* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Mass Spectrometer
Hydrogen atom wikipedia , lookup
Specific impulse wikipedia , lookup
Modified Newtonian dynamics wikipedia , lookup
Schiehallion experiment wikipedia , lookup
Nuclear physics wikipedia , lookup
Anti-gravity wikipedia , lookup
Electromagnetic mass wikipedia , lookup
Mass versus weight wikipedia , lookup
Negative mass wikipedia , lookup
Conservation of mass wikipedia , lookup
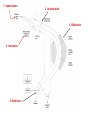
Mass Spectrometer 1. Vaporisation 3. Acceleration Mass Spectrometer 4. Deflection 2. Ionisation 5. Detection 1. Vaporisation Sample vaporised by oven if liquid, then injected into vacuum chamber at low pressure 3. Acceleration Negatively charged plates produce an electric field which accelerates the positively charged ions. They are focused into a narrow beam as they pass through slits 2. Ionisation •Sample bombarded by a stream of high energy electrons from an electron “gun”. •These high energy electrons knock an e- from a sample atom. Producing a positive ion. •X(g) X+(g) + e- Usually only one e- is removed to form X+(g) ions but X2+(g) are occasionally formed. Sometimes the particles are broken into fragments by the high energy electrons - fragmentation 4. Deflection •The ions are deflected by a strong, variable magnetic field •The extent of deflection is determined by the mass and charge of the ions. X+ By varying the strength of the magnetic field ions with specific m/z are focused through the collector slit m/z too small For a given magnetic field •The greater the mass, m, the less the ions are deflected m/z too large •The greater the positive charge, z, the more they are deflected The larger the value of the mass to charge ratio, m/z, the smaller the deflection 5. Detection All the counts together produce a mass spectrum. The peaks are proportional to the abundance of the positive ios Abundance When a positive ion reaches the detector it produces a tiny current. This signal is fed to a recorder which counts the number of signals for each setting of the magnetic field. X+ m/z The Mass Spectrum of Cl2 35Cl -35Cl Relative Abundance 35Cl 35Cl -37Cl Or Relative intensity 37Cl 37Cl -37Cl 35 37 Ratio 3 : 1 70 72 74 Ratio 9 : 6 : 1 m/z The Relative Atomic Mass (Ar) of an element The relative atomic mass (Ar) of an element is the average mass of one atom compared with one twelfth of the mass of an atom of carbon-12 Ar = average mass of 1 atom of element 1/12 mass of one atom of carbon-12 Ar = average mass of 1 atom of element x 12 mass of one atom of carbon-12 Calculating the Relative Atomic Mass (Ar) of an element Mass to charge ratio (m\z) 35 37 Relative Abundance (%) 75 25 Calculate a weighted average Ar of chlorine = (35 x 75) + (37 x 25) (75 + 25) Ar of chlorine = 2625 + 925 100 Ar of chlorine = 35.5 Calculating the Relative Atomic Mass (Ar) of Lead Mass to charge ratio (m\z) 204 206 207 208 Relative Abundance (%) 1.55 23.6 22.6 52.3 Ar of lead= (204 x 1.55) + (206 x 23.6) + (207 x 22.6) + 208 x 52.3) (1.55 + 23.6 + 22.6 + 52.3) Ar of lead = 316.2 + 4861.6 + 4678.2 + 10878.4 100.05 Ar of lead = 20734.4 100.05 Ar of lead = 207.2 The Relative Molecular Mass (Mr) of an element or compound • Relative atomic mass values (Ar) can be used to calculate the Relative molecular mass (Mr) of an element or compound • The relative molecular mass (Mr) of an element or compound is the sum of the relative atomic masses of all the atoms in the molecule, divided by one twelfth of the mass of an atom of carbon-12 • Mr = mass of 1 molecule 1/12 mass of one atom of carbon-12 rearranged to • Mr = mass of 1 molecule x 12 mass of one atom of carbon-12 Relative Molecular Mass (Mr) • Mass spectra can be used to find the Mr of a compound