* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File - Chemistry
Survey
Document related concepts
Transcript
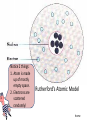
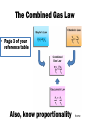
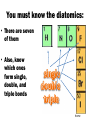
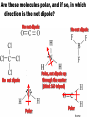
Chemistry EOC Goal 1 and 2 home • Please get out your reference table as you will be directed to pull certain information from it to help you solve specific problems home Competency Goal 1 • The learner will develop abilities necessary to do and understand scientific inquiry (4 %) home Goal 1.01 • Design, conduct and analyze investigations to answer questions related to chemistry You should be able to: home Goal 1.02 • Evaluate reports of scientific investigations from an informed scientifically-literate viewpoint including considerations of: home Goal 1.03 • Evaluate experimental designs with regard to safety and use safe procedures in laboratory investigations: -Use MSDS info -Acid into water, like you oughter! home Competency Goal 2 • The learner will build an understanding of the structure and properties of matter (34 %) home Goal 2.01 • Analyze the historical development of the current atomic theory - Let’s take a deeper look into this goal… home Describe the composition of the atom home The Atom: From Philosophical Idea to Scientific Theory I look pretty good for a dude 2400 years old!!! In Greek, the prefix "a" means "not" and the word "tomos" means cut. Our word atom therefore comes from atomos, a Greek word meaning uncuttable. home Dalton’s Atomic Theory 1. All matter is composed of small particles called atoms. 2. Atoms of a given element are identical; atoms of different elements are different 3. Atoms cannot be subdivided, created, or destroyed. 4. Atoms of different elements combine in simple whole-number ratios to form compounds 5. Atoms are combined, separated, or rearranged in chemical reactions home The Electron JJ Thomson--1897 Plum Pudding Model But the atom is Neutral!!!! While studying electric discharge in Cathode-ray tubes, Thomson discovered the electron!!! home The Nucleus Ernest Rutherford--1908 Rutherford’s Gold Foil Experiment In an attempt to build on Thomson’s model, I discovered some amazing stuff!!! home Notice 2 things: 1. Atom is made up of mostly empty space. 2. Electrons are scattered randomly! Rutherford’s Atomic Model home Nucleus Electrons Neils Bohr--1913 home Bohr Model of the Atom The Bohr Model has the following features: 1. There is a nucleus(Rutherford’s Discovery) 2. The electrons move about the nucleus in “stationary, stable states”(later referred to as orbits)! home The Wave Model of the Atom home Discovery of the neutron James Chadwick--1932 In 1932, Sir James Chadwick, working to interpret the results of other scientists, discovered the neutron!!! home Democritus/Dalton home Goal 2.02 • Examine the nature of atomic structure Subatomic Particle Proton Relative Mass Charge Where Found 1 +1 In Nucleus Neutron 1 0(neutral) In Nucleus Electron 1/2000 or 0 -1 Outside nucleus in electron cloud home • Atomic # (Z) – Number of protons • Mass # (A) – Number of protons + neutrons – Also found by rounding the average atomic mass Same # protons, different # NEUTRONS home Atomic # Atomic Mass A # = 27 M # = 59 # p = 27 # e = 27 # n = 32 M#=#p+#n 59 = 27 + #n home 2 ways of writing Isotopes H-1 H-2 H-3 • Each of these represents Hydrogen • The number represents the mass # • Each of these represents Carbon • The top # represents the mass # • The bottom # represents the Atomic # home Goal 2.03 • Apply the language and symbols of chemistry • Be able to name: – Ionic compounds – Covalent compounds – Polyatomic ions Number Prefix 1 Mono- 2 Di- 3 Tri- 4 Tetra- 5 Pentahome Name these: • • • • • • • • CaCl2 S2O5 PO43K2SO4 PO3 FeS C2H3O2Ba(OH)2 Take your time to look up the names of the elements and polyatomic ions! home A few more things you should know: • The names of the following acids: – HCl – HNO3 – H2SO4 – CH3COOH or HC2H3O2 – And all of the symbols: (s), (g), (l), (aq), etc. home Goal 2.04 • Identify substances using their physical properties: Most of this information can be obtained directly from the reference tables home Some examples include: • • • • • • Melting point Boiling point Specific heat Density Solubility rules And much more • You could be told an unknown substance has a specific heat of 0.129 J/g˚C … what is it? home Levels of Saturation Saturated solution—Solution has dissolved all the solute possible Unsaturated solution—solution “could” dissolve more solute. Supersaturated solution—Solution has dissolved all solute possible, but more solute is added anyway. *** Don’t forget… LIKE DISSOLVES LIKE home How many grams of KNO3 will dissolve in 100 g of water at 60o C? 110 g home At what temperature will approximately 95 g of NaNO3 dissolve in 100 g of water? 30 oC home At what temperature will the same amount of KNO3 and NaNO3 dissolve in 100 g of water? ~ 68 oC home Which term would describe a solution of 90 g of KCl dissolved in 100 g of water at 80 oC? Unsaturated Saturated Supersaturated Can’t but about 50 g dissolve at 80 oC. home Suppose 90 g of KI are placed in 100 g of water at 10 oC. What type of solution would you have? Unsaturated Saturated Supersaturated Could dissolve about 135 g. home Approximately, how much NH4Cl would dissolve in 200 g of water at 70 oC? Twice as much solute, because twice as much solvent!!! 120 g home Effects of Temperature on Solubility Gases in liquidsAs temperature increases, the solubility decreases. Solids in liquids – As temperature increases, the solubility increases. home Goal 2.05 • Analyze the basic assumptions of kinetic molecular theory and its applications. home The Combined Gas Law • Page 3 of your reference table Also, know proportionality home Tips for working gas law problems: • 1. • 2. • 3. • 4. home Ideal Gas Equation PV = nRT , where… P is pressure V is volume n is number of moles R is the Ideal gas constant T is the temperature You always have this!!! home Dalton’s Law of Partial Pressures • PT = P 1 + P 2 + P 3 … • Page 3 of your reference table • Remember, if a gas is collected over water, you have to account for water vapor pressure ***Conceptual understanding, calculations home Goal 2.06 • Assess bonding in metals and ionic compounds as related to chemical and physical properties • Ionic compound properties: high BP, high MP, high electrical conductivity (in molten or aqueous state, brittle home Do you remember why atoms bond? home Ionic Bond • Gain or loss of electrons to form ions --Electrostatic attraction between metals and nonmetals home Oxidation # home Metallic Bond • “Sea of Electrons” • Form between a metal and metal High luster, malleable, ductile, high conductivity, high MP, high BP home Goal 2.07 • Assess covalent bonding in molecular compounds as related to molecular geometry and chemical and physical properties. home Covalent Bond • Sharing of electrons to form a compound that is stable (octet rule) -- Forms between nonmetal and nonmetal home What type of bond forms between each of these pairs? • • • • • • • Sodium and bromine Ammonium and chlorine Zinc and copper Oxygen and oxygen Sulfur and oxygen Hydrogen and chlorine Gold and gold You also need to know the properties of ionic and covalent compounds! home Ionic vs. Covalent Compounds Ionic Compounds Covalent Compounds • Hard, but brittle • High MP and BP • Many can dissolve in water • Conductors in molten state • • • • Soft Lower MP and BP Good insulators Mostly gases at room temp. home You must know the diatomics: • There are seven of them • Also, know which ones form single, double, and triple bonds home Bond Energy vs Bond Length Bond Energy Bond Length Triple > double > single Single > double > triple Do you know why this is true? home VSEPR • What does VSEPR stand for? • Can you name the geometry, hybridization, and bond angle for each of the above molecules? home Lewis Dot Structures • The two things you have to remember in drawing covalent molecules are that: • 1. You have to satisfy the octet rule – Except hydrogen and helium • 2. You have to make sure you have the right number of electrons home Are these molecules polar, and if so, in which direction is the net dipole? home Covalent-Network Compounds • Covalent compounds that do not contain individual molecules, but instead can be pictured as continuous, 3-D, networks of bonded atoms home Intermolecular Forces • Hydrogen Bonds • London Dispersion Forces home Goal 2.08 • Assess the dynamics of physical equilibria home Heating Curve • Can you change temperature and phase at the same time? home Know these: home