* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Heart Rhythm Refresher Course 2014 Module 1: My Diagnostic
Cardiac contractility modulation wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Cardiovascular disease wikipedia , lookup
DiGeorge syndrome wikipedia , lookup
Turner syndrome wikipedia , lookup
Marfan syndrome wikipedia , lookup
Jatene procedure wikipedia , lookup
Down syndrome wikipedia , lookup
Cardiac surgery wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Heart arrhythmia wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Electrocardiography wikipedia , lookup
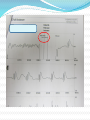
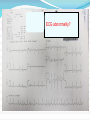
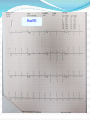
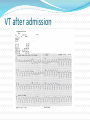
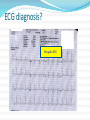
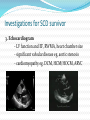
Dr Yue Chiu Sun Consultant and Head Division of Cardiology Dept of Medicine & Geriatrics United Christian Hospital 6 Apr 2014 Case 1 Mr. WWK age 51 Non-smoker, non-drinker PMH: psoriasis Mother diagnosed to have HOCM family screening previously recommended Presentation Played badminton after work -> sudden collapse Bystander CPR started Ambulance crew arrived VF At AED BP 133/92 P 120 bpm Given amiodarone infusion GCS 3/15 PERL Intubated for ventilatory support Consulted CCU-> admitted for further management ECG abnormality? At CCU Bedside Echo Fair echogenicity Overall satisfactory LV systolic function No apparent RWMA noted No LVH noted to suggest HCM or LVOT obstruction No pericardial effusion Seizure given sodium valproate 400 mg q8h iv Urgent CT brain: no SOL / haemorrhage What’s next step? Progress Consulted ICU Admitted ICU Therapeutic hypothermia sedation (dormicum, morphine) + muscle relaxant (atracurium) induction with 2.5L of 4°C ice cold saline aimed at body T 32-34°C; maintained with wet blankets continued rectal probe monitoring Inotropic support NG tube feeding started ECG : ST↑V2-3 and T V2-4 LDH 301 → 288; CK 395 → 932 TnT 33 → 1169 aspirin 160 mg daily PO started During Hypothermia Progress ECG : deep T over V2-6 (probable ACS : Anterior NSTEMI) Echo: - LV anteroseptal & apical hypokinesia; impaired LV function Re-warming with 0.25 °C per hr Reduced inotropic support gradually Extubated the next day -> transferred back to CCU To CCU Clopidogrel, Simvastatin, Metoprolol were added bedside echo: LVEF 50%, fair LV contraction; RWMA noted (similar to ICU findings) Fully alert, not in heart failure Coronary angiogram and PCI was performed R dominance, LM normal, LAD proximal 80%, mid LAD 70% stenosis, LCx mild disease, RCA normal Stenting to proximal and mid LAD Uneventful recovery and discharged home Post-PCI Anterior STEMI: Therapeutic Hypothermia and Primary PCI Post-Cardiac Arrest Care: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Recommend that comatose (ie, lack of meaningful response to verbal commands) adult patients with ROSC after out-ofhospital VF cardiac arrest should be cooled to 32°C to 34°C (89.6°F to 93.2°F) for 12 to 24 hours (Class I, LOE B) Induced hypothermia also may be considered for comatose adult patients with ROSC after in-hospital cardiac arrest of any initial rhythm or after out-of-hospital cardiac arrest with an initial rhythm of pulseless electric activity or asystole (Class IIb, LOE B) Circulation. 2010;122:S768-S786 Post–cardiac arrest care algorithm Peberdy M A et al. Circulation. 2010;122:S768-S786 Copyright © American Heart Association, Inc. All rights reserved. Case 2 Mr WWM age 52 History of idiopathic DCMP (CTCA-> normal coronary arteries) and AF Admitted for post-VT/VF arrest after being found collapse in street (pulseless VT/VF with prolonged resuscitation > 1 hr and repeated defibrillation); intubated for mechanical ventilation VT after admission Progress Acute renal and liver failure requiring temporary HD support Progressive improvement and extubated later Echo: poor LV function EF ~20% with global hypokinesia Coronary angiogram: mild CAD only ICD was implanted Transfer to convalescence hospital for rehabilitation, good neurological recovery Diagnostic Approach to SCD survivor 1. Clinical History eg. known IHD/congenital heart disease or past history of MI, CV risk profile, triggering factors (exercise, stress – eg. ACS, WPW, HCM, AS, CPVT ) 2. Family history of hereditary cardiac arrhythmia or congenital heart disease, FH of SCD 3. Transient and reversible causes 4. Evaluation for structural heart disease 5. Evaluation for those without structural heart disease Clinical History and Baseline Investigation Transient or reversible causes Evidence of ACS esp. STEMI Drug history: anti-arrhythmic drugs, psychiatric drugs, OTC drugs, herbal medicine, drug abuse Medications esp. those ↑ QTc Electrolyte and acid-base abnormalities: hypokalemia K, hyperkalemia ↑K, Ca, acidosis Investigations for SCD survivor 1. ECG - Myocardial ischemia or infarction (? ST elevation in V1 & aVR) - myocarditis (eg. T wave inversion, ST segment elevation or depression, heart block, ↑QT interval) - high grade AV block - Brugada Syndrome (polymorphic VT/VF) - Long QT or short QT - Wolf-Parkinson-White WPW syndrome esp. preexcited AF - CPVT, Early repolarization syndrome (ERS) ECG PR: short PR (WPW with preexcitation) Q wave: MI QRS: delta wave, BBB QT : congenital long QT, acquired QT : short QT ST : STEMI, myocarditis, Brugada syndrome ST : myocardial ischemia, NSTEMI, myocarditis T : tall T wave in K, hyperacute phase in AMI T : myocardial ischemia, NSTEMI, epsilon wave (ARVC) P and QRS relationship: high grade AV block J point elevation ECG diagnosis? Brugada ECG Brugada ECG Brugada syndrome: polymorphic VT/VF WPW with preexcited AF Early repolarization syndrome ERS ERS is defined as an elevation of the J point (the junction between the end of the QRS complex and the beginning of the ST segment) and/or ST segment by at least 0.1 mV from baseline Early repolarization syndrome ERS ERS can be divided into three subtypes: Type 1 - which is predominantly characterized by an ERP that is detected with lateral precordial leads, is prevalent among healthy male athletes and rarely seen in VF survivors Type 2 - which is predominantly detected through the inferior or inferolateral leads, is associated with a higher level of arrhythmia risk than type 1 ERS Type 3 - which involves ERPs that are observed globally through the inferior, lateral, and right precordial leads, is associated with the highest level of risk for malignant arrhythmias and often associated with VF storms Early repolarization syndrome ERS early repolarization in the inferior ECG leads has been associated with idiopathic VF and has been termed as ERS ERS has emerged as a marker of risk for idiopathic VF and sudden death. However, the incidental discovery of a J wave on routine screening should not be interpreted as a marker of “high risk” for sudden death Catecholaminergic Polymorphic VT (CPVT) Affected patients typically present with life-threatening VT or VF occurring during emotional or physical stress, with syncope often being the first manifestation of the disease. Although sporadic cases occur, this is a largely familial disease . The majority of known cases are due to mutations in the cardiac ryanodine receptor, which is the cardiac sarcoplasmic calcium release channel VT morphology may vary continuously, from beat to beat, or may appear as a bidirectional VT Congenital Long QT syndrome LQTS With 4 or more points the probability is high for LQTS, and with 1 point or less the probability is low. Two or 3 points indicates intermediate probability. QTc (Defined as QT interval / square root of RR interval) o >= 480 msec - 3 points o 460-470 msec - 2 points o 450 msec and male gender - 1 point Torsades de Pointes ventricular tachycardia - 2 points T wave alternans - 1 point Notched T wave in at least 3 leads - 1 point Low heart rate for age (children) - 0.5 points Syncope (one cannot receive points both for syncope and Torsades de pointes) o With stress - 2 points o Without stress - 1 point Congenital deafness - 0.5 points Family history (the same family member cannot be counted for LQTS and sudden death) o Other family members with definite LQTS - 1 point o Sudden death in immediate family (members before the age 30) - 0.5 points Long QT1 Congenital Long QT syndrome Syndrome Frequency Locus Protein (Functional Effect) KCNQ1 (LQT1) RWS, JLNS 40–55 11p15.5 Kv7.1 (↓) KCNH2 (LQT2) RWS 30–45 7q35–36 Kv11.1 (↓) SCN5A (LQT3) RWS 5–10 3p21–p24 NaV1.5 (↑) ANKB (LQT4) RWS <1% 4q25–q27 Ankyrin B (↓) KCNE1 (LQT5) RWS, JLNS <1% 21q22.1 MinK (↓) KCNE2 (LQT6) RWS <1% 21q22.1 MiRP1 (↓) KCNJ2 (LQT7) AS <1% 17q23 Kir2.1 (↓) CACNA1C (LQT8) TS <1% 12p13.3 L-type calcium channel (↑) CAV3 (LQT9) RWS <1% 3p25 Caveolin 3 (↓) SCN4B (LQT10) RWS <1% 11q23.3 Sodium channel-β4 (↓) AKAP9 (LQT11) RWS <1% 7q21–q22 Yotiao (↓) SNTA1 (LQT12) RWS <1% 20q11.2 Syntrophin α1 (↓) KCNJ5 (LQT13) RWS <1% 11q24 Kir3.4 (↓) Gene Short QT syndrome Genetic basis of SQTS QTc (msec) Locus Gene (cardiac ion channel) SQT 1 286 ± 6 11p15 KCNH2 (IKr) ↑ SQT 2 302 7q35 KCNQ1 (IKs) ↑ SQT 3 315-330 17q23 KCNJ2 (IK1) ↑ SQT 4 331-370 10p12 CACNB2b (ICa) ↓ SQT 5 346-360 12p13 CACNA1C (ICa) ↓ SQT 6 330 7q21-22 CACNA2D1 (ICa) ↓ Investigations for SCD survivor 2. Blood tests eg. RFT, ABG, cardiac biomarkers, Ca, Mg, drug level (digoxin, TCA etc.), toxicology screen Investigations for SCD survivor 3. Echocardiogram - LV function and EF, RWMA, heart chamber size - significant valvular disease eg. aortic stenosis - cardiomyopathy eg. DCM, HCM/HOCM, ARVC Investigations for SCD survivor 4. Coronary angiography and cardiac catheterization - r/o significant coronary artery disease, anomalous coronary arteries (? coronary artery course between aorta & pulmonary trunk), congenital heart disease 5. CT scan - aortic dissestion, coronary anomaly, congenital heart disease 6. Cardiac MRI - structural heart disease eg. HCM, ARVC, LV non-compaction Investigations for SCD survivor 7. EP study (limited role : post-cardiac arrest) - exclude other arrhythmias, ablative therapy (?WPW, VT foci) 8. Genetic testing, family screening and counselling eg. long QT, HCM, Brugada syndrome, CPVT - commercially available genetic testing exists for HCM, DCM, ARVC, LV non-compaction, LQTS, SQTS, CPVT, and BrS Genetic Testing for Potentially Lethal, Highly Treatable Inherited Cardiomyopathies/Channelopathies in Clinical Practice David J. Tester and Michael J. Ackerman Circulation. 2011;123:1021-1037 Indications for genetic testing. Provided is a table of possible indications for genetic testing for hypertrophic cardiomyopathy (HCM), long-QT syndrome (LQTS), catecholaminergic polymorphic ventricular tachycardia (CPVT), and Brugada syndrome (BrS). LVH indicates left ventricular hypertrophy; QTc, corrected QT interval; TdP, torsade de pointes; Neg., negative; and SUD, sudden unexplained death. The plus symbol ( ) represents a positive indication for genetic testing; the negative symbol ( ) represents an indicator that does not warrant genetic testing for the specific disorder; and the / symbol represents an indicator that may or may not warrant genetic testing. Utility of genetic testing. Shown is the current diagnostic, prognostic, and therapeutic utility of genetic testing for hypertrophic cardiomyopathy (HCM), long-QT syndrome (LQTS), catecholaminergic polymorphic ventricular tachycardia (CPVT), and Brugada syndrome (BrS). The plus symbol ( ) indicates the test has utility, the negative symbol ( ) indicates no current measurable utility, and the / symbol indicates the test may have some utility