* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download No Slide Title
Toxoplasmosis wikipedia , lookup
Neonatal infection wikipedia , lookup
Cryptosporidiosis wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
Toxocariasis wikipedia , lookup
Dirofilaria immitis wikipedia , lookup
Neglected tropical diseases wikipedia , lookup
Plasmodium falciparum wikipedia , lookup
West Nile fever wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Trichinosis wikipedia , lookup
Visceral leishmaniasis wikipedia , lookup
Onchocerciasis wikipedia , lookup
Hepatitis C wikipedia , lookup
Leishmaniasis wikipedia , lookup
Leptospirosis wikipedia , lookup
Sarcocystis wikipedia , lookup
Hepatitis B wikipedia , lookup
Coccidioidomycosis wikipedia , lookup
Lymphocytic choriomeningitis wikipedia , lookup
Schistosomiasis wikipedia , lookup
Fasciolosis wikipedia , lookup
Oesophagostomum wikipedia , lookup
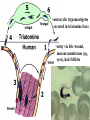
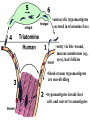
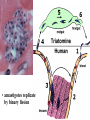
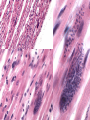
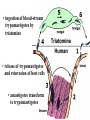
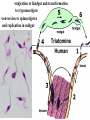
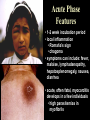
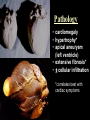
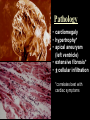
Trypanosoma cruzi • causative agent of Chagas disease • discovered by Carlos Chagas • named organism after mentor, Oswaldo Cruz • determined life cycle • described salient features of disease • 16-18 million infected • 100 million at risk • 50,000 deaths annually • leading cause of cardiac disease in S. and Central America Common Names • • • • • triatomine bugs reduviid bugs assassin bugs kissing bugs conenose bugs Genera • Triatoma • Rhodnius • Panstrongylus Triatomine Vectors • >100 species can transmit • 3 primary vectors • T. dimidiata (central Am.) • R. prolixis (Colombia and Venezuela) • T. infestans (‘southern cone’ countries) •metacyclic trypomastigotes excreted in triatomine feces •entry via bite wound, mucous membranes (eg., eyes), hair follicles •metacyclic trypomastigotes excreted in triatomine feces •entry via bite wound, mucous membranes (eg., eyes), hair follicles •blood-stream trypomastigotes are non-dividing •trypomastigotes invade host cells and convert to amastigotes • amastigotes replicate by binary fission • ingestion of blood-stream trypomastigotes by triatomine • release of trypomastigotes and reinvasion of host cells • amastigotes transform to trypomastigotes •migration to hindgut and transformation to trypomastigote •conversion to epimastigotes and replication in midgut Modes of Transmission SOURCE COMMENTS Natural transmission by triatomine bugs Vector through contamination with infected feces. A prevalent mode of transmission in urban Transfusion areas. Gentian violet treatment (24 hr) eliminates parasites in blood. Occurs during any stage of T. cruzi Congenital infection. Can result in premature labor, abortion neonatal death. Ingestion of food contaminated with metacyclic trypomastigotes. Laboratory Accidental accidents. Types of Vector Transmission Salivarian Stercorarian transmission via mouth parts very efficient infection rate in vector is low hind gut station acquired from feces or eating vector inefficient infection rate in vector is high Factors Influencing Human Transmission • ‘early’ defecation (i.e., during triatomine feeding) • colonization of human habitats - adobe walls - thatched roofs • para-domiciliary cycles - animal stalls adjacent to domicile • proximity to sylvatic cycle Trypanosoma cruzi in the U.S. • triatomine bugs found in U.S. • parasite common in wild animals • 5 confirmed autochthonous cases • why no autochthonous transmission? • late defecaters • zoophillic vectors • better houses inefficient transmission + limited vector-human contact Chagas Control • improvement of human dwellings • separation of animal stalls from house • health education • insecticides • synthetic pyrethroids • eg., Southern Cone Initiative • major in Chagas (T. infestans) • little affect with R. prolixis • gentian violet in blood for transfusions Clinical Course of Chagas • Acute Phase - active infection - 1-4 months duration - most are asymptomatic (children most likely to be symptomatic) • Indeterminate Phase - 10-30 years of latency - relatively asymptomatic with no detectable parasitemia - seropositive • Chronic Phase - 10-30% of infected exhibit cardiomyopathy or megasyndromes Acute Phase Features • 1-2 week incubation period • local inflammation • Romaña’s sign • chagoma • symptoms can include: fever, malaise, lymphadenopathy, hepatosplenomegaly, nausea, diarrhea • acute, often fatal, myocarditis develops in a few individuals • high parasitemias in myofibrils Chronic Chagas' Cardiomyopathy • long latency characterized by seropositivity and no parasitemia • higher prevalence of ECG abnormalities in asymptomatic seropositive persons • progressive development of abnormalities • right bundle branch block • left anterior hemiblock • clinical presentations include: • arrhythmias and conduction defects • congestive heart failure • thromboembolic phenomenon Pathology • cardiomegaly • hypertrophy* • apical aneurysm (left ventricle) • extensive fibrosis* • cellular infiltration *correlates best with cardiac symptoms Pathology • cardiomegaly • hypertrophy* • apical aneurysm (left ventricle) • extensive fibrosis* • cellular infiltration *correlates best with cardiac symptoms Megaviscerae • prevalence varies by geographical zones • Chili, central Brazil • colon and esophagus most frequently affected • megaesophagus • painful swallowing • regurgitation • megacolon • severe constipation • destruction of parasympathetic neurons dilation non-Chagas C = heart S = colon E = esophagus Chagas Basis of Pathogenesis autoimmunity? few (if any) parasites anti-self responses (humoral and cellular) slow development organ specificity parasite-mediated destruction? persistent low level parasitemia (PCR) inflammation correlates with parasites disease exacerbated by immune suppression successful treatment of chronic patients (?) • altered immune response? (Th1Th2 switch correlated with severe disease) • chagasic factor or toxin? (proposed by not found) DIAGNOSIS • history of living in infested house • bug bite, chagoma, Romaña's sign • cardiac or gastrointestinal symptoms • imaging • detection of parasite (acute stage) • serology (chronic stage) DIAGNOSIS • parasite detection • direct examination • stained blood smears • inoculation into mice • in vitro culture • xenodiagnosis • PCR • serological tests • hemagglutination • immunofluorescence • ELISA • complement fixation TREATMENT • acute stage –nifurtimox (8-16 mg/kg/day, 60-90 days) –benzidazole (5-7 mg/kg/day, 30-120 days) –allopurinol (experimental) –azole antifungal agents (experimental) • chronic stage –treat symptoms Viotta et al (1994) Am. Heart J. 127:151 benznidazole treatment (5 mg/kg/day, 30 d) followed for 8 years % of patients exhibiting treated (131) control (70) electrocardiogram changes deterioration in clinical condition sero-negative conversion 4% 30% 2% 17% 19% 6% Lauria-Pires et al (2000) AJTMH 63:111 • Brasília street cleaners treatment • standard treatment with nifurtimox or benznidazole • 10 year follow up • treated vs. untreated: • no parasitiological cure (PCR) • no sero-negative conversion • no ECG improvements • administration of nitroderivatives • severe side effects • compliance problems Trypanosoma rangeli • can be confused with T. cruzi • non-pathogenic for humans • pathogenic for triatomines mode of transmission? • found in both salivary glands and feces