* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Final Poster
Electrical ballast wikipedia , lookup
Current source wikipedia , lookup
Telecommunications engineering wikipedia , lookup
Pulse-width modulation wikipedia , lookup
Resistive opto-isolator wikipedia , lookup
Switched-mode power supply wikipedia , lookup
Optical rectenna wikipedia , lookup
Photomultiplier wikipedia , lookup
Rectiverter wikipedia , lookup
Immunity-aware programming wikipedia , lookup
Buck converter wikipedia , lookup
Alternating current wikipedia , lookup
Voltage regulator wikipedia , lookup
Stray voltage wikipedia , lookup
Opto-isolator wikipedia , lookup
Surge protector wikipedia , lookup
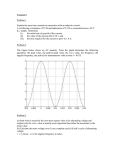
Determination of Intercellular Calcium Concentrations in Cardiac Myocytes Using Fluorescence and a Single Fiber Optic Method Paul Clark, Martin Garcia, Chris Gorga, John Ling, Jordan LoRegio Sponsor: Dr. Franz Baudenbacher INTRODUCTION RESULTS (cont…) EXPERIMENTAL SETUP DEVICE FABRICATION A Figure 1: X-rod Emission and Excitation Spectra. Also include in the figure above is the wavelength at which the laser and filter are set. C Figure 2: Standard Steps of Soft Lithography. The basic soft lithography used in fabricating the PDMS device integrated with an optical fiber, allowing for real-time data collection. Once plasma bonded to the glass slide the device can capture cells up to 10 microns in diameter. Proof of principle (i.e. changes in the magnitude of fluorescence can be quantified using the integrated optical fiber method) was achieved using a simple single channel device and fluoroscein solutions of varying concentrations. Figures 6a–6d, below, show an increase in the magnitude of the voltage changes as fluorescein concentration is increased. 3 3.5 3 2.8 2.7 2.6 2.5 2 1.5 1 2.5 0.5 0 2.4 0 10 20 30 40 0 50 20 40 60 80 100 Time (Seconds) Time (Seconds) Figure 6b: 10µM Fluoroscein Solution. The yield is approximately a 1.75V increase in voltage. Figure 6a: 1µM Fluoroscein Solution. The yield is approximately a 0.4V increase in voltage. OBJECTIVES o Design and fabricate a microfluidic device to measure the calcium flux of a single cardiac myocyte in real time as it contracts. 25 30 CONCLUSIONS 4 2.9 EXPERIMENTAL SETUP 6 4.5 4 5 Voltage (V) 3.5 3 2.5 2 1.5 1 4 3 2 o A microfluidic device has been developed allowing for the quantification of the magnitude of fluorescence using optical fibers as the detection method. o A second device has been developed that should allow for the capture and excitation of single cardiac myocytes. o Electrodes specific to this device must be designed for better alignment and larger platform for the device. o A better hole punching and fiber insertion technique must be developed to prevent channel collapse. 1 0.5 0 0 0 Figure 4b: Data acquisition setup. Shown here is the final setup with the device mounted on the microscope ready for data acquisition. 20 Figure 8b: Proper Device, Electrode Alignment. At right is the proper alignment of the electrodes over the cell loading area. Note the optical fiber just to the upper right as well as the collapsed channels, later found to be due to a damaged master. RESULTS Figure 4a: Completed device setup. Shown here is the completed device, mounted on the electrodes, and clamped into place. 15 Figure 8a: Loading of Live Myocytes. At the left three cardiac myocytes are experiencing suction in an attempt to load them via the channel on the left of the screen. A a collapsed channel prevented a successful loading. Figure 5: Overall Schematic of Experimental Setup. The system is capable of simultaneous dye excitement and fluorescence reading. This is possible because of the optical properties of X-rod couple with the dichroic filters. The PMT (photomultiplier) amplifies and converts the flourescence signal into a voltage which can be collected using LabView. DEVICE SPECIFICATIONS o Design and fabricate a microfluidic device showing calcium flux can be quantified using an optical detection method. 10 Figure 7. Fluoroscein as Correlated to Voltage Increase. The above trendline shows that the magnitude of voltage increases linearly with increasing fluoroscein concentration. B Figure 3: PDMS device for trapping cardiac myocytes. Note the cell inlet (A), main suction channel and debris reservoir (B), fine suction channel (C), and cell chamber (D), where the electrodes will be placed for excitation. 5 [Fluoroscein] (µM) Voltage (V) A microfluidic platform was chosen to isolate single cardiac myocytes for data acquisition via an integrated optical fiber. The cells are died with the fluorescent dye X-rod (emission and excitation spectra below), which fluoresces when exposed to calcium. Through electrical pulses the trapped cell is coaxed into releasing calcium for its sER (smooth endoplasmic reticulum), thus exciting the X-rod and showing a change in fluorescence intensity. 4.5 4 3.5 3 2.5 2 1.5 1 0.5 0 0 Voltage (V) The development of a method for the study of calcium signaling between cardiac myocytes will lead to a great understanding of many cellular processes, most notably contractile force. Once quantified, the calcium flux data (for individual myocytes) will be equated to a force, thus creating a normal line equating calcium concentration to contractile force. D Voltage (V) Heart contraction depends on the coordination of the electrical signals created by the individual cardiac myocyte. Extracellular calcium flux allows cells to communicate with neighboring cells, thus propagating the electrical signal which was initiated in the sinoatrial (SA) node. Volatage (V) [Fluoroscein] correlated to Voltage 10 20 30 40 50 60 70 80 Time (Seconds) Figure 6c: 15µM Fluoroscein Solution. The yield is approximately a 2.5V increase in voltage. 0 10 20 30 40 50 60 Time (Seconds) Figure 6d: 25µM Fluoroscein Solution. The yield is approximately a 4V increase in voltage. ACKNOWLEDGMENTS This work would not have been possible without the advice of our advisor, Dr. Baudenbacher or the help of graduate students Tobias Meyer, David Schaffer, and Raghav Venkataraman. This work was supported by funding from the Vanderbilt Institute for Biosystems Research and Education (VIIBRE).