* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download March2012--CassandraSalgado
Survey
Document related concepts
Transcript
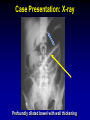
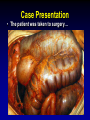
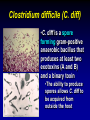
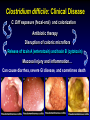
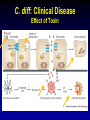
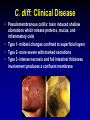
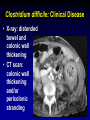
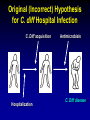
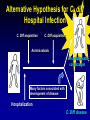
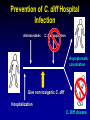
Clostridium Difficile: Epidemiology and Clinical Spectrum Cassandra D Salgado, MD, MS Associate Professor of Medicine Hospital Epidemiologist, MUSC March 15, 2012 I have no Disclosures to report relevant to the content of this presentation. Cassandra D Salgado, MD, MS Associate Professor of Medicine Hospital Epidemiologist, MUSC March 15, 2012 Case Presentation • 50 year old female was admitted for COPD exacerbation. She was treated with steroids, inhalers, and antibiotics. • On hospital day 6 she develops fever of 102°F hypotension, diarrhea, and increased WBC count with profound bandemia (WBC= 48,000 with 30% bands). • Septic workup was done… Case Presentation • Patient continued to decompensate with continued fevers and hypotension requiring vasopressors and transfer to MICU • Bowel movements ceased on hospital day 8 and patient was evaluated for toxic megacolon… Case Presentation: X-ray Profoundly dilated bowel with wall thickening Case Presentation • The patient was taken to surgery… Case Presentation • Colectomy was performed • Patient died on hospital day 9: autopsy revealed severe disease and pseudomembranous colitis Clostridium difficile (C. diff) •C. diff is a spore forming gram-positive anaerobic bacillus that produces at least two exotoxins (A and B) and a binary toxin •The ability to produce spores allows C. diff to be acquired from outside the host C. difficile: Epidemiology • “difficult” to isolate in laboratory • 1978: cause of pseudomembranous colitis • Most commonly recognized microbial cause of hospital-acquired diarrhea • 3-5% of healthy adults are colonized • 20-40% of hospitalized patients are colonized • Asymptomatic carriers outnumber patients with disease by several fold Bartlett JG. NEJM 1978;298:531. Viscidi R. Gastroenterol 1981;81:5. McFarland LV. NEJM 1989;320:204. Clostridium Difficile: Risk Factors • Exposure to antibiotics the preeminent risk factor for developing disease • More than 90% of healthcare-associated CDI occur while on antibiotics – Agents active against anaerobic organisms are considered to present the greatest risk (clindamycin) – Most cases are associated with β-lactam therapy – Growing number of reports of associations with fluoroquinolone therapy – Duration of antecedent therapy may be brief (surgical prophylaxis) C. Diff and Antibiotic Use • Broad-spectrum agents with a greater effect on the normal intestinal flora have been associated with CDAD • Meta-analysis: 49 studies Pooled Odds Ratio 95%CI Penicillin 2.0 1.1-4.5 1st Gen Ceph 2.6 1.8-3.7 Quinolones 8.0 4.5-14.3 Clindamycin 9.0 6.3-12.9 Antipseudo PCN 9.6 4.2-21.8 Cefoxitin 10.6 3.0-37.5 Ceftazadime 28.8 12.7-65.1 Cefotaxime 36.2 19.0-68.9 Bignardi GE. J Hops Infect 1998;40:1. Clostridium Difficile: Risk Factors • • • • • • • • • Cancer with or without chemotherapy GI surgery and other types of GI manipulation Older age Severity of illness Immunocompromising conditions Longer duration of stay in hospital ICU stay Exposure to an infected roommate Anti-ulcer medications Bignardi GE. J Hops Infect 1998;40:1. C. diff: Special Populations • Most cases of CDI occur in healthcare • Among hospitalized patients – Medical patients are at increased risk compared to surgical patients • C. diff is the most common cause of acute diarrheal illness in LTCF – Population is older, receive more medications known to increase risk of C. diff • Neonates may also be colonized with C. diff – Up to 70% – Neonates colonized with toxogenic strains less likely than adults to develop symptomatic disease • Neonates may lack receptors for toxin A in their immature enterocytes Sunenshine R. Clev Clinic J Med 2006;73:187. Clostridium difficile: Clinical Disease C. Diff exposure (fecal-oral) and colonization Antibiotic therapy Disruption of colonic microflora Release of toxin A (enterotoxin) and toxin B (cytotoxin) Mucosal injury and inflammation… Can cause diarrhea, severe GI disease, and sometimes death Clostridium difficile: Clinical Disease • • • • • Watery diarrhea (10-15 times per day) Lower abdominal cramping Low grade fever Leukocytosis (WBC>15,000) Symptoms generally occur during antibiotic therapy • Sometimes 5-10 days after completion • Rarely weeks after completion • Physical exam: abdominal tenderness CDI vs. AAD C. diff: Clinical Disease Effect of Toxin C. diff: Clinical Disease • Pseudomembranous colitis: toxin induced shallow ulcerations which release proteins, mucus, and inflammatory cells • Type 1- mildest changes confined to superficial layers • Type 2- more severe with marked secretions • Type 3- intense necrosis and full intestinal thickness involvement produces a confluent membrane C. diff: Clinical Disease • Fulminant colitis (rare) • Severe abdominal pain, abdominal distention, diarrhea, fever, hypovolemia, lactic acidosis. Marked leukocytosis (WBC>40,000) • Toxic Megacolon (rare) • Severe abdominal pain, abdominal distention, scant to no diarrhea, fever, shock, marked leokocytosis Clostridium difficile: Clinical Disease • X-ray: distended bowel and colonic wall thickening • CT scan: colonic wall thickening and/or pericolonic stranding CDI: Diagnosis • Cytotoxicity assay (tissue culture assay) – Gold standard for diagnosis – Add prepared stool to culture cells • If toxin present will cause histopathologic changes • Enzyme immunoassay (EIA) – – – Direct detection of toxin A or B Dependent on toxin level “three samples” rule • Anaerobic Culture – Often combined with other diagnostic methods • Endoscopy – Helpful for atypical presentations CDI: Diagnosis Test Time Sensitivity Advantages Disadvantages Endoscopy 2 hrs 51% Anaerobic culture 72 hrs 89-100% Tissue cytotoxin assay 48 hrs 94-100% ELISA toxin A and B 2 hrs Diagnostic of p colitis Low sensitivity Molecular typing No distinction of toxin production Detects A (most sensitive) and B 80-95% Easy to use False positives Decreased sensitivity for low level toxin Decreases PPV and limits clinical management Sunenshine R. Clev Clinic J Med 2006;73:187. Planche T. Lancet ID 2008;8:7772008;8:777-784 CDI: Diagnosis • PCR Based Testing – Directed towards toxin B or toxin A and B – Sensitivity and specificity 97-100% – No “three samples” – Compares well with toxinogenic culture – Considered by some to be the best clinical diagnostic tool Clostridium Difficile: At What Cost? • Estimated 3,000,000 new cases of C. diff diarrhea and colitis in US hospitals annually – Affects as many as 10% of patients hospitalized for more than 2 days – Relapsing disease occurs in 20-25% of adequately treated patients • Further relapses occur in 65% of patients who have suffered more than one relapse Clostridium Difficile: At What Cost? Study Mortality LOS (excess) Costs Miller MA. 2002 1.5% 13.6 days $85,000 per yr Kyne L. 2002 NR 10.2 (3.6) days $3,669 per pt McFarland LV. 1999 0% NR $10,970 per pt Wilcox MH. 1996 NR 46.5 (21.3) days £4,000 per pt Riley TV. 1995 NR 24.5 (18) days *$1,250,000 per yr Macgowan A. 1995 NR 49.8 (30.2) days NR Olson MM. 1994 0.6% NR NR Kofsky P. 1991 NR NR $2,000 to $5,000 per pt *Australian dollars • Clostridium Difficile – Spore forming toxin producing bacillus responsible for most nosocomial diarrhea. Risks for CDI include antibiotic exposure, other drugs, older age, and co-morbidities. CDI has been associated with notable morbidity, but traditionally low rates of severe disease and mortality. C. diff: New Epidemiologic Issues • C. diff rates are increasing • More severe disease with higher mortality and higher rates of colectomy • A common epidemic strain has been found in North America and Europe – Fluoroquinolone use may be driving its emergence CDI is Increasing • Average rate of CDI among NNIS hospitals from 1987-1998 – 12.2 cases per 10,000 pt-days • Teaching hospitals: 13.0 per 10,000 pt days • Non-teaching hospitals: 11.4 per 10,000 pt days • CDC reports that hospitalizations with a discharge diagnosis of CDI – Was 31 per 100,000 in 1996 – Was 61 per 100,000 in 2003 www.CDC.gov CDI is Increasing CDI is Increasing C. diff-Associated Infection Recent data: Incidence of healthcare-associated C. diff colonization and infection are 29.5 and 28.1 cases per 10,000 pt days, respectively. Incidence of CDI per 10,000 patient days NNIS 1987-2001 Archibald et al JID 2004;189:1585 C. diff: Increased Morbidity and Mortality • A hospital in Pittsburgh reports 253 nosocomial CDI cases in 2 years with an increase from 2.7 to 6.8 cases per 1,000 discharges (p<0.001)1 – 26 (10%) colectomies – 18 (7%) mortality • A Chicago hospital reports increased severity of CDI and increased mortality in MICU and oncology patients2 – high rates of shock – 37% mortality 1Muto et al. ICHE 2005;26:273., 2Patel et al. 2005 SHEA Annual Meeting #285. C. diff: Increased Morbidity and Mortality • Prospective study of 1719 CDI episodes in 12 Quebec hospitals – Determine incidence of CDI and complications • 22.5 per 1,000 admissions – Incidence increased with age • 30-day attributable mortality was 6.9% – Attributable mortality increased with age • 110 (6.5%) required ICU care • 33 (1.9%) required colectomies Loo VG et al. NEJM 2005;353(23):2442. C. diff: Pathogenicity • In general, C. diff strains either possess the entire genetic element (PaLoc) responsible for producing toxin (toxigenic strains) or lack this genetic element and thus, do not cause disease (non-toxigenic strains) C. diff: Pathogenicity • Mutation(s) in the tcd C gene can cause the C. diff strain to produce increased amounts of toxin causing more severe disease. – 16 x more toxin A and 23 x more toxin B • A binary toxin may also contribute, but clinical significance is not yet known. • This “epidemic strain” is PCR ribotype 027, PFGE type NAP1, and restriction endonuclease type B1. States with BI/NAP1/027 strain of C. difficile Recent data: among patients colonized or infected with C. diff, 36% and 63% have the NAP1 strain, respectively DC HI AK PR CDI: Risk Factors • Pittsburgh hospital outbreak (MVA) OR 1.02 95%CI 1.01-1.04 Age (per year) Diabetes 2.1 1.2-3.6 Organ Transplant 5.8 2.3-14.6 H2 blocker 2.0 1.1-3.5 PPI 2.4 1.3-4.4 Clindamycin 4.8 1.9-12.0 Ceftriaxone 5.4 1.8-15.8 *Levofloxacin 2.0 1.2-3.3 *Levofloxacin use significantly increased over the preceding months leading up to the outbreak Muto et al. ICHE 2005;26:273. CDI: Risk Factors • Quebec hospitals Outbreak – Fluoroquinolone receipt OR 3.9 (2.3-6.6) • All fluoroquinolones studies were independently associated with CDI – Cephalosporin receipt OR 3.8 (2.2-6.6) • All cephalosporins studies were independently associated with CDI – PPI, H2 blockers, or chemotherapy were not significantly associated with CDI – All isolates with tcdC gene mutations also had the binary toxin gene • Severe CDI was observed in 22 of 132 patients with both genetic elements • Severe CDI was not observed in any pt that did not have these genetic elements (p=0.03) Loo VG et al. NEJM 2005;353(23):2442. CDI: An Antibiotic-Resistance Issue? • Pittsburgh hospital outbreak – No isolates R to metronidazole or vancomycin – 85.7% R to clindamycin • Clindamycin was a significant risk – 94.5% R to levofloxacin • Levofloxacin was a significant risk • Quebec hospitals Outbreak – No isolates R to metronidazole, vancomycin or clindamycin • Clindamycin was not a significant risk – 100% of predominate type isolates R to ciprofloxacin, moxifloxacin, gatifloxacin, and levofloxacin • All FQ were associated with increased risk Muto et al. ICHE 2005;26:273., Loo VG et al. NEJM 2005;353(23):2442. CDI and Fluoroquinolones • Characterization of 187 C. diff isolates from 8 healthcare facilities from 2001-2003 and comparison to historic isolates – An epidemic strain was identified and was responsible for 50% or more of the isolates from most facilities (binary toxin and tcdC deletions in all) – This epidemic strain was in existence since 1984 – Compared to similar historic strains where no R to FQ was detected, all recent strains were R to gatifloxacin and moxifloxacin – Conclusion: A previously uncommon strain of C. diff with variation in toxin genes has become more R to fluoroquinolones and has emerged as a cause of geographically dispersed outbreaks of CDI McDonald LC et al. NEJM 2005;353(23):2433. C. diff: Community Setting • 31 yo woman 14 weeks pregnant with twins developed 3 weeks of intermittent diarrhea- stool specimens positive for C. diff – Only ABX exposure was TMP-SXT 3 mo prior – Treated but ultimately developed severe disease hospitalized for 18 days – Had recurrent disease 4 days after discharge, spontaneously aborted her fetuses, developed sepsis and died • CDC and Philadelphia Department of Public Health launched investigation C. diff: Community Setting • 33 cases of CA-CDI were reported from NH, PA, NJ, OH, all but 1 occurring in 2004-2005 – 15 (46%) required hospitalization or an ED visit – 13 (39%) had a relapse requiring treatment – 8 (24%) reported no ABX exposure in prior 3 months • 3 of these cases had exposure to patient with “diarrheal illness” (2 confirmed C. diff) – Of those who had received ABX (25 patients) • 3 had received <3 doses, 2 had received only 1 dose • 10 reported exposure to clindamycin – 10 among peripartum women • Transmission to close contacts evident for 4 patients – 23 among non-peripartum individuals • Ages 6 mo to 72 years (mean 26 years) C. diff: Community Setting • The estimated minimum annual incidence of CA-CDI in Philadelphia and its surrounding four counties – 7.6 per 100,000 population – One case for every 5,549 outpatient ABX prescriptions • Twice as high as the <1 case per 10,000 cited in earlier studies C. diff: Community Setting • Conclusions – These cases of severe CDI among individuals previously thought to be at low risk might reflect changing epidemiology – Certain features of CDI that have been uncommon in the past might be changing • • • • Close-contact transmission High recurrence rate Younger age patients Lack of ABX exposure – Because reporting was voluntary, the true incidence of CDI is likely higher • Clostridium Difficile – Emerging issues related to CDI include an increase in overall disease incidence associated with an epidemic strain that contains a mutation for increased toxin production and thus more severe disease with higher risk of death. This strain has an increased rate of resistance to certain antimicrobials, particularly fluoroquinolones, which may be driving the rates. Additionally, the epidemiology of CDI in the community, previously thought to be low risk, may be changing. CDI: Treatment • Stop the inciting antibiotic – Up to 25% will recover without further therapy • Metronidazole vs. Vancomycin – Several older retrospective studies comparing oral metronidazole to oral vancomycin • Metronidazole just as effective – Response rates >95% • Less expensive ($2 day vs. $70 day) • Less risk for VRE emergence and spread – A recent prospective observational study • Response rate to metronidazole was only 78% – Due to recent emergence of newer strain? CDI: Treatment • Metronidazole vs. Vancomycin – Both drugs have good in vitro activity • Median MICs <1.0 – Both drugs promote VRE overgrowth in the stool during treatment – Relapse rates after treatment are about the same for both drugs – Controversy regarding gut levels of metronidazole • Only present with active disease? Wafa N. AAC 2008;52(7):2403-2406. CDI: Treatment • Metronidazole vs. Vancomycin Cure by SOI Metronidazole Vancomycin P-value mild 37/41 (90) 39/40 (98) 0.36 severe 29/38 (76) 30/31 (97) 0.02 5/69 (7) 0.27 Relapse Rate 9/66 (14) Zar FA. CID 2007;45:302-307. CDI: Treatment • Metronidazole vs. Vancomycin Cure by SOI Metronidazole Vancomycin P-value mild 26/33 (79) 23/27 (85) NS moderate 40/53 (75) 58/73 (80) NS severe 37/58 (65) 28/33 (85) <0.05 27/103 (23) NS Relapse Rate 29/100 (29) Louie T. ICAAC 2007. Abstract K-4259. CDI: Treatment • Metronidazole vs. Vancomycin Metronidazole Vancomycin P-value LOS (days) 12.8 11.5 <0.001 Mortality 7.9% 6.8% 0.02 ICU stay 23.2 17.7 <0.001 Drug cost $2439 $2492 NS $14718 <0.001 Hospital cost $16953 Lahue B. ECCMID 2007. Abstract 1732. CDI: Treatment • Appropriate antibiotics directed towards C. diff should be given for 10 days for mild to moderate disease – Metronidazole (PO or IV), Vancomycin (PO) • Most experts recommend starting with metronidazole • Clinicians should be vigilant about monitoring response – If disease does not progress, should not consider treatment failure before 6-7 days – Vancomycin may be more appropriate for severe disease • Intraluminal or oral – Consider surgery if CDI progresses or if severe CDI: Treatment • Nitazoxanide vs. Vancomycin (severe disease) Nitazoxanide Vancomycin P-value EOT response 8/10 (80) 7/10 (70) NS Relapse 1/10 (10) 1/10 (10) NS Sustained response 7/10 (70) 6/10 (60) NS Musher D. CID 2009;48:e41-e46. CDI: Treatment • Fidaxomicin vs. Vancomycin Clinical Response EOT Sustained Response Dificid Vanco Difference (95% CI) Dificid Vanco Difference (95% CI) Trial 1 88% 86% 2.6% (-2.9-8.0) 70% 57% 12.7% (4.4-20.9) Trail 2 88% 87% 1.0% 72% (-4.8-6.8%) 57% 14.8% (5.9-23.3%) Some evidence to suggest that Dificid may be better for non-B1 isolates of C Diff when compared to vancomycin (redcued recurrence). Equivalent for B1 isolates. Package Insert DIFICID 2011. CDI: Treatment • Tolevamer (polymer): phase 3- inferior to vancomycin • Ramoplanin (antibiotic): phase 3 • Rifaximin (antibiotic): effective when used with vancomycin for recurrent disease • Monoclonal antibody: phase 2 • Vaccine: phase 2 CDI: Recurrent Disease • Meta-analysis of 12 studies to identify risks for recurrent disease – Continued use of non-CDI antibiotics • OR 4.23 95%CI 2.10-8.55, p<0.001 – Concomitant receipt of antacid medications • OR 2.15 95%CI 1.13-4.08, p=0.02 – Older age • OR 1.62 95%CI 1.11-2.36, p=001 Garey KW. JHI 2008;70:298-304. CDI: Treatment • No consensus on recurrent disease – Alternative agents, pulsed or tapering vancomycin courses – Ion-exchange resins and polymers – IVIG: some positive results in case reports • Do not treat asymptomatic colonization – Current therapies not effective for this purpose • Vancomycin effects not sustained and may place pts at risk for prolonged carriage after treatment • Can only obtain significant intraluminal levels of metronidazole in presence of diarrhea CDI: Alternative Regimens • Meta-analysis of RR by Type of Probiotic for Prevention of Antibiotic Associated Diarrhea #RCT Saccharomyces 6 Pooled RR 0.37 Lactobaccillus 6 0.31 95%CI, p-value 0.13-0.72, 0.006 0.26-0.52, <0.0001 • Meta-analysis of 6 RCT for Prevention of CDI – RR 0.59, 95%CI 0.41- 0.85, p=0.005 – Only Saccharomyces showed significant reduction for recurrent CDI McFarland LV. Am J Gastro 2006;101:812. CDI: Alternative Regimens • 2 patients died of unrelated causes • 1 patient had recurrent disease (6.3%) • 15 had no recurrent disease at 90 day follow-up C. Diff: Transmission • Two major potential reservoirs of C. diff in hospitals are patients and inanimate objects • Continued pressure from indiscriminate antibiotic use – Failure to stop antibiotics when they are no longer indicated • Patients with symptomatic intestinal infection probably contribute most – Contamination of the environment due to persistence of highly resistant spores • Can persist for many weeks or months after the patient has left the environment – Personnel hand carriage probably accounts for the majority of hospital transmission Original (Incorrect) Hypothesis for C. diff Hospital Infection C. Diff acquisition Hospitalization Antimicrobials C. Diff disease Alternative Hypothesis for C. diff Hospital Infection C. Diff acquisition C. Diff acquisition Antimicrobials Asymptomatic colonization Many factors associated with development of disease Hospitalization C. Diff disease Asymptomatic C. diff Colonization • Data from four prospective studies in which rectal swabs were obtained weekly from hospitalized patients: – Rates of CDI in non-colonized patients was 3.6% compared to 1.3% in colonized patients (p=0.02) – Rate also significantly decreased when only patients who received antibiotics were analyzed (p=0.02) Lancet 1998;351:633-636 Clostridium difficile: Control • Consider two distinct but related approaches – Efforts directed at interrupting horizontal spread (keep patient from acquiring organism) – Efforts to minimize the possibility that exposure will result in infection (reduce individual risk of disease) Control Measures: Prevention of CDI • • • • • • • Follow antimicrobial usage restrictions Avoid electronic thermometers Dedicate patient care items Use full barrier precautions for CDI pts Place CDI pts in private room Perform appropriate hand hygiene Perform adequate environmental decontamination (bleach) • Educate HCWs regarding CDI SHEA/IDSA Practice Recommendations. ICHE 2008;29:Suppl 1. Hand Hygiene and C. diff Risk • Hand washing with soap or chlorhexidine is equally effective in removing spores of C. diff from the seeded hands of volunteers • Alcohol hand hygiene products are not sporicidal and not likely to reduce the burden of spores on hands when used without hand washing • CDI outbreaks have NOT been directly related to use of alcohol hand hygiene products Control Measures: Prevention of CDI • Approaches that should not be considered for routine prevention – Testing of asymptomatic patients – Repeat testing of CDI pts after successful therapy • Unresolved issues – Use of contact precautions by family members – Standing nursing orders for testing – Restricting gastric acid lowering medications SHEA/IDSA Practice Recommendations. ICHE 2008;29:Suppl 1. Prevention of C. diff Hospital Infection Antimicrobials C. Diff acquisition Asymptomatic colonization Give non-toxigenic C. diff Hospitalization C. Diff disease • CDI: Treatment and Control – Effective treatment regimens have been developed for mild to moderate disease which include metronidazole, vancomycin, and fidaxomicin. Severe disease may require alternative uses of traditional drug regimens and possibly adjunctive agents. The transmissibility of C. diff has been well described in healthcare facilities and control measures have been recommended. Novel prevention methods are being considered. Summary • Clostridium difficile remains an important nosocomial pathogen • The epidemiology of Clostridium difficile is changing – Increased rates in acute care and long term care facilities – Increased morbidity and mortality – Possibly due to a genetically altered strain with increased virulence • Effective treatment regimens exist – Study of new treatment regimens should continue • Effective control measures have been described – Study of new or enhanced control measures should continue Questions…