* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 05. The concept of disease, controlled by the International Health
Survey
Document related concepts
Sociality and disease transmission wikipedia , lookup
Common cold wikipedia , lookup
Behçet's disease wikipedia , lookup
Rheumatic fever wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Kawasaki disease wikipedia , lookup
Childhood immunizations in the United States wikipedia , lookup
Transmission (medicine) wikipedia , lookup
Pathophysiology of multiple sclerosis wikipedia , lookup
Marburg virus disease wikipedia , lookup
Infection control wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Multiple sclerosis research wikipedia , lookup
Germ theory of disease wikipedia , lookup
Vaccination wikipedia , lookup
Transcript
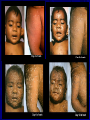
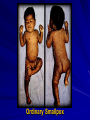
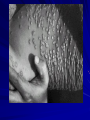
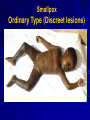
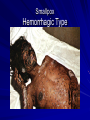
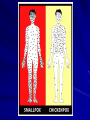
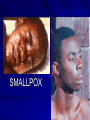
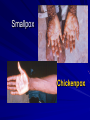
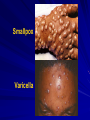
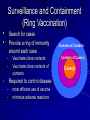
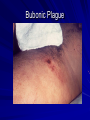
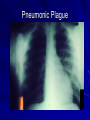
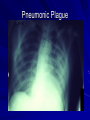
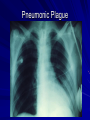
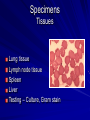
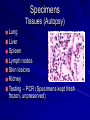
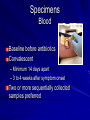
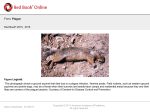
The concept of disease, controlled by the International Health Regulations Smallpox Plague Disease, Prevention, and Intervention Progression of Smallpox Incubation Period Prodrome Stage Macules Papules Vesicles Pustules Scabs Scars Smallpox Prodrome Incubation period 12 days (range 7-17 d) Prodrome – abrupt onset of fever >101oF – malaise, headache, muscle pain, nausea, vomiting, backache – lasts 1- 4 days Smallpox Rash Enanthem (mucous membrane lesions) appears ~ 24 hours before skin rash – Minute red spots on the tongue and oral/pharyngeal mucosa – Lesions enlarge and ulcerate quickly – Become infectious from lesions in mouth – Virus titers in saliva highest and most infectious during first week of exanthem (skin rash) Smallpox Rash • Exantham (skin rash) – (21 days) Stages: macules, papules, vesicles, pustules, scabs Pustules raised, round, firm - like small beads in the skin (“shotty”) Umbilication common • Begins and most dense on face and extremities • • (centrifugal distribution) Lesions on palms and soles (>50% of cases) Lesions in same stage and evolve slowly (1-2 days/stage) Day 4 of rash Ordinary Smallpox Pustular lesions on palms Flattened lesions on soles Smallpox Ordinary Type (Discreet lesions) Smallpox Ordinary Type (Dense lesions) Smallpox Flat-type Smallpox Hemorrhagic Type Varicella Differential Diagnosis SMALLPOX CHICKEN POX Deep, hard lesions Superficial Round, well circumscribed Not well circumscribed Confluent or umbilicated Lesions at same stage of development Confluence and umbilication uncommon Lesions at all stages of development CHICKENPOX SMALLPOX Smallpox Chickenpox Smallpox Varicella Smallpox Outbreak Control Activities and Strategies Surveillance and Containment (Ring Vaccination) • • Search for cases Provide a ring of immunity around each case - • Vaccinate close contacts Vaccinate close contacts of contacts Required to control disease - most efficient use of vaccine minimize adverse reactions Contacts of Contacts Contacts of Case(s) Case(s) Surveillance Pre-Event Surveillance – Identify cases with typical presentation – Rapid laboratory confirmation – Confirmation initiates contact vaccination – Passive with more specificity Post-Event Surveillance – Identify all potential cases (typical/atypical) – Clinical diagnosis can initiate contact vaccination – Active with increased sensitivity Epidemiologic Investigation Identify source of initial introduction(s) Identify contacts and other population(s) at risk Characterize and define outbreak – Identify unusual/unexpected features – Scope Monitor effectiveness of control measures Communicate information and define public health recommendations Contact Tracing Goal is to find as many contacts as possible – Contact with patient after onset of fever – Prioritize based on closeness, length, and date of exposure – If too many to find quickly, consider all people in same room (or possibly facility) with smallpox case after onset of fever as contacts Contact Tracing Examples: – Highest priority - people who live full-time in home and other face-to-face contacts after onset of fever – Next priority – non face-to-face contacts exposed in a medical care facility or in home – Last priority – people in same facility after onset of fever (other than home or hospital) but without face-to-face contact Isolation and Quarantine Goals of Smallpox Isolation Protect others from becoming infected – Healthcare personnel – Response personnel – Other patients Isolate smallpox patient – Prevent sharing of airspace (respiratory isolation) – Prevent direct contact (protective clothing) – Prevent contact with infectious materials (decontamination) Prevent sharing of airspace with potentially infectious patients Negative pressure isolation rooms. Separate facilities for larger groups. Respirators for unvaccinated careproviders. Personal Protective Equipment Use disposable gloves, gowns, and shoe covers. Reusable bedding and clothing should be autoclaved or laundered in hot water with bleach. Respiratory Protection Smallpox •Airborne precautions •Recommendation: fitted NIOSH N95 or greater respirators for personnel entering patient room Properly Fitted – air goes through mask filter Isolation Strategies 3 groups to consider – Confirmed or suspected smallpox cases – Febrile vaccinated contacts – Asymptomatic (vaccinated) contacts under surveillance Confirmed/Suspected Smallpox Cases (Few) Known or presumed infectious individuals Hospital isolation room(s) Rooms under negative pressure At least 6 to12 air changes/hour Air vented to outside Air not re-circulated to other rooms or areas Confirmed/Suspected Smallpox Cases (Many) Designated Facility for smallpox patients Aerosol precautions not needed if only potential smallpox cases in facility and no shared ventilation system – All people admitted/entering facility vaccinated Febrile Vaccinated Contacts Vaccinated contacts with fever (no rash) – – – – Two successive temps > 101º F (38ºC) Less need for medical care No shared ventilation system Can be housed in same facility with smallpox cases or separate facility – All persons in facility require vaccination Asymptomatic Vaccinated Contacts Not infectious – Own home or other lodging – No special ventilation or medical requirements All other persons staying in home must also be vaccinated – Household members with contraindications stay elsewhere Fever surveillance for 18 days from last exposure or 14 days from vaccination Decontamination Air: – UV Light Sensitive – Exhaust, Good Air Flow Surfaces: – Diluted bleach solution (Fresh every day) – Hospital disinfectants Blood, pus contaminated equipment: – Wash before disinfecting Decontamination Laundry: – Contain separately – Dissolving laundry bags if available – Don’t sort first, wash, then sort – Hot water with detergent and/or bleach Household: – Basic cleaning – Wash all clothing in hot w/ bleach if possible – Public health review of home Plague Objectives Identify plague bacterium Epidemiology Natural Occurrence Bio-Terror Threat Plague can cause large numbers of cases Could create panic Considered for use since 14th century Clinical Syndromes Bubonic Pneumonic Septicemic Plague Meningitis Pharyngeal “Safety Pin” Y. Pestis in blood Bubonic Plague Infected flea bite Exposure through break in skin No person-to-person Untreated progresses to pneumonic Pneumonic Plague Inhalation of plague bacteria Disease progression – Respiratory failure – Shock – Rapid death Person-to-person transmission Septicemic Plague Primary Form – Direct inoculation in bloodstream Secondary Form – Development of untreated pneumonic or bubonic plague Epidemiology Natural Reservoirs Bites of infected flea Blood meal from bacteremic animal Regurgitates into human/ animal host Common reservoirs – Deer mice – Ground squirrels Epidemiology Transmission Bite of infected flea Respiratory droplets Direct contact (1,5-2 m) Direct skin/mucous membrane less common BT event – Respiratory droplets or aerosols Plague Incidence Worldwide, 1970 - 1998 All inhabited continents, but Australia 1,500 to 3,000 cases annually Greatest Concentrations – Asia, South America Plague Bioterrorism Scenario Most dangerous as aerosol Outbreak of pneumonic Possibly pharyngeal or ocular Report all suspect cases to public health immediately Bubonic Plague Incubation: 2 to 6 days Symptoms – Lymphadenopathy, fever – Buboes at site of inoculation Disease Progression - Untreated – Septicemia – Secondary Pneumonic Plague – Meningitis (rare) Bubonic Plague Bubonic Plague Bubonic Plague Pneumonic Plague Incubation: 2 to 4 days (range 1 to 6 days) Symptoms – Acute fever, chills, malaise, myalgias – Productive cough – Watery mucoid sputum, may be bloody – Associated chest pain, increasing dyspnea Pneumonic Plague Disease Progression – Adult Respiratory Distress Syndrome – Refractory pulmonary edema – Signs of shock – Without treatment in less than 24 hours, almost universally fatal Pneumonic Plague Coughing patient can spread Respiratory precautions Rapidly expanding infiltrates Pulmonary parenchymal necrosis and hemorrhage Occasional pulmonary abscesses Enlarged hilar nodes and pleural effusions Pneumonic Plague Pneumonic Plague Pneumonic Plague Septicemic Plague Incubation: Most common as complication of pneumonic or bubonic plague Symptoms – Acute fever, chills, prostration, abdominal pain, nausea, vomiting Disease Progression – Purpura – DIC – Hypotension and other signs of shock – Fatal if not treated Infection Control Large numbers of plague bacilli Respiratory droplet spread in close direct contact Respiratory droplet precautions with suspect cases Contact public health Bubonic Plague Differential Diagnosis Streptococcal or staphylococcal adenitis Purulent/ inflamed lesion often distal to nodes (Staphylococcal aureus, Involved nodes more likely to be fluctuant Staphylococcal pyogenes) Ascending lymphangitis or cellulitis may be present Tularemia (Francisella tularensis) Cat scratch disease (B. henselae) Ulcer or pustule distal to nodes Rarely as fulminant as in plague Systemic toxicity uncommon History of cat contact/scratch Indolent clinical course Primary lesion at site of scratch No systemic toxicity Bubonic Plague Differential Diagnosis Mycobacterial infection, including scrofula (Mycobacterium tuberculosis and other Mycobacterium species) Lymphogranuloma venereum (Chlamydia trachomatis) • Adenitis occurs in cervical region • Usually painless • Indolent clinical course • More likely to occur in immunocompromised patients • Adenitis occurs in the inguinal region • Sexual exposure 10-30 days previously • Suppuration, fistula tracts common • Exquisite tenderness usually absent • Although patients may appear ill (headache, fever, myalgias), systemic toxicity not present Bubonic Plague Differential Diagnosis Chancroid (Hemophilus ducreyi) Adenitis occurs in inguinal region Ulcerative lesion present Systemic symptoms uncommon; toxicity does not occur Primary genital herpes Genital area Adenitis occurs in the inguinal region Severe systemic toxicity not present Primary or secondary syphilis (Treponema pallidum) Enlarged lymph nodes in inguinal region Lymph nodes generally painless Chancre may be noted Strangulated inguinal hernias Evidence of bowel involvement Primary Pneumonic Plague Differential Diagnosis Inhalational anthrax (Bacillus anthracis) Tularemia (Francisella tularensis) • Widened mediastinum and pleural effusions • Not true pneumonia • Minimal sputum production • Hemoptysis uncommon Not as rapid or fulminant as in pneumonic plague Primary Pneumonic Plague Differential Diagnosis Community-acquired bacterial pneumonia Mycoplasmal pneumonia (Mycoplasma pneumoniae) Pneumonia caused by Chlamydia pneumoniae Legionnaires' disease (Legionella pneumophila or other Legionella species) Psittacosis (Chlamydia psittaci) Other bacterial agents (e.g., Staphyloccocus aureus, Streptococcus pneumoniae, Haemophilus influenzae, Klebsiella pneumoniae, Moraxella catarrhalis) Rarely as fulminant Usually occur in persons with underlying pulmonary or other disease or in the elderly Bird exposure with psittacosis Gram stain may be useful Community outbreaks not as explosive as pneumonic plague outbreak S. pneumoniae usually institutional Community outbreaks of Legionnaires' disease often involve exposure to cooling systems Primary Pneumonic Plague Differential Diagnosis Viral pneumonia Influenza Hantavirus RSV CMV Q fever (Coxiella burnetii) Influenza generally seasonal History of recent cruise ship travel or travel to tropics Exposure to excrement of mice with Hantavirus RSV usually occurs in children CMV usually occurs in immunocompromised patients Exposure to infected parturient cats, cattle, sheep, goats Severe pneumonia not prominent Laboratory confirmation Critical for first cases Collect immediately and before antibiotics Store appropriately Presumptive diagnoses Specimens Respiratory Pharyngeal swabs Tracheal washes or aspirates Sputum specimens Trans-thoracic lung aspirates Pleural fluid collection Testing – Staining, culture, DNA amplification Specimens Tissues Lung tissue Lymph node tissue Spleen Liver Testing – Culture, Gram stain Specimens Tissues (Autopsy) Lung Liver Spleen Lymph nodes Skin lesions Kidney Testing – PCR (Specimens kept fresh frozen, unpreserved) Specimens Blood Baseline before antibiotics Convalescent – Minimum 14 days apart – 3 to 4 weeks after symptom onset Two or more sequentially collected samples preferred Laboratory Testing Pneumonic Plague Collect before initiating therapy Sputum, blood, lymph node aspirate Gram Stain and culture – Gram-negative coccobacillus – Bipolar (safety pin) staining Acute and convalescent bloods for public health Recommended Plague Treatment Contained Casualty - Adults Preferred Streptomycin 1g IM BID Gentamicin 5 mg/kg IM or IV daily OR 2 mg/kg loading with 1.7 mg/kg IM or IV TID Alternative Doxycycline 100mg IV BID or 200mg IV Ciprofloxacin 400mg IV BID Chloramphenicol 25 mg/kg IV QID Recommended Plague Treatment Mass Casualty - Adults Preferred Doxycycline 100 mg PO BID Ciprofloxacin 500 mg PO BID Alternative Chloramphenicol 25 mg/kg PO QID Plague Infection Control Precautions Bubonic Standard Pneumonic Standard and Respiratory Septicemic Standard Suspect Respiratory and Isolation Isolation for 48 hours of antibiotic treatment or until clinical improvement Plague Infection Control Cohort and Droplet precautions if no isolation available Isolation of contacts may increase in importance for outbreak control Corpses – Standard Precautions Shipping Biological Specimens Infectious Substance UN Hazard Class 6 UN Division 6.2 UN = United Nations Shipping Biological Specimens Infectious Substances Primary Container – Positive Seal Absorbent Material Shipping Biological Specimens Infectious Substances Secondary Packaging Water-tight Leak proof Shipping Biological Specimens Infectious Substances Must be approved by the United Nations. Designated as UN Certified Packaging or UN Specified Packaging.