* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slides - Clinical Trial Results
Survey
Document related concepts
Transcript
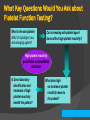
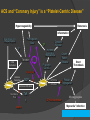
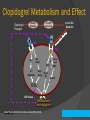
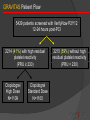
Mohan Sathyamoorthy, M.D., F.A.C.C. Vice-Chief, Cardiovascular Division Baylor All Saints Medical Center, Fort Worth, TX Adjunct Assistant Clinical Professor of Medicine Vanderbilt Medical Center, Nashville, TN Cardiologist, HeartPlace, Fort Worth, TX Learning Objectives Understand the key clinical data related to platelet function testing in clinical cardiovascular medicine Understand the key controversies related to these data Can one incorporate this data into your day to day clinical practice Disclosures Speakers Bureau for: Lantheus Medical Imaging Gilead Biosciences Consultant for: None Active Research Grants: None The Clinical Effectiveness of an Antiplatelet Agent…. A Platelet’s Response to a Drug vs. A Drug’s Bioavailability to a Platelet DRUG PLATELET Platelet Function Testing What Key Questions Would You Ask about Platelet Function Testing? What is the anti-platelet effect of clopidogrel, asa, and emerging agents? Can increasing anti-platelet agent doses affect high platelet reactivity? High platelet reactivity: quantifiable and modifiable risk factor 4) Does laboratory identification and treatment of high platelet reactivity benefit the patient? What does high on-treatment platelet reactivity mean to the patient? ACS and “Coronary Injury” is a “Platelet-Centric Disease” Restenosis Hypercoagulability Inflammation Shear Stress, PCI Plaque Rupture Procoagulant State TF Cytokine Release TF PLT-WBC Aggregation/ Microparticles Growth Factor Release Thrombin Initial Activation P-selectin CD-40L Stent Thrombosis Clopidogrel Granule Secretion ADP Sustained Activation Membrane PL’s X GPIIb/IIIa Activation X TxA 2 Aspirin X GP IIb/IIIa Inhibitor Platelet Aggregation Microaggregates Myocardiall Infarction What Affects a Platelet’s Response to a Drug Platelet Biology Surface Receptor Biology ○ P2Y12 ○ TP (Thromboplastin) ○ VW (VonWillenbrand. i.e. 2b3a) ○ PAR (Protease Activated Receptors) Granule Biology Levels of Circulating Receptor Agonists Central Role of Platelets and Interaction With Coagulation in the Genesis of Thrombosis Fibrinogen Shear, PCI Plaque Rupture Tissue Factor Thrombin Collagen vWF Amplification Initial Activation Amplification P2Y12 ADP TxA2 PAR-1 X Fibrin X TP Granule Secretion + GPIIb/IIIa Activation Activation X Platelet Aggregation X COX-1 Amplification Adapted from Gurbel PA et al. J Am Coll Cardiol. 2007;50:1822-1834. Platelet -Fibrin Clot Formation What Affects A Drug’s “Bio”Availability to A Platelet? Absorption kinetics Gut Pharmacokinetics and dynamics Initial and steady state Concomitant Medications Drug-drug interactions Metabolism, i.e. Pharmacogenetics Liver Cytochromes and SNPs Clopidogrel Metabolism and Effect Clopidogrel Prasugrel CYP P450 Conversion Intestinal Absorption Active Thiol Metabolite X Gq G12 PLC Gi Rho Kinase Ca++ Mobilization Adenylyl Cyclase PI3-K Shape Rap-1b Change Akt cAMP VASP-P Granule Secretion ADP Release GPIIb/IIIa Activation Platelet Aggregation Gurbel PA et al. Nat Clin Pract Cardiovasc Med. 2006;3:387-395. So How Do These Factors Relate to a Clinical Endpoint? Both of these phenomenon ultimately DETERMINE….. VARIABILITY OF RESPONSE TO THERAPY i.e. hyporesponsiveness or “resistance” Antiplatelet Drug “Resistance” / “Response Variability” An Emerging Clinical Problem Healthy Volunteer Crossover Study 100 Interpatient Variability N=66 60 40 20 Interpatient Variability IPA at 24 hours (%) 80 Clopidogrel Responder 0 Clopidogrel Non-responder -20 Response to Clopidogrel 300 mg From Brandt JT AHJ 153: 66e9,2007 Baylor All Saints CV Conference 2010: Advances in CV Therapy, M. Sathyamoorthy, MD, FACC Response to Prasugrel 60 mg Risk of any cardiovascular event in aspirin resistant patients Krasopoulos, G. et al. BMJ 2008;336:195-198 Baylor All Saints CV Conference 2010: Advances in CV Therapy, M. Sathyamoorthy, MD, FACC Risk of any cardiovascular event in aspirin or clopidrogel “resistant” patients Krasopoulos, G. et al. BMJ 2008;336:195-198 Baylor All Saints CV Conference 2010: Advances in CV Therapy, M. Sathyamoorthy, MD, FACC Assessing Platelet Function to Maximize a Therapeutic Response BOTTOM LINE: • Aspirin and clopidrogrel inhibit platelet aggregation but patients exhibit a variable response to both aspirin and clopidrogrel CLINCIAL IMPLICATION: • Maximum anti-platelet effects of aspirin and clopidrogrel may not be obtained with standard dosage • Up to 30% of patients may be resistant to the effects of these drugs • Patient assessment may be required prior to initiation of procedures where clopidrogrel is given • Pre-surgical testing of platelet function may assess the risk of bleeding Adapted from H. Aboul-Enein, Benha Faculty of Medicine So how do we assess “Responsiveness” in 2011 We test PLATELET FUNCTION We should now consider PHARMACOGENETIC TESTING i.e. liver cytochrome genotypes PLATELET FUNCTION TESTING in 2011 Test Advantages Disadvantages ASA Thienopyridines 2B3A Turbidometric PA (PFA-100) “gold Standard” Reproducibility Expense Time Sample Yes Yes Yes Impedance PA (MEA) Whole blood Reproducibility Expense Time Sample Yes Yes Yes VerifyNow Simple POC Whole Blood No instrument adjustment Yes Yes Yes Plateletworks Whole Blood Not well studied Yes Yes Yes TXAB2 COX-1 Indirect Yes No No Urinary 2,3DHTb2 COX-1 Indirect Renal fcn Yes No No Clin Cardiol (Suppl. 1) 31.3.I-10-1-16 (2008) E Braunwald et al: Assessing the current role of platelet function testing Baseline Platelet Reactivity* Determines Outcomes Following Coronary Stenting 1.0 Low Reactivity Group 0.9 0.8 0.7 High Reactivity Group 0.6 P = 0.01 0.5 0 100 Kabbani SS et al. Am J Cardiol. 2003;91:876-878. P = 0.006 200 Time (Days) P = 0.043 300 * Fibrinogen binding in response to 0.2 M ADP ** Composite MI, UR, Revascularization Studies Linking High Ex-Vivo Platelet Reactivity to ADP and Ischemic Events Study 1. Barragan et al. (Catheter Cardiovasc Interv. 2003;59:295) 2. Ajzenberg et al. (J Am Coll Cardiol. 2005;45:1753) 3. Gurbel et al. (CREST Study) (J Am Coll Cardiol. 2005;46:1827) 4. Buonamici (J Am Coll Cardiol. 2007;49:2312) 5. Matzesky et al. (Circulation. 2005;109:3171) 6. Gurbel et al. (Circulation. 2005;111:1153) (J Am Coll Cardiol;2006;48:2186) 7. Bliden et al. (J Am Coll Cardiol. 2007;49:657) 8. Cuisset et al. (J Thromb Haemost. 2006;3:542) 9. Lev et al. (J Am Coll Cardiol. 2006;47:27) 10. Cuisset et al. (J Am Coll Cardiol. 2006;48:1339) 11. Hochholzer et al. (J Am Coll Cardiol. 2006;48:1742) Results Clinical Relevance VASP-P levels Stent thrombosis Shear-induced aggregation Stent thrombosis VASP-P levels ADP-LTA Stimulated GP IIb/IIIa expression Stent thrombosis ADP-LTA Stent thrombosis ∆ ADP-LTA Recurrent cardiac events ADP-LTA Myonecrosis and inflammation pre-PCI ADP-LTA on chronic clopidogrel 1 year post-PCI events ADP-LTA 30-day post-PCI events ADP and AA-LTA Post-PCI myonecrosis ADP-LTA 30-day post-PCI events 600 mg – less events ADP-LTA 30-day MACE Prognostic significance of high on-clopidogrel platelet reactivity after percutaneous coronary intervention: systematic review and meta-analysis. METHODS CONCLUSIONS: • Systematic review and meta-analysis on the clinical relevance of HPR , observation studies 2003-2010 Outcome measures : cardiovascular (CV) death, definite/probable stent thrombosis (ST), nonfatal myocardial infarction (MI), and a composite end point of reported ischemic events. According to the meta-analysis, patients with HPR had a 3.4-fold higher risk for CV death compared with patients with normal ADP reactivity (odds ratio 3.35, 95% CI 2.39-4.70, P < .00001). High on-clopidogrel platelet reactivity, measured by an ADP-specific platelet function assay, is a strong predictor of CV death, MI, and ST in patients after percutaneous coronary intervention. • RESULTS • Twenty studies , 9,187 patients High on-clopidogrel platelet reactivity • MI: 3.00 [2.26-3.99, STEMI: 4.14 [2.74-6.25] Composite: 4.95 [3.34-7.34] P < .00001 for all cases ISSUES Methodology Definition of HPR between studies Meta-analysis/Review Aradi D , et al Am Heart J. 2010 Sep;160(3):543-51. VerifyNow Assay • Whole blood, closed-tube sampling with no pipetting required • Assay results in less than 5 minutes (assay time) • Good correlation with LTA and VASP Mixing Chamber Agonists: Aspirin Assay – AA P2Y12 assay – ADP + PGE1 GpIIbIIIa assay – iso-TRAP Light Source Agonist + Platelets in whole blood maximally activated by agonist in mixing chamber Fibrinogen-coated beads Agglutinated beads aggregate in clusters VerifyNow Point-of-Care Platelet Function Assay* Correlates with Clinical Outcomes Post-DES Deployment 1.00 99.0% Event-free Survival 0.95 P=0.004 91.5% 0.90 0.85 Not high platelet reactivity High platelet reactivity 0.80 0 *pre-discharge Price et al. Eur Heart J. 2008;29:992. 50 100 Time (days) 150 200 DES Thrombosis and Residual Platelet Reactivity Percent of patients free from definite or probable ST (%) 100 98 98 1 96 94 92 Responders Nonresponders* 90 88 91 3 86 84 Log rank P<0.001 82 80 0 30 60 90 120 150 180 Time(Days) (days) Time Buonamici P et al. J Am Coll Cardiol. 2007;49:2312-2317. *Residual platelet aggregation (10 µM ADP) ≥70% OPTIMUS (Optimizing Antiplatelet Therapy in Diabetes Mellitus) Impact of high clopidogrel maintenance dosing on platelet function in DM patients with suboptimal clopidogrel response VerifyNow P2Y12 substudy PRU %IPA 300 p=0.009 P2Y12 Reactivity Units Platelet inhibition (%) 80 60 40 20 p=0.007 250 200 150 100 50 0 0 75 mg 150 mg Angiolillo DJ et al. Circulation. 2007;115:708-16. Angiolillo DJ et al. Am J Cardiol. 2008;101:440-5. 75 mg 150 mg Primary Results of The Gauging Responsiveness with A VerifyNow Assay - Impact on Thrombosis And Safety Trial GRAVITAS AHA 2010 Matthew J. Price, MD On behalf of the GRAVITAS Investigators Point-of-Care Platelet Function Testing: Current Status • At least 7 studies involving more than 3,000 patients have concluded that high residual (on-clopidogrel) platelet reactivity measured by the VerifyNow P2Y12 test is associated with poor clinical outcomes after PCI. • A treatment strategy for patients with high residual platelet reactivity has not been tested in a large, randomized, clinical trial. GRAVITAS: Primary Hypothesis • High-dose clopidogrel for 6 months is superior to standard-dose clopidogrel for the prevention of adverse CV events after PCI in patients with high residual reactivity. GRAVITAS Study Design Elective or Urgent PCI with DES* VerifyNow P2Y12 Test 12-24 hours post-PCI PRU ≥ 230 R High-Dose Clopidogrel† clopidogrel 600-mg, then clopidogrel 150-mg daily X 6 months Standard-Dose Clopidogrel† clopidogrel 75-mg daily X 6 months Primary Efficacy Endpoint: CV Death, Non-Fatal MI, Stent Thrombosis at 6 mo Key Safety Endpoint: GUSTO Moderate or Severe Bleeding at 6 mo Pharmacodynamics: Repeat VerifyNow P2Y12 at 1 and 6 months *Peri-PCI clopidogrel per protocol-mandated criteria to ensure steady-state at 12-24 hrs †placebo-controlled All patients received aspirin (81-162mg daily) Inclusion and Exclusion Criteria Major Inclusion Criteria •DES for the treatment of stable or unstable CAD* Major Exclusion Criteria • Bleeding event or other major complication prior to platelet function testing • Recent glycoprotein IIb/IIIa inhibitor * STEMI pts were permitted after a protocol modification during the trial GRAVITAS Patient Flow 5429 patients screened with VerifyNow P2Y12 12-24 hours post-PCI 2214 (41%) with high residual platelet reactivity (PRU ≥ 230) Clopidogrel High Dose N=1109 Clopidogrel Standard Dose N=1105 3215 (59%) without high residual platelet reactivity (PRU < 230) Primary Endpoint: CV Death, MI, Stent Thrombosis Observed event rates are listed; P value by log rank test. Bleeding Events: Safety Population Severe or life-threatening: Fatal bleeding, intracranial hemorrhage, or bleeding that causes hemodynamic compromise requiring blood or fluid replacement, inotropic support, or surgical intervention Moderate: Bleeding that leads to transfusion but does not meet criteria for severe bleeding P by log rank test; observed event rates listed. HD, high-dose; SD, standard dose Secondary Comparison: High vs. Not High Reactivity Treated with Clopidogrel 75-mg daily Observed event rates are listed. P value by log-rank test. CV Events and Post-PCI PRU In Patients With High and Not High Reactivity Treated With Clopidogrel 75-mg Daily 500 Red dots: patients with CV death, MI, or ST 400 PRU 300 12 - 24 hrs post-PCI 230 PRU 200 100 0 N=1105 High Residual Reactivity ITT population N= 586 Not High Residual Reactivity GRAVITAS: Summary • Compared with standard-dose therapy, high-dose clopidogrel achieved a modest pharmacodynamic effect in patients with high residual reactivity. • In patients with high residual reactivity measured after PCI, 6-months of high-dose clopidogrel did not reduce the rate of cardiovascular death, nonfatal MI, or stent thrombosis and did not increase GUSTO severe or moderate bleeding. GRAVITAS: Possible Explanations • Underpowered: patients low-risk, low event rates? • Given HR of 1.01 with more than 2,200 patients, unlikely that a larger trial would show a clinically meaningful benefit • Pharmacodynamic effect of the intervention was too weak? • Stronger intervention and/or goal-directed therapy with serial measurements merit study (TRIGGER-PCI; ARCTIC; TARGET-PCI) GRAVITAS does not support a treatment strategy of high-dose clopidogrel in patients with high residual reactivity identified by a single platelet function test after PCI. TRIGGER-PCI Courtesy of F.J. Neumann Successful PCI with DES without major complication and NO GPIIb/IIIa use Post-PCI VerifyNow P2Y12 Assay (PRU) 2 - 4 hours after 1st MD of clopidogrel 75 mg at day 1 post-PCI N ~ 8800 Non-Responder Yes PRU ≥ 208? No Responder PRU ≥ 140? Random Selection A N = 1075 B N = 1075 C N = 550 D E N = 550 “Prasugrel arm” “Clopidogrel arm” “Prasugrel arm” “Clopidogrel arm” Prasugrel 60 mg LD Prasugrel 10 mg MD + Clopidogrel placebo Placebo LD Clopidogrel 75 mg MD + Prasugrel placebo Prasugrel 60 mg LD Prasugrel 10 mg MD + Clopidogrel placebo Placebo LD Clopidogrel 75 mg MD + Prasugrel placebo “Standard Therapy” Clopidogrel 75 mg Platelet function substudy: VerifyNow Assessment at day 2 (2 – 4 h after 1st MD of study drug) Clinical Follow-up and VerifyNow Assessment at 90 days, 180 days Primary Endpoint: 6 month CV Death and MI ASCET (PFA-100) AHA SS 11/2010 Primary Objective: Clinical outcome (composite USA+MI+CVA+Death) over 2 years in patients with “stable” symptomatic CAD on ASA therapy as related to initial aspirin response via PFA-100 Secondary Objective: Investigate the difference in composite event rate between patients treated with ASA vs Clopidrogel ASCET (PFA-100) Results AHA SS 11/2010 Conclusions Composite Endpoint No difference in when ASA no RPR (n=373) 10.5% vs ASA high RPR (n=130) 13.1% p=0.44 Composite Endpoint with RPR on ASA when treated with ASA 13.1% (n=130) or Clopidrogel 7.8% (n=129) p=0.16 26% of patients had RPR while on ASA by PFA-100 composite endpoints RPR by PFA-100 did NOT predict clinical outcome Trend seems to indicate fewer endpoints using clopidrogel in patients with high RPR vs. ASA ASCET STUDY DESIGN FLOWCHART ASA 160 mg QD N=503 Angiographic CAD w or w/o PCI N=1001 Composite endpoint of USA+MI+CVA +Death ASA 160 mg/d x 7 days then PFA-100 Clopidrogel 75mg QD N=503 ASA compliance was measured by TXAB2 levels Platelet reactivity after clopidrogrel treatment assessed with point-ofcare analysis (MEA) and early drug-eluting stent thrombosis. HYPOTHESIS: Does platelet reactivity to clopidogrel assessed with multiple electrode platelet aggregometry (MEA) correlate with the risk of early drug-eluting stent thrombosis? METHODS: 2/07-2/08, 1608 patients Before PCI, all patients received 600 mg clopidogrel. The ADP-induced platelet aggregation was assessed in whole blood with a MEA. The primary end point was definite ST at 30 days. CONCLUSIONS: RESULTS: Compared with normal responders (n = 1,285), low responders (MEA upper qunitile, n = 323) had a significantly higher risk of: definite ST within 30 days (2.2% vs. 0.2%; odds ratio [OR]: 9.4; 95% confidence interval [CI]: 3.1 to 28.4; p < 0.0001) Mortality rates were 1.2% in low versus 0.4% in normal responders (OR: 3.2; 95% CI: 0.9 to 11.1; p = 0.07). Composite of death or ST was higher in low versus normal responders (3.1% vs. 0.6%; OR: 5.1; 95% CI: 2.2 to 11.6; p < 0.001). Low response to clopidogrel assessed with MEA is significantly associated with an increased risk of ST. Further studies are warranted to evaluate the ability of MEA to guide antiplatelet therapy in patients undergoing PCI. Sibbing D et al, J Am Coll Cardiol. 2009 Mar 10;53(10):849-56. Clopidogrel response status assessed with Multi-plate point-of-care analysis (MEA) and the incidence and timing of stent thrombosis over six months following coronary stenting. 6 Month followup data from Sibbing D et al, J Am Coll Cardiol. 2009 Mar 10;53(10):849-56. Cumulative incidence of definite ST within six months was significantly higher in low responders [2.5% vs. 0.4%; OR 6.5; 95% CI, 2.4-17.0; P<0.001]. The combined incidence of definite or probable ST was higher as well in low vs. normal responders [4.1% vs. 0.7%; OR 5.8; 95% CI, 2.8-12.3; P<0.0001]. A significant inverse correlation of MEA values and the timing of definite or probable ST (in days) was observed (Spearman coefficient = -0.45; P=0.04) CONCLUSION MEA measurements are highly predictive for the occurrence of ST during the first six months following coronary stenting. In the majority of clopidogrel low responders suffering ST, the ischemic event occurs early in the course after the procedure. Sibbing D, , et al Thromb Haemost. 2010 Jan;103(1):151-9. Epub 2009 Oct 26. VASP Guided PCI After a 600mg clopidogrel LD, poor responders (PRI ≥ 50%) received additional 600mg bolus (max 2400 mg) until reaching therapeutic target. Mean ±SD VASP after first LD, % VASP after adjustment, % Control VASP-guided p 68 ±11 69 ±10 0.4 38 ±14* *<0.001 Log rank p =0.007 MACE: CV death, MI, revascularization Courtesy of Angiolillo Bonello et al. J Am Coll Cardiol 2008 VASP and LTA: limitations Not-user friendly Time consuming Require experienced lab personnel Require expensive equipment Not universally available Overall,….expensive Courtesy of Angiolillo Mechanism of Clopidogrel Response Variability Limited absorption capacity with ceiling effect at 600mg loading dose Clopidogrel Bisulfate Intestinal Absorption ? P-glycoprotein (MDR1 3435T genotype)2 Esterases 85% Inactive Carboxylic Acid Metabolite 15% Hepatic P450 Cytochromes CYP3A4 lipophilic statins CYP3A5 Genetic polymorphisms Genetic polymorphisms CYP2C19 CYP1A2 Smoking, proton pump inhibitors CYP2B6, 2C19 Multistep Conversion Active Thiol Metabolite P2Y12 Receptor Inhibition of Platelet Aggregation (Wide Response Variability)1 ~30% 75mg/day for 30days Post-PCI ~30%-40% 75mg/day for 5-7days volunteers ~30%-40% 300 mg load Post-PCI ~30%-50% 600 mg load Post-PCI ~1.4x 150mg/d vs. 75mg/d for 30days Post-PCI3 1. Gurbel PA et al. Thromb Res. 2007;120:311-21. 2. Taubert et al. Clin Pharmacol. 2006;80:486-501. 3. von Beckerth et al. Eur Heart J.2007;28:1814-9. Residual Platelet Aggregation Stratified by CYP2C19*2 Genotype 60 CYP2C19 *1/*1 CYP2C19 *1/*2 CYP2C19 *2/*2 40 20 0 Baseline Trenk D et al. J Am Coll Cardiol. 2008:51:1925-1934. Before PCI Pre-discharge Clopidogrel Non-Responsiveness Correlation With CYP3A4 Enzyme Activity Nonresponders (25%) 80 9 5.0 Responders (75%) 2.7 1.0 60 1.9 0.7 40 2.0 20 0 3.0 1.0 Platelet aggregation 4 hours post clopidogrel* *450 mg PO (P=0.0002); **P=0.15 Lau WC et al. J Am Coll Cardiol. 2003;41:225A. CYP3A4** activity 0 exhaled/h (%) 37 20 4.0 2 80 14CO Aggregation (%) 100 Pharmacogenetics of antiplatelet therapy CYP2C19 and CVD, MI, or Stroke PRASUGREL CLOPIDOGREL 14 14 Carriers CYP2C19 Reduced-Function Allele Carriers 13 12.1 12 12 11 11 10 9 8.0 Non-carriers 8 7 6 5 4 Hazard Ratio 1.53 3 0 30 90 Carriers 8.5 CYP2C19 Reduced-Function Allele Carriers 7 6 5 4 2 Hazard Ratio 0.89 (95% CI 0.60-1.31) 360 270 180 Days After Randomization 450 P=0.27 N=1,466 1 Number at Risk: Non-Carrier Carrier 8 P=0.014 0 9.8 9 3 N=1,477 Non-carriers 10 (95% CI 1.07-2.19) 2 1 CV Death, MI, or Stroke (%) CV Death, MI, or Stroke (%) 13 0 0 30 90 1048 991 407 383 982 376 180 270 360 Days After Randomization 450 Number at Risk: 1064 1009 999 980 870 755 542 395 364 360 348 306 270 181 * Carriers ~30% of the population Non-Carrier Carrier Courtesy of Angiolillo 951 364 849 320 750 276 541 188 Mega et al. NEJM 2009 Strategies to Improve/Overcome Clopidogrel Response Increase clopidogrel dose (What data do we believe?? GRAVITAS implies NO!) Prasugrel! New pharmacologic agents coming….(ticagrelor, AZD6140, cangrelor) Is there a role for CYP inducers (St. John’s Wort, etc.) to enhance conversion? Triple antiplatelet regimens (cilostazol, dipirydamole)? Learning Objectives Understand the key clinical data related to platelet function testing in guiding clinical decision making OPTIMUS GRAVITAS TRIGGER-PCI ASCET MEA Studies (JACC 2010, TH 2010) Mega et al, NJEM 2009 Learning Objectives Understand the key controversies related to these data… Is there really a standard definition of “responsiveness or resistance” Cut points matter, and we fundamentally DO NOT have a consensus What is the best test? Do we combine platelet function testing and Genotype testing in a POC method? Learning Objectives Can one incorporate this data into your day to day clinical practice and do we have GUIDELINES supporting this? ISTH Consensus Statement: Do not test ASA or clopidogrel resistance outside of clinical trials or to change therapy based on such testing. Why? NO clinically meaningful, standardized definition of resistance based on data linking therapy-dependent laboratory tests to clinical outcomes . Correct treatment of patients whose platelets are hyporesponsive to antiplatelet agents is unknown. Guidelines Continued AHA/ACC/ASCI: Daily ASA therapy after PCI for patients without ASA ‘‘resistance,’’ but no definition of resistance is offered. Class IIb, level C recommendation For clopidogrel, the guidelines state that ‘‘ . . .in patients in whom stent thrombosis may be catastrophic or lethal. . . Platelet aggregation studies may be considered and the dose of clopidogrel increased to 150 mg per day if less than 50% inhibition of platelet aggregation is demonstrated.’’ What about GRAVITAS??? Method to assess platelet inhibition is not described. HOW DO WE MOVE FORWARD? We need adequately powered clinical trials to…. 1. Determine what is the most simple, inexpensive, and rapid test of platelet function and/or metabolism genotyping that best predicts clinical outcomes of antiplatelet therapy for specific patient subgroups? AND STICK TO IT….results in trials discussed seem to vary by assay! 2. Determine if individual outcomes are affected when treatment is changed in response to the testing results. i.e. what is our “threshold” or “cutoff” values 3. What the benefit of supplementary treatment with an agent having a different mechanism of action in patients with known hypo-response to a given antiplatelet agent? 4. With emerging agents, is any of this ultimately going to matter? Adapted from H. Aboul-Enein, Benha Faculty of Medicine