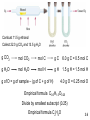* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chapter 3 Stoichiometry: Calculations with Chemical
Size-exclusion chromatography wikipedia , lookup
Chemical reaction wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Strengthening mechanisms of materials wikipedia , lookup
Process chemistry wikipedia , lookup
Biochemistry wikipedia , lookup
Chemical element wikipedia , lookup
Drug discovery wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Transition state theory wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Rate equation wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Metalloprotein wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Chemical bond wikipedia , lookup
History of chemistry wikipedia , lookup
Isotopic labeling wikipedia , lookup
Computational chemistry wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Implicit solvation wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Molecular dynamics wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
History of molecular theory wikipedia , lookup
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and Equations Reaction Types Combination Reactions • Two or more substances react to form one product • Examples: N2 (g) + 3 H2 (g) 2 NH3 (g) C3H6 (g) + Br2 (l) C3H6Br2 (l) 2 Mg (s) + O2 (g) 2 MgO (s) 2 Mg (s) + O2 (g) 2 MgO (s) Link to Video Decomposition Reactions • One substance breaks down into two or more substances • Examples: CaCO3 (s) CaO (s) + CO2 (g) 2 KClO3 (s) 2 KCl (s) + O2 (g) 2 NaN3 (s) 2 Na (s) + 3 N2 (g) Link to Video Combustion Reactions • Rapid reactions that produce a flame • Most often involve hydrocarbons reacting with oxygen in the air • Examples: CH4 (g) + 2 O2 (g) CO2 (g) + 2 H2O (g) C3H8 (g) + 5 O2 (g) 3 CO2 (g) + 4 H2O (g) Balancing Reactions Law of Conservation of Mass “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical experiments depends.” --Antoine Lavoisier, 1789 Dalton’s Postulates Atoms of an element are not changed into atoms of a different element by chemical reactions; atoms are neither created nor destroyed in chemical reactions. They just rearrange! A process in which one or more substances is changed into one or more new substances is a chemical reaction. A chemical equation uses chemical symbols to show what happens during a chemical reaction. reactants products 3.7 How to “Read” Chemical Equations 2 Mg + O2 2 MgO 2 atoms Mg + 1 molecule O2 makes 2 formula units MgO 2 moles Mg + 1 mole O2 makes 2 moles MgO 48.6 grams Mg + 32.0 grams O2 makes 80.6 g MgO IS NOT 2 grams Mg + 1 gram O2 makes 2 g MgO 3.7 Balancing Chemical Equations 1. Write the correct formula(s) for the reactants and the product(s). Ethane reacts with oxygen to form carbon dioxide and water C2H6 + O2 CO2 + H2O 2. Change the numbers in front of the formulas (coefficients) to make the number of atoms of each element the same on both sides of the equation. Do not change the subscripts. 2C2H6 NOT C4H12 3.7 Balancing Chemical Equations 3. Start by balancing those elements that appear in only one reactant and one product. C2H6 + O2 2 carbon on left C2H6 + O2 6 hydrogen on left C2H6 + O2 CO2 + H2O start with C or H but not O 1 carbon on right multiply CO2 by 2 2CO2 + H2O 2 hydrogen on right 2CO2 + 3H2O multiply H2O by 3 3.7 Balancing Chemical Equations 4. Balance those elements that appear in two or more reactants or products. C2H6 + O2 2 oxygen on left 2CO2 + 3H2O multiply O2 by 7 2 4 oxygen + 3 oxygen = 7 oxygen (3x1) on right (2x2) C2H6 + 7 O2 2 2CO2 + 3H2O 2C2H6 + 7O2 4CO2 + 6H2O remove fraction multiply both sides by 2 3.7 Balancing Chemical Equations 5. Check to make sure that you have the same number of each type of atom on both sides of the equation. 2C2H6 + 7O2 4CO2 + 6H2O 12 4 CH (2 6) 14 O(2 (7xx2) 2) 4(6 C 2)6) 1412 OH (4 x 2x + Reactants 4C 12 H 14 O Products 4C 12 H 14 O 3.7 Remember 2 Mg + O2 2 MgO IS NOT 2 grams Mg + 1 gram O2 makes 2 g MgO So How do we relate masses of reactants and products? That’s what we can observe! 3.7 Formula Weights Formula Weight (FW) • Sum of the average atomic weights for the atoms in a chemical formula • So, the formula weight of calcium chloride, CaCl2, would be Ca: 1(40.1 amu) + Cl: 2(35.5 amu) 111.1 amu • These are generally reported for ionic compounds Molecular Weight (MW) • Sum of the average atomic weights of the atoms in a molecule • For the molecule ethane, C2H6, the molecular weight would be C: 2(12.0 amu) + H: 6(1.0 amu) 30.0 amu Percent Composition One can find the percentage of the mass of a compound that comes from each of the elements in the compound by using this equation: (number of atoms)(atomic weight) % element = (FW of the compound) x 100 Percent Composition So the percentage of carbon in ethane is… (2)(12.0 amu) %C = (30.0 amu) 24.0 amu x 100 = 30.0 amu = 80.0% Moles Using Moles Moles provide a bridge from the molecular scale to the real-world scale Avogadro’s Number • 6.02 x 1023 • 1 mole of 12C has a mass of 12 g The mole (mol) is the amount of a substance that contains as many elementary entities as there are atoms in exactly 12.00 grams of 12C 1 mol = NA = 6.0221367 x 1023 Avogadro’s number (NA) 3.2 eggs Molar mass is the mass of 1 mole of shoes in grams marbles atoms 1 mole 12C atoms = 6.022 x 1023 atoms = 12.00 g 1 12C atom = 12.00 amu 1 mole 12C atoms = 12.00 g 12C 1 mole lithium atoms = 6.941 g of Li For any element atomic mass (amu) = molar mass (grams/mol) 3.2 Percent composition revisited n x molar mass of element x 100% molar mass of compound n is the subscript number … Assume one mole of substance and the math goes like this. 2 x (12.01 g) x 100% = 52.14% 46.07 g 6 x (1.008 g) %H = x 100% = 13.13% 46.07 g 1 x (16.00 g) %O = x 100% = 34.73% 46.07 g %C = C2H6O 52.14% + 13.13% + 34.73% = 100.0% 3.5 Question • What is the mass on one atom of carbon 12? • What is the mass in grams of one amu? Mole Relationships • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Do You Understand Molar Mass? How many atoms are in 0.551 g of potassium (K) ? 3.2 Do You Understand Molar Mass? How many atoms are in 0.551 g of potassium (K) ? 1 mol K = 39.10 g K 1 mol K 0.551 g K x 39.10 g K = 0.0141 moles K 3.2 Molecular mass (or molecular weight) is the sum of the atomic masses (in amu) in a molecule. 1S SO2 2O SO2 32.07 amu + 2 x 16.00 amu 64.07 amu For any molecule molecular mass (amu) = molar mass (grams) 1 molecule SO2 = 64.07 amu 1 mole SO2 = 64.07 g SO2 3.3 Do You Understand Molecular Mass? How many H atoms are in 72.5 g of C3H8O ? 3.3 Do You Understand Molecular Mass? How many H atoms are in 72.5 g of C3H8O ? 1 mol C3H8O = (3 x 12.01) + (8 x 1.008) + 16.00 = 60.09 g C3H8O 1 mol C3H8O molecules = 8 mol H atoms 1 mol H = 6.022 x 1023 atoms H 72.5 g C3H8O x 1 mol C3H8O 8 mol H atoms x 60.09 g C3H8O 1 mol C3H8O = 9.65 moles H or 5.82 x 1024 atoms H 3.3 Mass Changes in Chemical Reactions 1. Write balanced chemical equation 2. Convert quantities of known substances into moles 3. Use coefficients in balanced equation to calculate the number of moles of the sought quantity 4. Convert moles of sought quantity into desired units 3.8 Methanol burns in air according to the equation 2CH3OH + 3O2 2CO2 + 4H2O If 209 g of methanol are used up in the combustion, what mass of water is produced? grams CH3OH moles CH3OH molar mass CH3OH 209 g CH3OH x moles H2O grams H2O molar mass coefficients H2O chemical equation 4 mol H2O 18.0 g H2O 1 mol CH3OH = x x 32.0 g CH3OH 2 mol CH3OH 1 mol H2O 235 g H2O 3.8 Finding Empirical Formulas Calculating Empirical Formulas One can calculate the empirical formula from the percent composition Calculating Empirical Formulas The compound para-aminobenzoic acid (you may have seen it listed as PABA on your bottle of sunscreen) is composed of carbon (61.31%), hydrogen (5.14%), nitrogen (10.21%), and oxygen (23.33%). Find the empirical formula of PABA. Calculating Empirical Formulas Assuming 100.00 g of para-aminobenzoic acid, C: H: N: O: 1 mol 12.01 g 1 mol 5.14 g x 1.01 g 1 mol 10.21 g x 14.01 g 1 mol 23.33 g x 16.00 g 61.31 g x = 5.105 mol C = 5.09 mol H = 0.7288 mol N = 1.456 mol O Calculating Empirical Formulas Calculate the mole ratio by dividing by the smallest number of moles: C: 5.105 mol 0.7288 mol = 7.005 7 H: 5.09 mol 0.7288 mol = 6.984 7 N: 0.7288 mol 0.7288 mol = 1.000 O: 1.458 mol 0.7288 mol = 2.001 2 Calculating Empirical Formulas These are the subscripts for the empirical formula: C7H7NO2 Combustion Analysis • Compounds containing C, H and O are routinely analyzed through combustion in a chamber like this – C is determined from the mass of CO2 produced – H is determined from the mass of H2O produced – O is determined by difference after the C and H have been determined Elemental Analyses Compounds containing other elements are analyzed using methods analogous to those used for C, H and O Stoichiometric Calculations The coefficients in the balanced equation give the ratio of moles of reactants and products Stoichiometric Calculations From the mass of Substance A you can use the ratio of the coefficients of A and B to calculate the mass of Substance B formed (if it’s a product) or used (if it’s a reactant) Stoichiometric Calculations C H O + 6 O 6 CO + 6 H O 6 12 6 2 2 2 Starting with 1.00 g of C6H12O6… we calculate the moles of C6H12O6… use the coefficients to find the moles of H2O… and then turn the moles of water to grams Real Life Analysis • What is the percent composition of an organic substance called ethanol? Combust 11.5 g ethanol Collect 22.0 g CO2 and 13.5 g H2O 3.6 Combust 11.5 g ethanol Collect 22.0 g CO2 and 13.5 g H2O g CO2 mol CO2 mol C gC 6.0 g C = 0.5 mol C g H2O mol H2O mol H gH 1.5 g H = 1.5 mol H g of O = g of sample – (g of C + g of H) 4.0 g O = 0.25 mol O Empirical formula C0.5H1.5O0.25 Divide by smallest subscript (0.25) Empirical formula C2H6O 3.6 Limiting Reactants How Many Cookies Can I Make? • You can make cookies until you run out of one of the ingredients • Once this family runs out of sugar, they will stop making cookies (at least any cookies you would want to eat) How Many Cookies Can I Make? • In this example the sugar would be the limiting reactant, because it will limit the amount of cookies you can make Limiting Reactants The limiting reactant is the reactant present in the smallest stoichiometric amount Limiting Reactants • The limiting reactant is the reactant present in the smallest stoichiometric amount – In other words, it’s the reactant you’ll run out of first (in this case, the H2) Limiting Reactants In the example below, the O2 would be the excess reagent Do You Understand Limiting Reagents? In one process, 124 g of Al are reacted with 601 g of Fe2O3 2Al + Fe2O3 Al2O3 + 2Fe Calculate the mass of Al2O3 formed. g Al mol Al g Fe2O3 124 g Al x mol Fe2O3 needed OR mol Al needed mol Fe2O3 1 mol Al 27.0 g Al x g Fe2O3 needed 1 mol Fe2O3 2 mol Al Start with 124 g Al 160. g Fe2O3 = x 1 mol Fe2O3 g Al needed 367 g Fe2O3 need 367 g Fe2O3 Have more Fe2O3 (601 g) so Al is limiting reagent 3.9 Use limiting reagent (Al) to calculate amount of product that can be formed. g Al mol Al mol Al2O3 2Al + Fe2O3 124 g Al x 1 mol Al 27.0 g Al x 1 mol Al2O3 2 mol Al g Al2O3 Al2O3 + 2Fe 102. g Al2O3 = x 1 mol Al2O3 234 g Al2O3 3.9 Theoretical Yield • The theoretical yield is the amount of product that can be made – In other words it’s the amount of product possible as calculated through the stoichiometry problem • This is different from the actual yield, the amount one actually produces and measures Percent Yield A comparison of the amount actually obtained to the amount it was possible to make Actual Yield Percent Yield = Theoretical Yield x 100