07/08 03/08 Cameras and door alarms installed 10/06/08 3 8/08 9/08 10/08 11/08 12/08 01/09 02/09 03/09 04/09 06/10/08 Hand hygiene compliance calculated with the use of remote video auditing 07/04/10 Remote video auditing with feedback continues Hand Hygiene Measurement • Measurement: Hand hygiene with soap and water or an alcohol based hand sanitizer – Pass: hand hygiene observed in a patient room or neighboring area within 10 seconds (before or after) of entry or exit to a patient room – Fail: no hand hygiene observed as per protocol – Discarded events: entries/exits by non-clinical staff or visitor and multiple entries/exits within 60 seconds of another • Quality control audits: 5% of the recorded events to ensure consistency and accuracy Inclusion/Exclusion Criteria • Inclusion: Nurses, aides, house staff, and other clinicians wearing any type of scrub or uniform were classified into the category of other health care professional, and physicians not wearing scrubs were classified as attending physician • Exclusion: Non-clinical workers and visitors Figure Without and With Feedback Internal Self-Auditing Scores Start Feedback 10/06/08 • • Without feedback: hand hygiene rates of <10% (3,833/60,066) With feedback the rates were >86% (223,187/261,091) (p<0.001) Partnership for Patients • Healthcare Association of New York State/Greater New York Hospital Association initiative to decrease: – CLABSI – CAUTI • Goal: – To eliminate and sustain reductions in CLABSI and CAUTIs by >40% by 11/2013. IPRO 10th Scope of Work • Aim: – Prevention, Reduction, Elimination • • • • CLABSI reduction of 50% by 03/13 CAUTI reduction of 25% by 03/13 Clostridium difficile Surgical Site Infections (SSIs) Health Care Personnel Vaccination • Average vaccination rate -~45%. • 20111/2012 vaccinate rate -58%. • Highest vaccination rate was when New York State Department of Health mandated the influenza vaccine in 2009/2010 - 79%. • 2012/2013 plan: 100% program participation: – accept the vaccine or – declining with knowledge regarding placing yourself and others at risk [email protected]