* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download here
Trichinosis wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Anthrax vaccine adsorbed wikipedia , lookup
Microbicides for sexually transmitted diseases wikipedia , lookup
Dirofilaria immitis wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Oesophagostomum wikipedia , lookup
Schistosomiasis wikipedia , lookup
Neonatal infection wikipedia , lookup
Hepatitis C wikipedia , lookup
Whooping cough wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Coccidioidomycosis wikipedia , lookup
Hepatitis B wikipedia , lookup
Neisseria meningitidis wikipedia , lookup
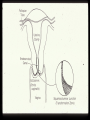
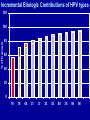
Prophylactic HPV vaccines to prevent cervical cancer —but what else? Aimee R. Kreimer, Ph.D. [email protected] National Cancer Institute March 20, 2010 American Medical Writers Association Mid-Atlantic Chapter (AMWA-MAC) Cervical Cancer History • 1842: Italian investigator D. Rigoni-Stern reported that prostitutes had an unusually high rate of cervical cancer, while nuns had virtually no cases of the disease (vice versa for breast cancer). • 1940’s: Papanicolaou develops his cytology test (Pap smear); cervical cancer rates decline by >75% in U.S. over the next ~50 years • 1960’s-1980’s: Infectious agents implicated---HSV2 Cervical Cancer History • 1974: Zur Hausen identifies human papillomavirus (HPV) DNA in cervical cancer tissue • Late 1980s: Epidemiologic studies show association of HPV and cervical cancer; PCR assays developed • Mid to Late 1990’s: ≥95% cervical CA’s have HPV DNA • 2003: FDA approval of HPV DNA Test for CxCA Screening • 2003-5: Vaccines based on Capsid Proteins Protect Against HPV infection and CIN Summary of Today’s Talk • • • • • Global Epidemiology of cervical cancer Etiology/biology Anatomy/physiology 2° prevention through screening 1° prevention through prophylactic vaccination – Details of the HPV vaccine Cervical Cancer epidemiology • Worldwide ~500,000 new cases per year – 3rd most common cancer in women • Worldwide ~250,000 deaths per year – ~1/10th of all female cancer deaths • Incidence and survival rates vary by race and geographical region Age-standardized incidence and mortality rates of cervical cancer, 2002 Mortality rate per 100,000 women 4.0 11.2 Incidence rate per 100,000 women 10.3 19.1 0 5 10 15 20 Developed Countries Less Developed Countries GLOBOCAN 2002 25 Cervical Neoplasia risk factors The Primary Risk Factor for Cervical Cancer: Human Papillomavirus (HPV) Cervical Neoplasia risk factors • Risk factors for HPV acquisition – lifetime # of sexual partners – sexual practices of male partner • Co-factors for progression – – – – immunosuppression other infectious agents (HSV-2, chlamydia) Parity, oral contraceptives smoking? dietary factors? genetics? HPV • Small DNA virus (~8000bp) • More than 100 types identified based on the genetic sequence of the outer capsid protein L1 HPV epidemiology • Most common viral sexually transmitted infection – MEN: oral cavity, penis, scrotum, urethra, anus – WOMEN: oral cavity, cervix, vagina, vulva, anus • Manifestation of infection depends on the site of infection and the genotype of HPV – Carcinogenic neoplasia and cancer of cervix, oropharynx, and anus; less likely to cause cancer at other sites, ex: HPV16 and 18 – Non-carcinogenic anogenital warts, ex: HPV6 and 11 Infection From Time of First Sexual Intercourse Study of female college students (N=603) 1 Cumulative Incidence of HPV Infection 0.8 0.6 0.4 0.2 0 0 4 8 12 16 20 24 28 32 36 40 44 48 52 56 Months Since First Intercourse From Winer RL, Lee S-K, Hughes JP, Adam DE, Kiviat NB, Koutsky LA. Genital human papillomavirus infection: incidence and risk factors in a cohort of female university students. Am J Epidemiol. 2003;157:218–226. Reprinted with the permission of Oxford University Press. Secondary Prevention: Interrupting disease progression In the right settings, cervical cancer can be prevented with a moderately sensitive test • Forgiving disease: long pre-clinical detectable phase • Almost all precancer can be treated when detected early • With regular screening, an insensitive test will detect all but the most rapidly developing cases! The Natural History of Disease Outcome Biologic Disease Symptoms Onset of detectable by Disease screening Preclinical phase Detectable preclinical phase Usual time of Diagnosis Screening for cervical cancer Screening for precursor lesions and HPV infections Frequent pap smear cytology HPV DNA testing (age 30+) Treatment of precursor lesions by excisional therapy is > 95% effective If Pap Smears successfully prevented cervical cancer in the mainstream U.S., why not use simply use them to screen everyone else? A. Three-visit cycle: 1. Cytology; 2. Colposcopy; 3. Treatment B. Cytology is an insensitive test (negative test = poor reassurance); repeat iterations (e.g., regular or annual Pap smears) are necessary. C. Cytology is a poorly reproducible (subjective) test and difficult to maintain performance D. Colposcopy is not as good as you think it is. E. Bottom Line: U.S. program costs billions of $ annually F. Tests with better test characteristics, less visits per cycle, fewer cycles per lifetime Incremental Etiologic Contributions of HPV types 120 % of Cancers 100 80 60 40 20 0 16 18 45 31 X 33 52 58 35 59 56 Cumulative Incidence of Cervical Precancer or Cancer (≥CIN3) after a Single Oncogenic HPV Test 25% Cumulative Incidence Rate of ≥CIN3 HPV+ HPV- 30 Years and Older, Cytologically Normal Women 20% 15% 10% 5% 0% 0.0 4.5 15.0 27.0 39.0 51.0 63.0 75.0 87.0 99.0 111.0 119.5 Follow-Up Time (Months) Portland Sherman et al., JNCI, 2003 HPV Test as Screening Cumulative Incidence Rate of ≥CIN3 25% HPV16+ 20% HPV18+ 15% 10% 5% HPV+ HPV+/ HPV16-/18- 0% 0.0 4.5 15.0 27.0 39.0 51.0 63.0 75.0 Follow-Up Time (months) Portland 87.0 99.0 111.0 119.5 HPV- Khan et al., JNCI, 2005 HPV testing • • • • Sensitive - 98% But not specific so high false+ rates • Useful for equivocal Pap results • Useful for certain ages Useful if can find persistent infection Useful for infrequent screening! Pap testing HPV testing Current Recommendations in US Women 30 Years and Older Cytology Negative Oncogenic HPV Testing Oncogenic HPV- Oncogenic HPV+ Repeat Tests in 3 Years Repeat Tests in 6--12 Months Concerns about Screening Using HPV Tests • Will the tests include the right types and proper thresholds for positivity? • Will screening be used incorrectly, e.g., among young women? • Will the companies act and price fairly? • Will the “winner” be the best test? • How can “home-brew” assays be qualitycontrolled or discouraged? Primary Prevention: Prophylactic vaccination Current Prophylactic Vaccines are Based on Purified Papillomavirus-Like Particles (VLPs) • Empty viral capsid composed of the L1 major virion protein • Non-infectious and non-oncogenic (Subunit vaccine) HPV VLP Vaccines Manufacturer Merck GlaxoSmithKline Trade name Gardasil® Cervarix™ HPV types 6,11,16,18 16,18 Theoretic coverage 70% of CxCa 90% GW 70% of CxCa Adjuvant Alum ASO4 (Alum+MPL) 0, 2, 6 months 0, 1, 6 months Dosing Schedule Phase III Trials--Main Results •Design: double blind, placebo controlled trials of >10,000 women •Main Finding: protection against cervical precancer caused by HPV16/18 among women naïve to these types during the vaccination period: VE ~100% •Tolerability: slightly more injection site pain than control vaccine •Immunogenicity: 99.5% seroconversion with titers 10-50 fold higher than natural infection •Safety: no evidence of elevated adverse events among vaccinees How durable is protection? Serum HPV16 antibody titers postvaccination plateau at levels higher than in natural infection Vaccinees Naturally infected Uninfected From Mao et al, Obstet Gynecol 107:18-27, 2006 Can the vaccine be used to treat existing infections and/or disease? Vaccine efficacy among women already infected with HPV16 or 18 • Vaccine efficacy against viral clearance: 2.5% (95% CI -9.8 to 13.5%) • HPV vaccination does not accelerate viral clearance among those infections • No evidence of a therapeutic effect of this vaccine Hildesheim et al. JAMA 2007; 298(7):743-53 Does the vaccine protect against other HPV types? (cross-protection) Efficacy Against 12 month persistent HPV infection Other High Risk HPV Types GSK HPV16/18 Vaccine ITT Analysis; ca. >700 per arm: 3.5 yr follow-up* HPV Type # Vaccine # Placebo Efficacy (95%CI) 45 10 27 63 (18-85) 31 21 102 79 (66-88) 33 31 50 52 150 143 5 (-34-18) 58 64 56 -15 (-71-23) 31 35 16 33 58 52 X X 34 18 45 39 68 38 (18-85) 26 51 30 53 56 66 High-risk HPV types *Paavonen et al, Lancet 2009 Does the vaccine protect against HPV infections at other anatomic sites? Burden of HPV in all cancers Site Attributable to HPV (%) Of which, HPV16/18 (%) Total Cancers Attributable to HPV % of all cancers Cervix 100 70 492,800 492,800 4.54 Penis 40 63 26,300 10,500 0.1 Vulva, vagina 40 80 40,000 16,000 0.15 Anus 90 92 30,400 27,300 0.25 Mouth 3 95 274,300 8200 0.08 Oropharynx 12 89 52,100 6200 0.06 10,862,500 561,100 5.17 All sites Adapted from Parkin and Bray, Vaccine 2006 Protection against other cancers • Vaginal/vulvar- 100% VE • Anal- studies on-going • Penile- high protection against penile precancer • Oral- no data yet Important considerations • ~ $375 for the 3 dose series in the USA • Requires a cold chain and 3 visits • It doesn’t protect women with past infection • Still have to screen for other carcinogenic types (~30% of cancer) How do we integrate HPV vaccination into current screening programs? (goal: maximize impact in a costeffective manner) Summary of Vaccine Findings in Young Women • High level prevention of precancerous cervical lesions when administered to females without infection by vaccine-type HPVs – Not therapeutic • Generally safe and well-tolerated – Cannot cause infection or cancer (b/c only contains 1 protein from each targeted type) • High antibody titers sustained for ~8 years • Additional results – High efficacy for prevention of HPV-related vaginal/extra-genital lesions Summary of questions that still need addressed • How long is the duration of protection (past 10 yrs)? • Will the vaccine protect against infections and disease at other anatomic sites? • Will the vaccine work in special populations (i.e.: HIV-positive)? • How should the vaccine be integrated into existing screening programs? • How should the vaccine be introduced in countries without screening programs?